Arthroscopic Suprascapular Nerve Release: A Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on arthroscopic suprascapular nerve release. We'll meticulously cover patient selection, detailed surgical anatomy, precise intraoperative steps from portal placement to nerve decompression, and crucial pearls to ensure optimal outcomes. Learn to navigate the suprascapular and spinoglenoid notches, identify vital neurovascular structures, and manage potential complications for this challenging procedure.
Comprehensive Introduction and Patho-Epidemiology
Defining the Pathology of Suprascapular Nerve Entrapment
Suprascapular nerve entrapment is a complex, insidious, and frequently debilitating peripheral neuropathy that presents a unique diagnostic and therapeutic challenge to the orthopedic surgeon. Historically underdiagnosed, this condition is characterized by the mechanical compression or dynamic traction of the suprascapular nerve as it traverses the narrow fibro-osseous boundaries of the shoulder girdle. This entrapment fundamentally disrupts the axoplasmic flow and microvascular perfusion of the nerve, leading to a cascade of focal demyelination, subsequent axonal degeneration, and profound denervation of the supraspinatus and infraspinatus musculature. The resulting clinical picture is not merely one of localized pain, but of profound, progressive weakness and irreversible muscle atrophy that significantly compromises glenohumeral kinematics and the patient's overall quality of life.
The pathology typically manifests as a focal constriction at one of three critical anatomical junctures, each presenting distinct biomechanical and structural challenges. The most frequent site of compression is the suprascapular notch, where the nerve is tethered beneath the transverse scapular ligament (TSL). Less frequently, entrapment occurs within the supraspinatus fossa itself, often secondary to a space-occupying lesion such as a paralabral ganglion cyst originating from a posterosuperior glenoid labral tear. Finally, the nerve may be compromised at the spinoglenoid notch as it sharply navigates around the lateral margin of the scapular spine, a site particularly vulnerable to traction injuries in overhead athletes or compression by a hypertrophied spinoglenoid ligament.
Understanding this pathology requires an appreciation of the dynamic interplay between scapular dyskinesia, glenohumeral instability, and intrinsic nerve compliance. When conservative measures—including targeted physical therapy, non-steroidal anti-inflammatory medications, and image-guided perineural injections—fail to arrest the progression of denervation or alleviate intractable pain, surgical intervention becomes paramount. The advent of advanced arthroscopic techniques has revolutionized our approach, offering a precise, highly magnified, and minimally invasive solution to decompress the nerve while simultaneously addressing concomitant intra-articular pathology.
Epidemiological Considerations and Patient Demographics
The epidemiological profile of suprascapular nerve entrapment is diverse, reflecting the multifactorial etiology of the condition. While historically considered a rare clinical entity, advancements in diagnostic imaging and heightened clinical suspicion have revealed a significantly higher prevalence, particularly among specific athletic and occupational cohorts. It is now recognized as a leading cause of isolated shoulder pain and weakness in overhead athletes, including elite volleyball players, baseball pitchers, tennis players, and swimmers. In these populations, the repetitive, high-velocity eccentric loading of the posterior cuff, combined with extreme ranges of external rotation and abduction, induces microtrauma and dynamic traction on the nerve at the spinoglenoid notch.
Beyond the athletic population, suprascapular neuropathy is frequently encountered in the aging demographic, often presenting concomitantly with massive, retracted rotator cuff tears. In these scenarios, the medial retraction of the supraspinatus tendon exerts secondary traction on the motor branches of the suprascapular nerve, a phenomenon termed the "sling effect." This tethering exacerbates the degenerative process and complicates the surgical management of the rotator cuff tear, as the compromised neural drive significantly impairs postoperative rehabilitation and functional recovery. Furthermore, laborers engaged in heavy, repetitive overhead lifting are also at an elevated risk due to chronic microtrauma and subsequent fibrotic thickening of the transverse scapular ligament.
Interestingly, anatomical variants play a crucial role in the epidemiological distribution of this entrapment neuropathy. Morphological studies of the scapula have identified various shapes of the suprascapular notch, ranging from a wide, shallow "U" shape to a narrow, deep "V" shape, or even a completely ossified foramen. Patients possessing a V-shaped notch or a completely ossified transverse scapular ligament are genetically predisposed to entrapment, as the inherent reduction in cross-sectional area leaves zero tolerance for transient edema or hypertrophic changes. Recognizing these demographic and anatomical predispositions is critical for maintaining a high index of suspicion during the initial clinical evaluation.
Pathogenesis and the Natural History of Nerve Compression
The pathogenesis of suprascapular nerve entrapment is best understood through the lens of the "double-crush" or "multiple-crush" hypothesis, wherein the nerve is subjected to a combination of static compression and dynamic tension. At the suprascapular notch, static compression is frequently the primary culprit. Trauma, localized hematoma, or chronic inflammatory changes can lead to the fibrosis and hypertrophy of the transverse scapular ligament. As the fibro-osseous space narrows, the epineurial microvasculature is compromised, leading to intraneural edema, localized ischemia, and a localized conduction block (neurapraxia). If the compression persists, it progresses to axonotmesis, characterized by Wallerian degeneration distal to the site of entrapment.
Conversely, dynamic traction is the predominant pathomechanical force at the spinoglenoid notch. During the late cocking and early acceleration phases of throwing, the scapula undergoes extreme protraction and the humerus is maximally externally rotated. This specific kinematic chain forces the suprascapular nerve to stretch acutely against the rigid fulcrum of the spinoglenoid ligament and the bony margin of the scapular spine. Over time, this repetitive stretch-induced trauma exceeds the nerve's viscoelastic limits, resulting in intraneural fibrosis, reduced axonal transport, and eventual denervation of the infraspinatus. This isolated infraspinatus atrophy is pathognomonic for spinoglenoid notch entrapment and is a classic finding in elite volleyball attackers.
The natural history of untreated suprascapular nerve entrapment is generally one of progressive functional decline. While transient neurapraxia secondary to acute, blunt trauma may resolve spontaneously with rest and rehabilitation, chronic entrapment rarely demonstrates spontaneous recovery. The continuous denervation leads to irreversible fatty infiltration and profound atrophy of the supraspinatus and infraspinatus muscles. This loss of dynamic stabilization alters glenohumeral arthrokinematics, leading to superior humeral head migration, secondary subacromial impingement, and accelerated joint degeneration. Therefore, early recognition and timely surgical decompression are imperative to halt the progression of neuropathy and maximize the potential for motor endplate reinnervation.
Detailed Surgical Anatomy and Biomechanics
Origin, Course, and the Neural Highway
A profound, three-dimensional mastery of the suprascapular nerve's anatomy is the absolute prerequisite for safe and effective arthroscopic decompression. The suprascapular nerve is a mixed peripheral nerve carrying both motor and sensory fibers. It originates primarily from the ventral rami of the fifth and sixth cervical nerve roots (C5, C6), forming the upper trunk of the brachial plexus. In approximately 50% of individuals, it receives a variable contribution from the fourth cervical nerve root (C4). The nerve courses laterally and inferiorly through the posterior triangle of the neck, running deep to the trapezius and the omohyoid muscles, before plunging into the suprascapular region.
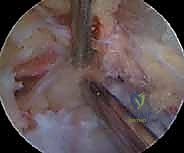
As it approaches the superior border of the scapula, the nerve travels along the "neural highway," a trajectory that guides it toward the suprascapular notch. During this descent, the nerve is accompanied by the suprascapular artery and vein. However, a critical anatomical divergence occurs just proximal to the notch. The suprascapular artery and vein typically elevate to pass superior to the transverse scapular ligament, while the suprascapular nerve dives deep to pass inferior to the ligament, directly through the bony notch. This anatomical relationship is famously memorized by surgical trainees with the mnemonic: "Army (Artery) goes over the bridge, Navy (Nerve) goes under the bridge."
Understanding this specific neurovascular divergence is critical during arthroscopic dissection. The surgeon must meticulously isolate the transverse scapular ligament from the overlying vascular bundle before initiating the release. Inadvertent injury to the suprascapular artery during ligamentous transection results in catastrophic hemorrhage that rapidly obscures the arthroscopic visual field, necessitating immediate conversion to an open procedure and risking significant morbidity. Furthermore, aberrant anatomical variations, such as the nerve passing superior to the ligament or the artery passing inferior to it, occur in a small percentage of the population, demanding meticulous, millimeter-by-millimeter dissection.
The Suprascapular Notch and Transverse Scapular Ligament
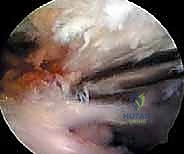
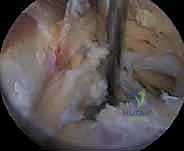
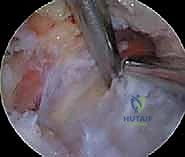
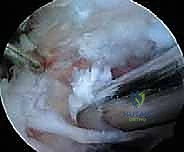
The suprascapular notch represents the primary anatomical bottleneck for the suprascapular nerve. Located on the superior border of the scapula, just medial to the base of the coracoid process, the notch is transformed into a rigid fibro-osseous foramen by the superior transverse scapular ligament (TSL). The morphology of this notch is highly variable. Rengachary's classification system defines six distinct types of notches, ranging from a wide, shallow depression (Type I) to a deep, V-shaped notch (Type IV), and ultimately to a completely ossified ligament forming a bony foramen (Type VI). Types IV, V, and VI are intrinsically predisposed to entrapment due to their severely restricted cross-sectional area.
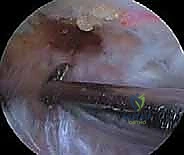
The superior transverse scapular ligament itself is a dense, fibrous band that bridges the notch. It is not merely a static structure but serves as an attachment site for the omohyoid muscle medially. During arthroscopic dissection, the TSL must be definitively identified and completely transected to achieve adequate decompression. The ligament is often obscured by a layer of fibrofatty tissue and the overlying coracoclavicular and coracohumeral ligaments. The surgeon must carefully navigate this soft tissue envelope, using the medial border of the coracoid base as the primary osseous landmark to guide the approach to the TSL.
Once the nerve traverses the suprascapular notch, it enters the supraspinatus fossa. Here, it immediately gives off two distinct branches. The first is a sensory branch that travels laterally to provide nociceptive and proprioceptive innervation to the coracoclavicular ligament, the subacromial bursa, and the posterior capsule of the glenohumeral joint. The second is a robust motor branch that arborizes extensively to innervate the supraspinatus muscle. The main trunk of the nerve then continues its journey, tethered to the bony floor of the supraspinatus fossa, coursing laterally and posteriorly toward the spinoglenoid notch.
The Spinoglenoid Notch and Distal Arborization
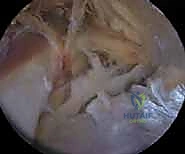
The spinoglenoid notch constitutes the second major site of potential entrapment. This anatomical juncture is defined medially by the lateral border of the scapular spine and laterally by the posterosuperior aspect of the glenoid neck. As the suprascapular nerve reaches this point, it must execute a sharp, nearly 90-degree turn around the base of the scapular spine to transition from the supraspinatus fossa into the infraspinatus fossa. This acute angulation renders the nerve highly susceptible to dynamic traction and friction, particularly during the extreme ranges of motion demanded by overhead throwing sports.
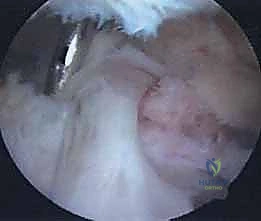
Bridging the spinoglenoid notch is the spinoglenoid ligament (inferior transverse scapular ligament). Unlike the robust TSL at the suprascapular notch, the spinoglenoid ligament is morphologically inconsistent. It may present as a distinct, thick fibrous band, a thin aponeurotic membrane, or it may be entirely absent in up to 20% of the population. When present and hypertrophied, it creates a secondary fibro-osseous tunnel that tightly constricts the nerve. Furthermore, the close proximity of the spinoglenoid notch to the posterosuperior glenoid labrum makes it the classic location for paralabral ganglion cysts, which extrude from labral tears and mechanically compress the nerve against the bony floor of the notch.
After navigating the spinoglenoid notch, the nerve enters the infraspinatus fossa. Here, it branches extensively, typically dividing into two to four terminal motor branches that penetrate the deep surface of the infraspinatus muscle. These terminal branches are responsible for the entire motor innervation of the infraspinatus, a critical external rotator of the humerus. Because the motor branches to the supraspinatus diverge proximal to the spinoglenoid notch, an entrapment strictly at the spinoglenoid level results in isolated infraspinatus weakness and atrophy, while preserving supraspinatus function—a crucial clinical distinction that guides both diagnosis and the specific site of surgical intervention.
Critical Neurovascular Relationships and Anatomic Variants
The arthroscopic approach to the suprascapular nerve is fraught with potential hazards due to the dense concentration of critical neurovascular structures in the immediate vicinity. The most prominent risk factor is the suprascapular artery. While typically coursing superior to the TSL, it can be remarkably tortuous and frequently sends small anastomotic branches inferiorly that intertwine with the ligament and the nerve. Meticulous hemostasis using precise radiofrequency ablation is essential during the exposure of the TSL to prevent bleeding that can obscure the surgical field and lead to iatrogenic nerve injury during blind dissection.
Another critical relationship is the proximity of the brachial plexus and the subclavian vessels, located just anterior and medial to the suprascapular notch. While these structures are generally safe if the surgeon strictly maintains instruments posterior to the coracoid base and directs dissection laterally and posteriorly, a loss of spatial orientation can lead to catastrophic anterior penetration. The surgeon must constantly reference the medial border of the coracoid base and the anterior margin of the supraspinatus muscle to ensure dissection remains within the safe anatomical boundaries of the suprascapular region.
Anatomic variants further complicate the surgical landscape. The suprascapular nerve may occasionally bifurcate proximal to the suprascapular notch, resulting in two distinct nerve bundles passing through the foramen. Alternatively, the nerve may pass superior to the TSL, running alongside the artery, or pass through an anomalous osseous foramen located entirely within the body of the scapula, inferior to the true notch. Surgeons must rely on careful tactile feedback with a probe and meticulous visual inspection to identify these variants before proceeding with any definitive ligamentous release. Failure to recognize a bifurcated nerve can result in an incomplete decompression or inadvertent transection of a motor branch.
Clinical Evaluation and Diagnostic Modalities
The Diagnostic Puzzle: History and Physical Examination
When a patient presents with suspected suprascapular nerve entrapment, the history is often complex, frequently mimicking rotator cuff pathology, cervical radiculopathy, or primary subacromial impingement. The classic presentation involves a deep, poorly localized, aching pain in the posterior or posterolateral aspect of the shoulder, often radiating down the arm or into the neck. Patients typically report an insidious onset of symptoms, exacerbated by overhead activities, cross-body adduction, or prolonged positioning of the arm in abduction and external rotation. A hallmark symptom is a profound sense of "heaviness" or fatigue in the shoulder, which is a direct manifestation of progressive motor unit loss.
The physical examination requires a meticulous and systematic approach to unmask the subtle signs of neuropathy. Meticulous inspection of the shoulder girdle, with the patient disrobed from the waist up, is paramount. The surgeon must look for asymmetric hollowing or atrophy in the supraspinatus and infraspinatus fossae, comparing the affected side to the contralateral shoulder. Atrophy is often the most reliable clinical indicator of chronic nerve compression. Isolated infraspinatus atrophy suggests a spinoglenoid notch lesion, whereas combined supraspinatus and infraspinatus atrophy points toward a more proximal lesion at the suprascapular notch.
Strength testing must be rigorous and specific to isolate the affected musculature. We test for isolated weakness of the supraspinatus using the "empty can" (Jobe's) test and the "full can" test. Infraspinatus weakness is assessed via resisted external rotation with the arm adducted to the side, and by evaluating for a positive external rotation lag sign. The Whipple test (resisted forward flexion with the arm internally rotated and abducted to 90 degrees) is also highly sensitive for eliciting supraspinatus weakness. Crucially, palpation of the rotator cuff insertions typically reveals no palpable defect, helping to distinguish entrapment neuropathy from a massive, primary rotator cuff tear.
Advanced Imaging: MRI and Ultrasound
Magnetic Resonance Imaging (MRI) is the imaging modality of choice for evaluating suspected suprascapular nerve entrapment. A high-resolution MRI, ideally utilizing a dedicated shoulder coil and including T1, T2 fat-suppressed, and proton density sequences, provides invaluable diagnostic information. The primary utility of MRI is twofold: identifying the specific etiology of the compression and assessing the status of the target musculature. MRI is exquisitely sensitive for detecting space-occupying lesions, particularly paralabral ganglion cysts at the spinoglenoid notch. It also allows for the detailed evaluation of the glenoid labrum to identify the underlying tear that frequently feeds the cyst.
Furthermore, MRI provides a quantitative assessment of muscle viability. In the setting of chronic denervation, MRI will reveal characteristic patterns of muscle edema (in the acute to subacute phase, visible on T2-weighted images) followed by progressive fatty infiltration and volume loss (visible on T1-weighted images) within the supraspinatus and infraspinatus bellies. The Goutallier classification system is frequently utilized to grade the severity of this fatty degeneration. Importantly, the MRI will typically demonstrate an intact rotator cuff tendon, confirming that the muscle atrophy is secondary to denervation rather than a primary tendinous avulsion.
In recent years, high-resolution diagnostic ultrasound has emerged as a valuable, dynamic adjunct to MRI. Ultrasound allows for the real-time, dynamic assessment of the suprascapular nerve as it traverses the notch. It can identify thickened transverse scapular ligaments, vascular anomalies, and ganglion cysts. Moreover, ultrasound facilitates precise, image-guided perineural injections of local anesthetic and corticosteroids. A positive diagnostic response to an ultrasound-guided suprascapular nerve block—characterized by immediate, albeit temporary, pain relief—strongly corroborates the diagnosis of entrapment and serves as a positive prognostic indicator for surgical decompression.
Electromyography and Nerve Conduction Studies
While clinical examination and advanced imaging provide critical clues, Electromyography (EMG) and Nerve Conduction Studies (NCS) remain the definitive "gold standard" for confirming the diagnosis of suprascapular neuropathy. These neurodiagnostic studies should ideally be performed by a neurologist or physiatrist with specific expertise in proximal entrapment lesions of the upper extremity. The timing of the study is crucial; EMG changes typically take 3 to 4 weeks to manifest following the onset of acute nerve injury or compression.
Nerve conduction studies evaluate the latency and amplitude of the electrical signal as it travels along the nerve. In suprascapular nerve entrapment, NCS will typically demonstrate prolonged distal motor latencies to the supraspinatus and/or infraspinatus muscles, indicating a focal demyelinating block at the site of compression. A reduction in the compound muscle action potential (CMAP) amplitude suggests axonal loss. The specific pattern of latency prolongation helps localize the lesion: prolonged latencies to both muscles suggest a suprascapular notch lesion, while isolated prolongation to the infraspinatus points to a spinoglenoid notch lesion.
Electromyography involves the insertion of a needle electrode directly into the muscle bellies to evaluate electrical activity at rest and during voluntary contraction. In the setting of active denervation, the EMG will reveal abnormal spontaneous activity, such as fibrillation potentials and positive sharp waves, indicating that the muscle fibers have lost their neural connection. During voluntary contraction, the study may show a reduced recruitment pattern and the presence of large, polyphasic motor unit action potentials (MUAPs), which represent the nerve's attempt at reinnervation via collateral sprouting. The combination of these EMG and NCS findings provides definitive proof of neuropathy and localizes the exact level of entrapment.
Exhaustive Indications and Contraindications
Formulating the Surgical Rationale
The decision to proceed with arthroscopic suprascapular nerve release is predicated on a careful synthesis of the patient's clinical presentation, imaging findings, and electrodiagnostic data. Surgical intervention is rarely the first line of treatment. A comprehensive trial of nonoperative management—including activity modification, targeted physical therapy focusing on periscapular stabilization and posture correction, NSAIDs, and potentially image-guided perineural injections—should be exhausted over a period of 3 to 6 months. However, when conservative measures fail to halt the progression of symptoms, or when objective evidence of severe denervation is present, surgical decompression is definitively indicated.
The primary indication for surgery is persistent, debilitating pain and progressive weakness that correlates with positive EMG/NCS findings confirming focal entrapment at either the suprascapular or spinoglenoid notch. Furthermore, the presence of a space-occupying lesion, such as a large symptomatic ganglion cyst identified on MRI, constitutes a strong indication for early surgical intervention, as these mechanical blocks rarely resolve spontaneously and will continue to exert deleterious pressure on the nerve. In such cases, the surgical plan must include both nerve decompression and the definitive management of the underlying labral pathology to prevent cyst recurrence.
Conversely, the surgeon must be acutely aware of the contraindications to surgical release. The most critical differential diagnosis to exclude is Parsonage-Turner Syndrome (idiopathic neuralgic amyotrophy). This viral or autoimmune condition presents with acute, excruciating shoulder pain followed by profound weakness and atrophy, often mimicking mechanical entrapment. However, Parsonage-Turner is a self-limiting inflammatory neuritis, not a mechanical compression, and surgical release provides no benefit and may exacerbate the condition. Similarly, cervical radiculopathy (particularly C5-C6 root compression) must be ruled out via cervical spine MRI and careful clinical examination, as the symptoms can closely mimic suprascapular neuropathy.
Table of Indications and Contraindications
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Indications | Refractory pain/weakness (>3-6 months) despite conservative care. | Indicates failure of non-operative modalities; risk of permanent axonal loss outweighs surgical risk. |
| Positive EMG/NCS confirming focal entrapment. | Objective evidence of demyelination/axonal loss localized to the suprascapular or spinoglenoid notch. | |
| MRI evidence of a space-occupying lesion (e.g., ganglion cyst) causing compression. | Mechanical compression requires physical removal/decompression; addresses the root cause directly. | |
| Progressive, isolated atrophy of supraspinatus/infraspinatus with intact rotator cuff. | Clinical hallmark of chronic denervation requiring immediate intervention to halt muscle degeneration. | |
| Relative Indications | Concomitant massive, retracted rotator cuff tear with severe traction neuropathy. | Releasing the nerve may improve mobilization of the cuff and enhance postoperative neural drive. |
| Absolute Contraindications | Parsonage-Turner Syndrome (Neuralgic Amyotrophy). | Inflammatory neuritis; mechanical decompression is ineffective and potentially harmful. |
| Cervical Radiculopathy (C5-C6) as the primary source of symptoms. | Symptoms originate proximally at the spinal root level; peripheral release will not resolve the pathology. | |
| Active local or systemic infection. | Standard surgical contraindication; risk of seeding the joint or deep fascial spaces. | |
| Advanced, irreversible fatty infiltration (Goutallier Grade 4) with no pain. | If pain is absent and muscle is entirely replaced by fat, motor recovery is impossible; surgery offers no functional benefit. |
Pre-Operative Planning, Templating, and Patient Positioning
Patient Counseling and Operative Room Setup
Meticulous preoperative planning is the cornerstone of a successful arthroscopic suprascapular nerve release. The process begins in the clinic with exhaustive patient counseling. The surgeon must set realistic expectations regarding the postoperative timeline. Patients must understand that while pain relief is often immediate following decompression, the recovery of muscle strength and the reversal of atrophy are protracted processes. Nerve regeneration occurs at a rate of approximately 1 millimeter per day; therefore, functional motor recovery may take 6 to 12 months, and in cases of chronic, severe atrophy, full strength may never be completely restored. Informed consent must explicitly detail the risks of neurovascular injury, incomplete release, and the potential need for open conversion.
The operative room setup requires specific attention to detail. General anesthesia is the standard of care, ensuring complete muscle relaxation and patient immobility during delicate perineural dissection. We routinely employ a preoperative regional interscalene nerve block, administered by the anesthesia team under ultrasound guidance. This provides excellent intraoperative and postoperative analgesia, significantly reducing the
Clinical & Radiographic Imaging Archive