Autologous Chondrocyte Implantation: A Masterclass in Cartilage Restoration

Key Takeaway
Welcome, fellows, to an in-depth operative masterclass on Autologous Chondrocyte Implantation (ACI). This two-stage procedure is a cornerstone for treating focal, full-thickness articular cartilage defects in young, active patients. We'll meticulously cover patient selection, detailed surgical anatomy, precise intraoperative technique, critical pearls, potential pitfalls, and comprehensive postoperative rehabilitation. Prepare to scrub in and master this advanced cartilage restoration technique.
Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues, fellows, and residents, to the operating theater and the academic lectern. Today, we delve into a profound and technically demanding biologic procedure: Autologous Chondrocyte Implantation (ACI). This intervention transcends the traditional boundaries of standard knee arthroscopy; it represents a sophisticated, cellular-level biological reconstruction. It is a testament to the orthopedic community's relentless pursuit of true hyaline cartilage restoration, moving away from the palliative, fibrocartilaginous outcomes of marrow stimulation techniques. Once relegated to the realm of experimental salvage procedures, ACI has robustly matured into an indispensable, highly successful surgical tool for a meticulously selected subset of patients. It is particularly targeted toward young, high-demand individuals suffering from debilitating, full-thickness focal chondral lesions that threaten their functional longevity.
The epidemiological burden of articular cartilage defects is staggering, with retrospective analyses of routine knee arthroscopies revealing that up to sixty percent of patients exhibit some degree of chondral damage. While many of these lesions are asymptomatic incidentalomas, a significant percentage progress to symptomatic, full-thickness defects that profoundly impair joint kinematics and patient quality of life. The natural history of an untreated, full-thickness articular cartilage defect is notoriously unforgiving. Due to the avascular, aneural, and alymphatic nature of hyaline cartilage, intrinsic healing is virtually nonexistent. The resulting altered joint contact pressures inevitably lead to progressive rim overload, shear stress on adjacent healthy cartilage, and the accelerated onset of early unicompartmental osteoarthritis.
The fundamental essence and biological triumph of ACI lie in its meticulously orchestrated two-stage approach. The primary stage involves the arthroscopic harvest of healthy, autologous chondrocytes from minor load-bearing regions of the knee, followed by their ex vivo enzymatic digestion and extensive laboratory expansion. The secondary stage, performed weeks later, necessitates an arthrotomy to implant these millions of expanded cells into a radically debrided and prepared chondral defect. The ultimate surgical objective is to reconstitute the articular surface with a durable, functional "hyaline-like" repair tissue. Extensive long-term clinical data and histological biopsies have definitively proven that ACI-generated tissue offers vastly superior compressive stiffness, shear resistance, and documented clinical survivorship when directly compared to the transient fibrocartilage produced by microfracture.
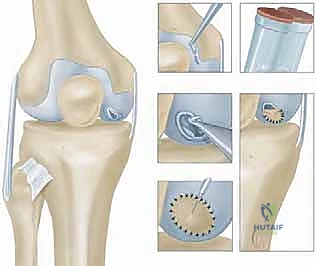
Detailed Surgical Anatomy and Biomechanics
Before a scalpel is ever passed, the operating surgeon must possess an absolute, granular understanding of the target tissue. Articular cartilage is not merely an inert shock absorber; it is a dynamic, highly specialized, and metabolically active connective tissue covering the epiphyseal ends of diarthrodial joints. Its primary biomechanical mandates are twofold: to act as a resilient, viscoelastic load-bearing structure that distributes monumental compressive forces across the subchondral bone, and to provide a remarkably low-friction tribological surface, boasting a coefficient of friction significantly lower than ice sliding on ice.
Microscopic Architecture and Zonal Organization
Histologically, normal articular cartilage is a masterpiece of structural engineering, comprising four distinct, highly organized zones. Each zone is characterized by unique chondrocyte morphology, specific collagen fibril orientation, and varying proteoglycan concentrations, all tailored to meet specific biomechanical demands. The Superficial (Tangential) Zone is the thinnest outermost layer, containing flattened chondrocytes and densely packed Type II collagen fibers oriented strictly parallel to the articular surface. This precise parallel orientation is critically responsible for resisting the immense shear forces generated during joint articulation and acts as a semi-permeable barrier regulating the influx and efflux of large macromolecules.

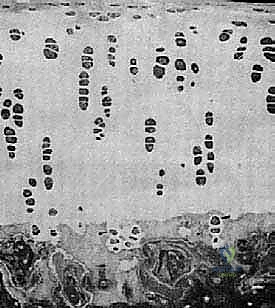
Deep to the superficial layer lies the Middle (Transitional) Zone, which constitutes a larger volume of the total cartilage thickness. Here, the chondrocytes adopt a more spherical morphology, and the collagen fibers transition into an oblique, less organized, decussating arrangement. This zone serves as the primary shock absorber, transitioning shear forces into compressive forces. Beneath this is the Deep (Radial) Zone, characterized by chondrocytes arranged in vertical columns and thick collagen fibers oriented strictly perpendicular to the articular surface. This vertical orientation provides the maximal resistance to compressive loads and securely anchors the uncalcified cartilage to the underlying structures.


The deepest layer is the Calcified Zone, which serves as the critical transitional interface securing the articular cartilage to the rigid subchondral bone. It is separated from the deep radial zone by a highly distinct basophilic line known as the "tidemark." The tidemark is a vital anatomical and biomechanical boundary; it represents the absolute limit of nutritional diffusion from the synovial fluid and plays a crucial role in tethering the collagen fibrils of the deep zone. In surgical cartilage restoration, violating the subchondral bone plate and disrupting the calcified zone inadvertently invites marrow elements and bleeding, which universally results in inferior fibrocartilaginous repair rather than the desired hyaline regeneration.
Chondrocyte Biology and Extracellular Matrix
The chondrocyte is the solitary cellular architect of articular cartilage, constituting a mere two to five percent of the tissue's total wet weight. Despite their sparse population, these highly specialized cells are entirely responsible for the synthesis, maintenance, and internal turnover of the complex extracellular matrix (ECM). Because cartilage is devoid of a vascular supply, chondrocytes exist in a profoundly hypoxic environment, relying entirely on anaerobic glycolysis for energy and on cyclic mechanical loading to facilitate the diffusion of nutrients and oxygen from the synovial fluid.
The ECM synthesized by these cells is a biphasic material composed primarily of tissue fluid and a solid structural macromolecular framework. Water constitutes an astonishing sixty-five to eighty percent of the total wet weight of cartilage. The interaction of this fluid with the solid matrix is the foundation of cartilage biomechanics. The solid framework is dominated by Type II collagen (providing tensile strength and structural scaffolding) and large aggregating proteoglycans, primarily aggrecan.
Aggrecan molecules are densely populated with negatively charged glycosaminoglycan (GAG) side chains, such as chondroitin sulfate and keratan sulfate. These negative charges repel each other while simultaneously attracting massive amounts of water through the Donnan osmotic effect. This creates an immense internal swelling pressure that is counteracted and contained by the tensile strength of the Type II collagen network. When an external compressive load is applied to the joint, water is forced out of the ECM; when the load is removed, the hydrophilic proteoglycans aggressively draw the water back in. This dynamic fluid exudation and imbibition is the cornerstone of cartilage's viscoelastic properties and is precisely the micro-architecture we aim to replicate with ACI.
Exhaustive Indications and Contraindications
The clinical success of Autologous Chondrocyte Implantation is entirely predicated upon meticulous, uncompromising patient selection. The literature is replete with evidence demonstrating that ACI yields spectacular results in the correctly chosen patient, but fails predictably when applied outside of strict parameters. The orthopedic surgeon must evaluate not only the isolated focal lesion but the entire joint as an integrated biomechanical organ. We are seeking the optimal physiological candidate with a discrete structural defect within an otherwise pristine intra-articular environment.
Patient Selection Criteria
The ideal candidate for ACI is a physiologically young, highly active patient presenting with a symptomatic, full-thickness, unipolar focal chondral or osteochondral defect of the knee. While chronological age is a consideration—typically limited to patients between the ages of 15 and 55—biological age, tissue quality, and patient compliance are far more critical determinants. The patient must possess the cognitive and psychological fortitude to endure a rigorous, multi-phase rehabilitation protocol that spans up to twelve or eighteen months.
The specific lesion must meet stringent arthroscopic criteria, ideally classified as an Outerbridge Grade III or IV defect. Outerbridge Grade III indicates deep fissuring extending down to, but not through, the subchondral bone, often accompanied by loose cartilaginous flaps. Grade IV indicates complete full-thickness loss with exposed subchondral bone. ACI is particularly advantageous, and often the primary treatment of choice, for large lesions exceeding 2.0 square centimeters. For massive defects (e.g., >4.0 cm²), ACI and its modern iterations (such as Matrix-Induced ACI or MACI) are the only biologically viable restorative options, as microfracture in these dimensions universally fails. Furthermore, ACI is an excellent salvage strategy for failed prior marrow stimulation techniques or as a definitive resurfacing procedure following the bone grafting of large, unstable osteochondritis dissecans (OCD) lesions.
Absolute and Relative Contraindications
It is equally, if not more, important to recognize the absolute contraindications to ACI. ACI is fundamentally a surface restoration technique for focal defects; it is categorically not a treatment for degenerative joint disease or diffuse osteoarthritis. The presence of bipolar "kissing" lesions (e.g., full-thickness loss on both the femoral condyle and the opposing tibial plateau) or radiographic evidence of Fairbanks changes (joint space narrowing, osteophytosis, subchondral sclerosis) absolutely precludes the use of ACI. Implantation into a globally degenerative joint will result in rapid mechanical destruction of the delicate cellular graft.
Furthermore, any uncorrected biomechanical abnormalities represent absolute contraindications until they are surgically addressed. The knee joint must be stable, aligned, and possess intact meniscal load-sharing capabilities. Co-morbidities such as ligamentous laxity (e.g., ACL deficiency), significant meniscal loss (which exponentially increases focal contact pressures), or coronal plane malalignment (varus or valgus deformity overloading the affected compartment) will subject the maturing ACI graft to catastrophic shear and compressive forces. These issues must be corrected either in a staged fashion prior to ACI or as a concomitant procedure during the implantation stage.
| Category | Specific Contraindications for ACI | Management Strategy |
|---|---|---|
| Absolute | Diffuse Osteoarthritis (Kellgren-Lawrence Grade III/IV) | Arthroplasty or conservative management. ACI strictly contraindicated. |
| Absolute | Bipolar "Kissing" Lesions | Alternative resurfacing or arthroplasty. |
| Absolute | Active joint infection or inflammatory arthropathy (e.g., Rheumatoid) | Medical management; biologic restoration contraindicated. |
| Relative/Correctable | Coronal Malalignment (Varus/Valgus overloading the defect) | Concomitant or staged corrective osteotomy (HTO or DFO). |
| Relative/Correctable | Ligamentous Instability (ACL/PCL/MCL/LCL deficiency) | Concomitant or staged ligamentous reconstruction. |
| Relative/Correctable | Significant Meniscal Deficiency in the affected compartment | Concomitant or staged Meniscal Allograft Transplantation (MAT). |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock of a successful ACI procedure. The assessment must be exhaustive, evaluating the precise dimensions of the cartilage defect, the integrity of the subchondral bone, the status of the menisci and ligaments, and the overall mechanical axis of the lower extremity. A failure to identify and plan for concomitant pathologies will inevitably compromise the survival of the implanted chondrocytes.
Advanced Imaging Modalities
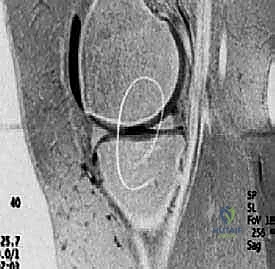
The diagnostic workup begins with a comprehensive suite of plain radiographs. We require weight-bearing anteroposterior (AP), 45-degree flexion posteroanterior (Rosenberg or notch), Merchant or sunrise patellofemoral views, and a true lateral view. While articular cartilage is radiolucent, these films are critical for identifying joint space narrowing, subchondral sclerosis, osteophyte formation, and the bony architecture of osteochondritis dissecans (OCD) lesions. Crucially, full-length, weight-bearing lower extremity alignment films (hip-to-ankle) are mandatory to assess the mechanical axis. Any deviation that places the weight-bearing line through the compartment containing the defect necessitates a corrective osteotomy.


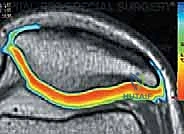
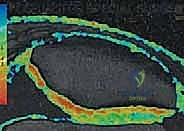
Magnetic Resonance Imaging (MRI) is the undisputed gold standard for non-invasive cartilage evaluation. Standard fast spin-echo proton density and T2-weighted sequences provide excellent visualization of lesion size, depth, and the presence of subchondral edema or cystic changes. However, modern cartilage restoration requires advanced MRI techniques. T2 mapping allows for the biochemical assessment of the collagen network's integrity and hydration status, providing a color-coded topographical map of cartilage quality. Delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) can quantify the glycosaminoglycan (GAG) concentration, offering a microscopic look at the surrounding "healthy" cartilage to ensure it provides a stable rim for the future ACI graft.



Patient Positioning and Operating Room Setup
For both Stage 1 (Harvest) and Stage 2 (Implantation), meticulous patient positioning is paramount. The patient is placed supine on a standard radiolucent operating table. A high thigh tourniquet is applied to the operative leg, though it is typically only inflated during the open implantation phase to ensure a bloodless field, which is critical for fibrin glue adhesion. The entire lower extremity is meticulously prepped and draped free from the iliac crest down to the toes.
The ability to manipulate the knee through a full, unrestricted range of motion is absolutely essential. During the arthrotomy in Stage 2, extreme hyperflexion is often required to access posterior condylar lesions, while full extension is needed for patellofemoral defects. Therefore, rigid leg holders that restrict flexion past 90 degrees should be avoided. Instead, a simple lateral post or a sandbag taped to the table allows for dynamic positioning. Fluoroscopy must be readily available in the room, particularly if concomitant procedures like an osteotomy or hardware removal are planned.
Step-by-Step Surgical Approach and Fixation Technique
The execution of Autologous Chondrocyte Implantation is a true test of surgical precision and respect for tissue biology. The procedure is inherently divided into two distinct operative events, separated by a period of three to six weeks to allow for the in vitro expansion of the harvested chondrocytes.
Stage One: Diagnostic Arthroscopy and Chondrocyte Harvest
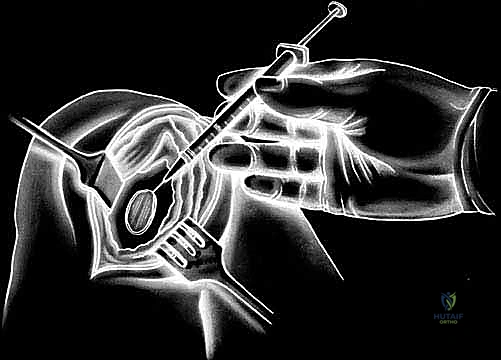
The first stage is an outpatient arthroscopic procedure. The primary goals are to definitively confirm the preoperative diagnosis, meticulously map the size and location of the chondral defect, assess the status of the opposing articular surfaces and menisci, and harvest the chondrocytes. Using standard anterolateral and anteromedial portals, a systematic diagnostic sweep of the joint is performed. The defect is probed to determine the stability of the surrounding cartilage rim. If the lesion is deemed appropriate for ACI, we proceed to the harvest.

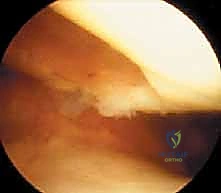
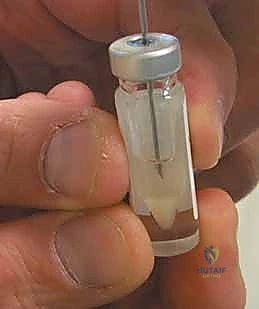
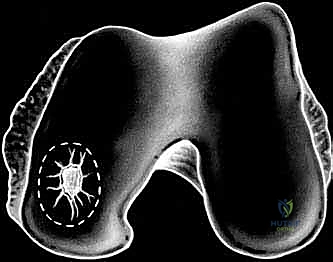
The harvest site must be chosen from a strictly non-weight-bearing region of the articular surface to minimize donor site morbidity. The most common and reliable sites are the superomedial edge of the femoral trochlea or the lateral aspect of the intercondylar notch. Using a sharp ring curette or a specialized cartilage gouge, thin shavings of full-thickness articular cartilage are meticulously harvested. It is imperative to avoid penetrating the subchondral bone, as bleeding will contaminate the sample with marrow elements, macrophages, and fibroblasts, which can overtake the chondrocyte culture in the lab. Approximately 200 to 300 milligrams of cartilage (roughly the size of two Tic-Tac candies) is required. This pristine tissue is immediately placed into a sterile, specialized transport medium and couriered to the cell-processing laboratory under strict temperature control.
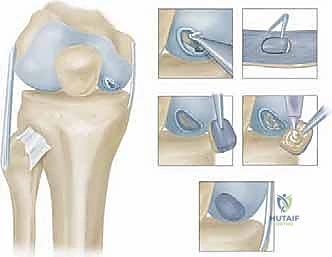
Stage Two: Defect Preparation and Implantation
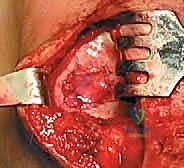
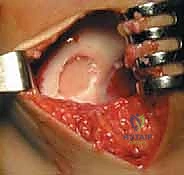
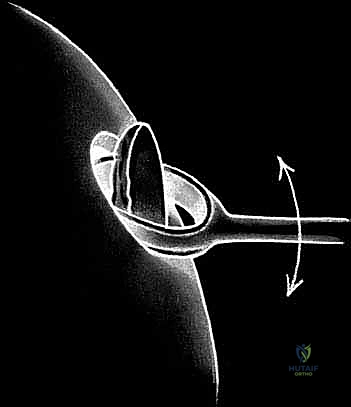
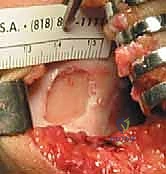
Stage two is the definitive open implantation. Depending on the lesion's location, a mini-medial or lateral parapatellar arthrotomy is utilized. The exposure must be adequate to visualize the entire defect without excessive retraction that could damage the surrounding healthy cartilage. Once exposed, the critical step of defect preparation begins. Using sharp curettes and a scalpel, the damaged cartilage is radically debrided back to entirely healthy, stable, vertically oriented margins. This creates the "ring of fire," a stable perimeter essential for containing the cellular implant.



The base of the defect is then meticulously debrided of all calcified cartilage down to the subchondral bone plate. However, the surgeon must exercise extreme caution not to breach the subchondral bone. Any pinpoint bleeding from the bone must be meticulously controlled with topical epinephrine or precise application of fibrin thrombin, as blood entering the defect will introduce fibroblasts and drive the repair toward inferior fibrocartilage. Once the defect is perfectly prepared, a sterile aluminum foil template is pressed into the defect to capture its exact dimensions.



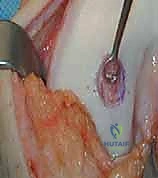
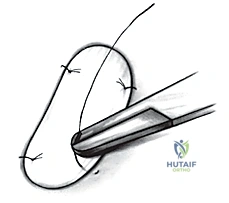
Historically, a periosteal patch was harvested from the proximal tibia to cover the defect. However, due to high rates of graft hypertrophy, modern techniques utilize a synthetic Type I/III collagen membrane (ACI-C) or a matrix seeded with the cells directly (MACI). The template is used to cut the collagen membrane to the exact size of the defect. If using the traditional liquid suspension ACI, the membrane is sutured over the defect using closely spaced 6-0 absorbable sutures (e.g., Vicryl), leaving a small opening at the superior pole. The suture line is then meticulously sealed with fibrin glue to create a watertight compartment.


Clinical & Radiographic Imaging Archive