Autologous Chondrocyte Implantation for Talar Osteochondral Lesions: An Intraoperative Masterclass

Key Takeaway
This masterclass provides an immersive, detailed guide to Autologous Chondrocyte Implantation (ACI) for symptomatic osteochondral lesions of the talus (OLTs). We'll cover patient selection, comprehensive surgical anatomy, meticulous intraoperative execution from chondrocyte harvest to implantation, critical pearls, and comprehensive postoperative management, ensuring fellows gain a deep understanding of this advanced cartilage repair technique.
Comprehensive Introduction and Patho-Epidemiology
The management of osteochondral lesions of the talus (OLTs) remains one of the most formidable challenges in modern orthopedic sports medicine and foot and ankle surgery. The talus, uniquely devoid of muscular attachments and covered by articular cartilage over approximately 70% of its surface area, operates within a highly congruent joint subjected to immense biomechanical forces. When the articular surface is compromised, the intrinsic healing capacity of the hyaline cartilage is notoriously poor due to its avascular, aneural, and alymphatic nature. Autologous Chondrocyte Implantation (ACI), and its subsequent iterations including Matrix-Induced Autologous Chondrocyte Implantation (MACI), represents a sophisticated, restorative biologic intervention. Unlike marrow stimulation techniques (e.g., microfracture) that yield structurally inferior fibrocartilage (predominantly Type I collagen), ACI is designed to regenerate a durable, hyaline-like cartilage repair tissue (predominantly Type II collagen) capable of withstanding the rigorous shear and compressive loads of the tibiotalar joint.
To fully appreciate the utility of ACI, one must understand the patho-epidemiology of OLTs. These lesions typically arise from two primary etiologies: acute traumatic shear forces and osteochondritis dissecans (OCD). Traumatic injuries often involve a single, high-energy inversion or eversion event leading to a localized impaction or shear fracture of the chondral surface and underlying subchondral bone plate. Conversely, OCD is classically observed in adolescents and young adults, characterized by a localized ischemic necrosis of the subchondral bone, secondary to repetitive microtrauma or an idiopathic vascular watershed phenomenon. Over time, the overlying cartilage, deprived of its mechanical subchondral support, softens, fibrillates, and ultimately detaches.
While degenerative, "wear-and-tear" lesions do occur, ACI is strictly preserved for focal, contained defects rather than diffuse osteoarthritic disease. The natural history of an untreated, symptomatic, full-thickness OLT larger than 1.5 to 2.0 cm² is a predictable progression toward early-onset osteoarthritis. Marrow stimulation techniques have proven highly efficacious for smaller lesions, but longitudinal outcome studies demonstrate a precipitous decline in clinical success for lesions exceeding this critical size threshold, as the fibrocartilaginous repair tissue degrades under cyclic loading. Therefore, ACI emerges as the definitive salvage—and increasingly, the primary—procedure for large, symptomatic, full-thickness chondral and osteochondral defects of the talus.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of ankle anatomy and biomechanics is the cornerstone of successful ACI. The ankle joint is a highly constrained, modified hinge joint. Unlike the knee, which relies heavily on ligamentous structures for stability, the ankle derives significant inherent stability from its osseous architecture—the mortise. The talar dome is wider anteriorly than posteriorly, meaning dorsiflexion tightly wedges the talus within the mortise, maximizing congruency and stability. The contact area of the ankle joint is significantly smaller than that of the knee or hip, yet it routinely transmits forces equivalent to three to five times body weight during normal gait, and up to ten times during athletic activities. Consequently, the articular cartilage of the talus is subjected to extraordinary contact pressures.
The topography of OLTs follows a predictable, albeit not absolute, pattern characterized by the mnemonic "DIAL a PIMP" (Dorsiflexion/Inversion = Anterior Lateral; Plantarflexion/Inversion = Posterior Medial). Approximately 62% of lesions occur on the medial talar dome. These are typically posterior, deep, cup-shaped, and morphologically consistent with OCD. Conversely, 34% are located on the lateral talar dome; these are usually anterior, shallow, wafer-shaped, and strongly associated with acute trauma. Central lesions are exceedingly rare, accounting for less than 5% of cases.
Surgical access to these lesions requires meticulous navigation through complex neurovascular corridors. Medial approaches, often necessitating a medial malleolar osteotomy for adequate perpendicular access to the lesion, place the posterior tibial tendon (PTT) and the neurovascular bundle (tibial nerve, posterior tibial artery) at significant risk. The bundle lies immediately posterior to the PTT and must be protected with malleable retractors during osteotomy completion. Anterior and anterolateral arthroscopic portals, utilized during the Stage 1 harvest, imperil the superficial peroneal nerve (SPN). The SPN demonstrates highly variable branching patterns as it crosses the ankle joint, necessitating careful superficial dissection—often utilizing a "nick and spread" technique—to avoid devastating iatrogenic neuromas. Lateral approaches for lateral OLTs may require fibular osteotomies or extensive ligamentous releases (ATFL, CFL), placing the sural nerve and peroneal tendons at risk. Furthermore, the tenuous blood supply of the talus, derived primarily from the artery of the tarsal canal, the artery of the sinus tarsi, and deltoid branches, must be respected to prevent iatrogenic avascular necrosis, particularly when performing extensive subchondral debridement or bone grafting.
Exhaustive Indications and Contraindications
Patient selection is arguably the most critical determinant of success in ACI. This procedure demands a highly compliant patient, a pristine biological environment, and strict adherence to specific morphological criteria. The ideal candidate is a young, biologically active individual with a focal, contained, full-thickness chondral defect who has exhausted conservative measures and, often, prior marrow stimulation procedures.
The primary indication for ACI in the talus is a symptomatic, full-thickness osteochondral lesion (arthroscopic Grade III or IV) measuring greater than 2.0 cm². While primary ACI can be considered for these massive lesions, it is most frequently utilized as a secondary, salvage procedure following failed microfracture. Age is a crucial consideration; while chronological age is less absolute than physiological age, patients between 18 and 50 years exhibit the most robust chondrogenic potential. Pediatric patients with open physes are generally managed with conservative care or alternative biologic interventions, while patients over 50 often exhibit diminished cellular proliferation rates and a higher propensity for concomitant degenerative joint disease.
Contraindications must be rigorously respected to prevent catastrophic failure. "Kissing lesions" (bipolar disease involving both the talus and the tibial plafond) represent an absolute contraindication, as the altered biomechanics and sheer stress will inevitably destroy the delicate chondrocyte graft. Uncorrected axial malalignment (varus or valgus hindfoot) and uncorrected ligamentous instability are similarly prohibitive; these mechanical derangements must be addressed either prior to or concurrently with the ACI procedure. Diffuse osteoarthritis, inflammatory arthropathies, and active joint infections are absolute contraindications.
| Category | Specific Criteria for Talar ACI |
|---|---|
| Primary Indications | Symptomatic full-thickness OLT > 2.0 cm²; Failed prior marrow stimulation (microfracture); Hepple Stage V / Ferkel Stage I/IIa (with concurrent bone grafting). |
| Ideal Patient Profile | Age 18–50 years; BMI < 30; Non-smoker; Highly compliant with complex, prolonged rehabilitation protocols. |
| Absolute Contraindications | Diffuse tibiotalar osteoarthritis; "Kissing lesions" (tibial plafond involvement); Active infection; Inflammatory arthropathy (e.g., Rheumatoid Arthritis); Uncorrected malalignment or instability. |
| Relative Contraindications | Age > 50 years (assessed on a case-by-case basis regarding biological age); Subchondral cysts > 10mm deep requiring massive bone grafting (though manageable with a "sandwich" technique); Heavy smoking history. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock of a flawless intraoperative execution. The clinical examination must be comprehensive, extending beyond the ankle joint to assess the entire lower extremity kinetic chain. The surgeon must evaluate for subtle hindfoot varus or valgus, which may necessitate a concurrent calcaneal osteotomy. Ligamentous integrity is assessed via the anterior drawer and talar tilt tests; chronic lateral ankle instability is a frequent concomitant pathology that must be addressed via a Broström-Gould repair or lateral ligament reconstruction to protect the nascent cartilage graft from abnormal shear forces.
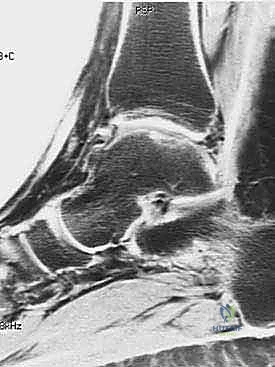
Advanced imaging is non-negotiable. While weight-bearing plain radiographs (AP, Mortise, Lateral) are obtained to rule out diffuse joint space narrowing and assess overall osseous alignment, Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the articular cartilage and subchondral bone. Fluid-sensitive sequences (T2-weighted with fat suppression or STIR) exquisitely detail the extent of cartilage loss, subchondral edema, and the presence of fluid undermining an unstable osteochondral fragment.
When subchondral cysts are identified on MRI (Hepple Stage V), a fine-cut Computed Tomography (CT) scan is mandatory. CT provides unparalleled spatial resolution of the osseous architecture, allowing the surgeon to precisely quantify the volumetric dimensions of the cyst. This dictates the necessity and volume of autologous bone grafting required during Stage 2. The Ferkel and Sgaglione CT classification system is instrumental here, guiding the surgical approach and the extent of subchondral preparation needed.
Patient positioning for both stages requires meticulous attention to detail. For Stage 1 (Arthroscopic Harvest), the patient is positioned supine. A thigh tourniquet is utilized to provide a bloodless field, optimizing arthroscopic visualization. For Stage 2 (Implantation), the patient is also positioned supine, but the use of a vacuum mattress is highly recommended.
The vacuum mattress allows the surgeon to dynamically position the operative leg—securing it in internal rotation for medial approaches or external rotation for lateral approaches—without the need for an assistant to hold the limb for prolonged periods. A bump is placed under the ipsilateral hip to correct natural external rotation if a medial approach is planned. Prophylactic intravenous antibiotics are administered prior to tourniquet inflation.
Step-by-Step Surgical Approach and Fixation Technique
Stage 1: Arthroscopic Diagnostic Evaluation and Chondrocyte Harvest
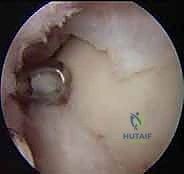
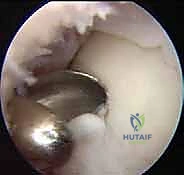
The initial stage is a minimally invasive arthroscopic procedure aimed at confirming the diagnosis, assessing the lesion's suitability for ACI, and harvesting healthy chondrocytes. Standard anteromedial and anterolateral portals are established. The anterolateral portal requires extreme caution; superficial transillumination and blunt dissection are utilized to avoid the superficial peroneal nerve. Upon joint entry, a systematic diagnostic sweep is performed. The OLT is probed to determine its size, stability, and the quality of the surrounding cartilage borders.
Once ACI is deemed appropriate, the harvest is performed. The goal is to obtain 200 to 300 milligrams of pristine, full-thickness articular cartilage from a strictly non-weight-bearing region. In the ankle, the anterior non-articulating aspect of the talar dome is the preferred donor site. Alternatively, if the detached OLT fragment contains viable hyaline cartilage, it can be utilized, thereby eliminating donor site morbidity. Using a sharp ring curette or arthroscopic biopsy forceps, the cartilage is harvested down to, but meticulously avoiding penetration of, the subchondral bone plate. The harvested tissue is immediately transferred into a sterile, specialized transport medium provided by the cell-culturing facility. Strict adherence to chain-of-custody protocols is mandatory. The cells are subsequently expanded in vitro over 3 to 6 weeks.
Stage 2: Open Implantation and Defect Preparation
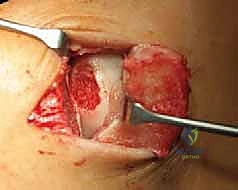
The definitive implantation procedure demands an open approach to achieve perpendicular access to the lesion. For the majority of lesions (medial talar dome), a medial malleolar osteotomy is required. An incision is made over the medial malleolus, curving distally along the line of the posterior tibial tendon. The periosteum is incised, and the proposed osteotomy site is marked. We favor a chevron-type or step-cut osteotomy over a simple transverse cut, as it provides superior rotational stability and increases the surface area for osseous healing. Prior to completing the osteotomy, two parallel drill holes are created using a 2.5mm drill bit, and tapped for 4.0mm partially threaded cancellous screws. This pre-drilling ensures perfect anatomic reduction during closure. The osteotomy is then completed using an oscillating saw and an osteotome, meticulously protecting the posterior tibial tendon and neurovascular structures with a retractor. The medial malleolus is reflected inferiorly, exposing the talar dome.
Lesion preparation is the most critical technical step. The defect is radically debrided using sharp curettes to remove all fibrous tissue, necrotic cartilage, and the sclerotic subchondral bone base. The goal is to create a contained defect with perfectly vertical, healthy hyaline cartilage walls—the "ring of fire." The subchondral bone is debrided until punctate bleeding is observed, signifying a viable vascular bed. However, aggressive penetration into the deeper cancellous bone must be avoided, as this invites the influx of marrow-derived mesenchymal stem cells, which will produce inferior fibrocartilage and compromise the ACI construct.
If a deep subchondral cyst is present (Hepple Stage V), it is thoroughly curetted. The resulting void must be filled with autologous cancellous bone graft, typically harvested from the ipsilateral distal tibia or calcaneus. The bone graft is tamped into the defect to reconstitute the subchondral bone plate, leveling it flush with the surrounding native subchondral bone. This is known as the "sandwich technique."
Membrane Templating, Fixation, and Cell Delivery
Once the bed is prepared, a sterile aluminum foil template is pressed into the defect to capture its exact dimensions. This template is then used to cut the covering membrane. Historically, an autologous periosteal patch harvested from the proximal tibia was utilized (first-generation ACI). However, due to high rates of graft hypertrophy, contemporary techniques almost exclusively utilize a synthetic Type I/III collagen bilayer membrane (second-generation ACI) or a matrix seeded with the cultured chondrocytes (third-generation MACI).
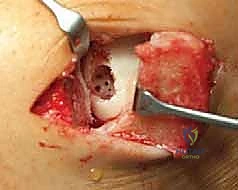
If using a liquid cell suspension (second-generation), the collagen membrane is sutured over the defect. Using 6-0 Vicryl or PDS on a small spatulated needle, the membrane is secured to the surrounding healthy cartilage with interrupted sutures spaced 1 to 2 millimeters apart. The knot must be buried to avoid mechanical irritation. A small opening is left at the superior pole.

The suture line is then sealed with commercial fibrin glue to ensure a watertight construct. The cultured chondrocytes, suspended in a liquid medium, are injected beneath the membrane through the small opening using a flexible catheter. The final opening is then sutured and sealed with fibrin glue.
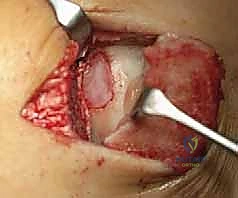
If utilizing MACI (third-generation), the technique is significantly streamlined. The cultured chondrocytes are already seeded within the collagen matrix. The membrane is simply cut to the templated size and secured within the defect using fibrin glue alone, or occasionally supplemented with a few fine sutures. This eliminates the technically demanding watertight suturing and the risk of liquid cell leakage. Following implantation, the joint is put through a gentle range of motion to ensure graft stability and the absence of impingement. The medial malleolar osteotomy is anatomically reduced and secured with the previously drilled 4.0mm screws. The wound is closed in layers, and the limb is placed in a well-padded short leg splint in neutral dorsiflexion.
Complications, Incidence Rates, and Salvage Management
While ACI offers transformative outcomes, it is a technically demanding procedure with a distinct complication profile. The surgeon must be prepared to identify and manage these complications to prevent ultimate failure of the reconstructive effort. Complications can be broadly categorized into graft-related issues, approach-related morbidity, and general surgical risks.
Historically, the most common complication associated with first-generation (periosteal) ACI was symptomatic graft hypertrophy, occurring in up to 25-30% of cases. The periosteal patch, driven by intra-articular growth factors, would overgrow, causing painful catching and impingement. This necessitated a secondary arthroscopic debridement (shaving) of the hypertrophic tissue. The advent of collagen membranes (cACI) and matrix-induced ACI (MACI) has drastically reduced this complication rate to less than 5%.
Graft delamination or failure of integration is a catastrophic complication, often resulting from inadequate subchondral bed preparation, failure to achieve vertical cartilage walls, or premature weight-bearing. If delamination occurs, salvage options are limited and typically involve osteochondral autograft transfer (OATS) or, in older patients, progression to arthrodesis or total ankle arthroplasty.
Approach-related complications primarily involve the medial malleolar osteotomy. Nonunion or delayed union occurs in 1-3% of cases. Meticulous surgical technique, avoiding thermal necrosis during the saw cut, and rigid internal fixation are paramount to mitigating this risk. Iatrogenic nerve injury (SPN during arthroscopy, tibial nerve during osteotomy) is a devastating complication that underscores the necessity of precise anatomic dissection.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Graft Hypertrophy | < 5% (with MACI/cACI); 25-30% (historical pACI) | Biologic overactivity of periosteum; uneven membrane tension. | Arthroscopic debridement/shaving of hypertrophic tissue. |
| Graft Delamination | 3 - 7% | Poor defect preparation; inadequate fixation; premature weight-bearing; uncorrected instability. | Revision ACI (rare); OATS procedure; Arthrodesis; Arthroplasty. |
| Osteotomy Nonunion | 1 - 3% | Thermal necrosis; inadequate fixation; smoking; poor bone quality. | Revision open reduction internal fixation (ORIF) with bone grafting. |
| Nerve Injury (SPN, Tibial) | < 1% | Aberrant anatomy; aggressive retraction; poor portal placement. | Neuroma excision; nerve grafting (rarely successful for SPN). |
| Deep Vein Thrombosis (DVT) | 1 - 2% | Prolonged immobilization; tourniquet use; genetic predisposition. | Therapeutic anticoagulation protocol. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following ACI is as critical as the surgical execution itself. The protocol is heavily dictated by the biology of chondrocyte maturation. The nascent graft requires mechanical stimulation to differentiate into load-bearing hyaline cartilage, yet it is highly susceptible to shear forces and excessive compression during the early phases of healing. The rehabilitation is divided into four distinct, biologically driven phases.
Phase 1: Protection and Early Motion (Weeks 0 - 6)
The primary goal is graft protection and the prevention of intra-articular adhesions. The patient is strictly Non-Weight-Bearing (NWB). The initial splint is removed at 10-14 days for suture removal. The patient is then transitioned to a removable controlled ankle motion (CAM) boot. Continuous Passive Motion (CPM) is initiated immediately, typically for 4 to 6 hours daily. CPM provides the essential hydrodynamic stimulation required for chondrocyte metabolism and matrix synthesis without subjecting the graft to compressive loads. Active-assisted range of motion (ROM) in the sagittal plane (dorsiflexion/plantarflexion) is encouraged, but inversion/eversion is strictly avoided to prevent shear stress on the graft and tension on the malleolar osteotomy.
Phase 2: Progressive Loading (Weeks 6 - 12)
Radiographic healing of the medial malleolar osteotomy must be confirmed before progressing. Once cleared, the patient begins a meticulously graduated Partial Weight-Bearing (PWB) protocol. This typically starts at 20% of body weight, increasing by 20% weekly, utilizing crutches and the CAM boot. Aquatic therapy is highly beneficial during this phase, utilizing the buoyancy of water to facilitate gait mechanics with reduced joint contact forces. Stationary cycling with low resistance can be initiated.
Phase 3: Strengthening and Proprioception (Months 3 - 6)
The patient transitions to Full Weight-Bearing (FWB) in standard footwear. The focus shifts to restoring muscular strength, endurance, and neuromuscular control. Closed kinetic chain exercises, proprioceptive training (e.g., BAPS board, single-leg stance), and progressive resistance exercises for the gastroc-soleus complex, tibialis anterior, and peroneal musculature are heavily emphasized. The cartilage graft is maturing, but high-impact activities are still prohibited.
Phase 4: Return to Sport (Months 6 - 12+)
The final phase involves sports-specific functional training. Progression to running, jumping, and cutting maneuvers is initiated only when the patient demonstrates full, pain-free ROM, isometric strength within 90% of the contralateral limb, and normal functional movement patterns. MRI evaluation (often utilizing T2 mapping or dGEMRIC sequences to assess glycosaminoglycan content) may be utilized at the 12-month mark to confirm structural integration and maturation of the repair tissue before clearing an elite athlete for full competition.
Summary of Landmark Literature and Clinical Guidelines
The evolution of ACI in the ankle is built upon a foundation of rigorous scientific inquiry and clinical trials. The pioneering work of Brittberg et al. in 1994, initially describing ACI in the knee, established the biologic plausibility of utilizing cultured autologous chondrocytes to regenerate hyaline cartilage. The extrapolation of this technology to the talus was spearheaded by Giannini and colleagues in the late 1990s and early 2000s. Giannini's early series demonstrated that ACI could yield excellent clinical outcomes and durable, hyaline-like repair tissue in the ankle, even in the setting of massive, cystic lesions.
The literature reflects a clear evolutionary trajectory from first-generation periosteal ACI (pACI) to second-generation collagen-covered ACI (cACI), and currently, third-generation Matrix-Induced ACI (MACI). Studies by Niemeyer et al. and others have definitively shown that cACI and MACI significantly reduce the incidence of graft hypertrophy and the need for subsequent reoperation compared to pACI, while maintaining or exceeding the clinical efficacy.
Current clinical guidelines from major orthopedic societies (such as the American Orthopaedic Foot & Ankle Society - AOFAS, and the International Cartilage Regeneration & Joint Preservation Society - ICRS) position ACI as a second-line therapy for lesions < 2.0 cm² that have failed marrow stimulation, and as a viable first-line option for primary lesions > 2.0 cm² or those with significant subchondral cystic involvement. While the regulatory landscape (e.g., FDA approval status for specific anatomic joints) continues to evolve, the vast body of peer-reviewed literature supports ACI as the premier biologic restorative procedure for complex osteochondral lesions of the talus, offering patients a definitive pathway to joint preservation and functional restoration.
