Anterior Drainage of Pediatric Septic Hip: A Masterclass in the Modified Smith-Peterson Approach
Key Takeaway
Join us in the OR for an immersive masterclass on anterior drainage of the pediatric septic hip. We'll meticulously cover comprehensive anatomy, preoperative planning, and granular, real-time surgical execution using the modified Smith-Peterson approach. Learn critical pearls, avoid pitfalls, and ensure optimal outcomes for these vulnerable patients. This guide provides an exhaustive breakdown from incision to closure and beyond.
Comprehensive Introduction and Patho-Epidemiology
Septic arthritis of the pediatric hip represents one of the most critical and time-sensitive surgical emergencies encountered in orthopedic practice. Unlike many conditions in pediatric orthopedics where a "watch and wait" approach may be permissible, a purulent infection within the confines of the hip joint demands immediate, decisive, and meticulous surgical intervention. The overarching principles governing the management of this condition are unequivocal: rapid and accurate diagnostic confirmation, emergent surgical decompression and drainage, targeted antimicrobial therapy, and vigilant, long-term surveillance for potential sequelae. As orthopedic surgeons, we must recognize that the most catastrophic outcomes—ranging from complete destruction of the articular cartilage to devastating avascular necrosis (AVN) of the femoral head—are almost invariably the direct consequence of delayed diagnosis or inadequate definitive treatment. Our primary objective is not merely to eradicate the infection, but to meticulously preserve the biomechanical integrity and long-term function of the hip joint.
The pathogenesis of pediatric septic hip arthritis is multifaceted, affecting patients across the entire developmental spectrum, from the immunologically fragile neonate to the robust, active adolescent. The sterile joint cavity is typically inoculated via one of three primary mechanisms. The most common route is hematogenous dissemination, where transient bacteremia leads to the direct seeding of the highly vascularized subsynovial layer of the joint capsule. Alternatively, the joint may be inoculated indirectly via the contiguous spread from a primary focus of osteomyelitis in the proximal femoral metaphysis. Less frequently, an adjacent acetabular osteomyelitis or a severe pelvic infection can decompress directly into the intra-articular space. Regardless of the route of entry, the subsequent inflammatory cascade is both rapid and highly destructive.
Once pathogenic bacteria establish a foothold within the synovial fluid, a massive influx of polymorphonuclear leukocytes (PMNs), plasma proteins, and inflammatory cytokines ensues. This rapid accumulation of purulent exudate leads to a tense, high-pressure joint effusion. In the non-expansile compartment of the hip capsule, this elevated intracapsular pressure is our primary adversary. It acts as a mechanical tamponade, severely compromising the delicate, low-pressure venous and arterial blood supply to the femoral head, specifically the retinacular vessels. Concurrently, the biological milieu becomes profoundly toxic. Proteolytic enzymes released from hyperplastic synovial membrane cells, coupled with interleukin-1 (IL-1) and tumor necrosis factor-alpha (TNF-α) secreted by activated monocytes, trigger the release of potent matrix metalloproteinases (MMPs) from both chondrocytes and synoviocytes. Animal models have chillingly demonstrated the sheer velocity of this destruction: critical proteoglycan matrix can be irreversibly lost within 5 days, and the structural collagen framework within 9 days of infection onset. It is imperative to understand that even the most aggressive, broad-spectrum intravenous antibiotic therapy cannot completely halt this enzymatic degradation if the mechanical pressure and purulent burden are not surgically relieved.
The pediatric proximal femur possesses a unique anatomical vulnerability that fundamentally alters the pathophysiology of local infections. In infants and young children, the metaphysis of the proximal femur is entirely intra-articular, enveloped by the capsular attachments that extend down to the intertrochanteric line. This critical anatomical reality dictates that a primary hematogenous osteomyelitis originating in the metaphyseal bone—a common site due to sluggish blood flow in metaphyseal venous sinusoids—can easily and rapidly rupture through the thin metaphyseal cortex directly into the hip joint. Consequently, what begins as an isolated bone infection rapidly escalates into a catastrophic joint emergency. This intra-articular metaphyseal anatomy necessitates a high index of suspicion; whenever a child presents with proximal femoral osteomyelitis, concomitant septic arthritis must be assumed until definitively proven otherwise.
Detailed Surgical Anatomy and Biomechanics
Before initiating any surgical incision, a profound and exhaustive understanding of the regional anatomy is absolutely paramount. The anterior approach to the hip, specifically the modified Smith-Peterson approach, navigates through a complex, three-dimensional landscape of critical neurovascular structures and dynamic muscular intervals. Mastery of these anatomical relationships is the cornerstone of a successful surgical outcome, allowing for rapid exposure while meticulously avoiding iatrogenic complications.
Vascularity of the Proximal Femur
The vascular supply to the developing femoral head is a highly complex, dynamic, and fragile system. It is predominantly derived from the medial femoral circumflex artery (MFCA) and, to a significantly lesser extent, the lateral femoral circumflex artery (LFCA). These vessels, which are primary branches of the deep femoral artery (profunda femoris), anastomose to form a robust extracapsular arterial ring situated at the base of the femoral neck. From this extracapsular ring, multiple subsynovial retinacular vessels branch off, penetrate the dense hip joint capsule, and ascend along the surface of the femoral neck. These retinacular vessels are the terminal lifeline to the femoral head epiphysis.
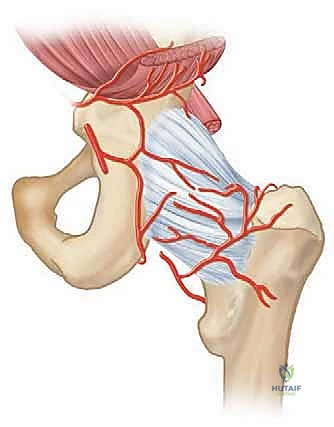
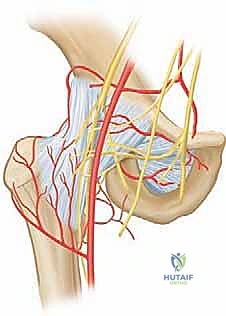
Crucially, in the pediatric patient, these ascending retinacular vessels do not cross the cartilaginous proximal femoral physis. The physis acts as an impenetrable barrier to vascular anastomosis. Therefore, the epiphysis relies entirely on these lateral epiphyseal vessels. When a tense, purulent effusion develops within the rigid hip capsule, the intracapsular pressure rapidly exceeds the perfusion pressure of these delicate retinacular vessels. This mechanical tamponade leads to acute ischemia of the femoral head. If surgical decompression is delayed, this ischemia progresses to irreversible avascular necrosis (AVN)—a devastating complication that can lead to complete collapse of the femoral head, severe limb length discrepancy, and early-onset osteoarthritis.
Muscular Intervals and Surgical Corridors
The modified Smith-Peterson approach is universally favored for pediatric hip drainage because it elegantly exploits true internervous and intermuscular planes, minimizing denervation and muscle morbidity. The approach is conceptually divided into superficial and deep dissections.
The superficial layer relies on the interval between the sartorius muscle and the tensor fasciae latae (TFL). The sartorius, a long, strap-like muscle originating from the anterior superior iliac spine (ASIS), runs inferomedially and is innervated by the femoral nerve. Conversely, the TFL originates from the outer lip of the iliac crest and the ASIS, running distally to merge with the iliotibial band; it is innervated by the superior gluteal nerve. The fascial plane between these two muscles is a true internervous corridor, relatively avascular, and provides a safe, direct trajectory toward the deeper structures of the anterior hip.
The deeper dissection layer navigates the interval between the rectus femoris and the iliopsoas complex. The rectus femoris is a powerful bi-articular muscle featuring two distinct origins: a "straight" head originating from the anterior inferior iliac spine (AIIS) and a "reflected" (indirect) head originating from the ilium just superior to the acetabular rim, intimately blending with the anterior hip capsule. It is innervated by the femoral nerve. Medial to our working zone lies the iliopsoas muscle, formed by the convergence of the psoas major and iliacus muscles. The iliopsoas tendon crosses directly anterior to the hip joint capsule, acting as a dynamic anterior stabilizer. The surgeon must work meticulously along the lateral border of the rectus femoris, retracting it medially or laterally depending on the specific variation of the approach, to expose the underlying joint capsule.
Neurovascular Risks in the Anterior Approach
While the modified Smith-Peterson approach is anatomically sound, it is fraught with specific neurovascular hazards that demand constant vigilance. The most perilous structure encountered during the superficial dissection is the Lateral Femoral Cutaneous Nerve (LFCN). This purely sensory nerve exhibits high anatomical variability; it typically exits the pelvis by passing under, through, or over the inguinal ligament, immediately medial to the ASIS. However, aberrant branches frequently cross directly over the ASIS or traverse the proximal sartorius-TFL interval. The LFCN is highly susceptible to traction injury from overzealous retractor placement or direct transection during the superficial fascial incision. Injury results in meralgia paresthetica, characterized by chronic, distressing dysesthesia and numbness over the anterolateral thigh.
Deeper in the dissection, the femoral nerve represents a critical risk. Located deep and medial to our primary dissection plane, it resides in the protective groove between the psoas and iliacus muscles. While generally shielded by the bulk of the iliopsoas during a standard anterior approach, aggressive medial retraction—particularly with sharp-tipped Hohmann retractors—can compress or injure the nerve, leading to devastating quadriceps weakness. Furthermore, the primary femoral neurovascular bundle (Femoral Artery, Vein, and Nerve - NAVEL) lies medial to the iliopsoas. Provided the surgeon maintains a strict lateral trajectory and respects the medial boundary of the iliopsoas, these major vessels remain safely out of the operative field. However, the ascending branches of the lateral femoral circumflex artery (LFCA) consistently cross the operative field in the deep interval between the rectus femoris and iliopsoas; these must be meticulously identified, isolated, and ligated to prevent obscuring hemorrhage.
Exhaustive Indications and Contraindications
The decision to proceed with emergent surgical drainage of a pediatric hip is predicated on a synthesis of clinical presentation, laboratory markers, and imaging findings. The classic Kocher criteria remain a foundational tool in differentiating true septic arthritis from transient synovitis. The presence of a fever > 38.5°C, inability to bear weight, an Erythrocyte Sedimentation Rate (ESR) > 40 mm/hr, and a serum White Blood Cell (WBC) count > 12,000 cells/mm³ strongly predict septic arthritis. When all four criteria are met, the probability of septic arthritis approaches 99%. Subsequent studies by Caird et al. added a C-Reactive Protein (CRP) > 20 mg/L as an independent, highly sensitive predictor.
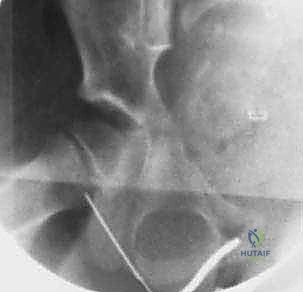
Surgical intervention is definitively indicated when clinical suspicion is high, supported by inflammatory markers, and confirmed by either a purulent joint aspirate or unequivocal advanced imaging (MRI) demonstrating intra-articular pus or severe capsular distension with concomitant marrow edema. It is critical to emphasize that if a joint aspirate yields frank pus, one must not delay surgery while awaiting formal Gram stain or culture results; the mechanical decompression must occur immediately.
| Category | Specific Parameters / Conditions | Clinical Rationale / Action |
|---|---|---|
| Absolute Indications | Purulent fluid on joint aspiration (>50,000 WBC/mm³, >90% PMNs). | Confirms gross infection; requires immediate mechanical decompression to prevent cartilage necrosis. |
| Absolute Indications | Positive Gram stain from synovial fluid aspirate. | Definitive evidence of bacterial presence; emergent arthrotomy required. |
| Strong Relative Indications | 3 or 4 Kocher Criteria met + elevated CRP (>20 mg/L) + US showing effusion. | High probability of septic hip. If aspiration is dry or equivocal, open exploration is generally favored over observation. |
| Strong Relative Indications | MRI evidence of concomitant proximal femoral osteomyelitis with joint effusion. | High risk of intra-articular decompression of the bone infection; requires simultaneous bone drilling and joint drainage. |
| Relative Contraindications | Overlying severe soft tissue infection/cellulitis at the planned anterior incision site. | Risk of seeding a sterile joint (if misdiagnosed) or complicating wound closure. Consider alternative approaches (e.g., medial/Ludloff) or ultrasound-guided aspiration first. |
| Absolute Contraindications | Severe hemodynamic instability or uncorrected coagulopathy. | Patient cannot tolerate general anesthesia. Must stabilize medically; consider bedside needle aspiration as a temporizing measure. |
| Alternative Diagnoses | Confirmed Transient Synovitis, Lyme Arthritis, or Juvenile Idiopathic Arthritis (JIA). | Requires medical management (NSAIDs, antibiotics for Lyme, rheumatologic workup). Surgery is contraindicated and adds unnecessary morbidity. |
Contraindications to the anterior surgical approach are exceedingly rare, given the life- and limb-threatening nature of the condition. However, profound hemodynamic instability or severe, uncorrected coagulopathy may necessitate a delay in open surgery, prompting a temporizing bedside ultrasound-guided needle aspiration under local anesthesia to relieve pressure while the patient is medically optimized. Additionally, if severe cellulitis or a cutaneous abscess overlies the anterior hip, an alternative surgical corridor, such as a medial (Ludloff) or anterolateral approach, should be strongly considered to avoid dragging superficial pathogens into the deep articular space.
Pre-Operative Planning, Diagnostic Imaging, and Patient Positioning
The preoperative phase is a high-stakes period requiring rapid, coordinated action between orthopedic surgery, pediatrics, infectious disease, and radiology. The patient, typically a febrile, irritable toddler refusing to bear weight and holding the affected hip in a position of flexion, abduction, and external rotation (the position of maximum capsular volume), requires immediate evaluation.
Initial laboratory studies must include a complete blood count (CBC) with differential, ESR, CRP, and aerobic/anaerobic blood cultures. Blood cultures are paramount, as they yield the causative organism in 40-50% of cases, guiding subsequent antimicrobial therapy even if joint fluid cultures remain sterile. The CRP is particularly vital; it rises rapidly within 6-8 hours of infection onset and falls quickly with successful treatment, making it an excellent barometer for monitoring therapeutic response, in contrast to the ESR, which may remain elevated for weeks.
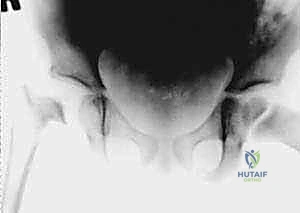
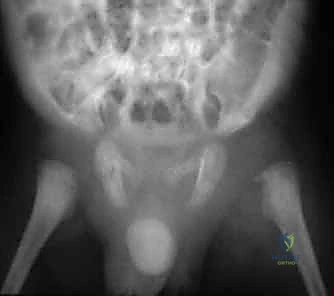
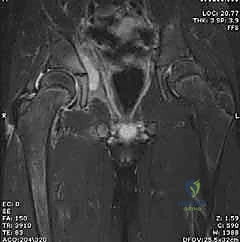
Diagnostic imaging begins with plain anteroposterior (AP) and frog-leg lateral radiographs of the pelvis. While early radiographs are often completely normal regarding osseous structures, they are crucial for ruling out trauma, tumors, or Legg-Calvé-Perthes disease. Subtle radiographic signs of a septic hip include widening of the medial joint space (teardrop distance), blurring of the pericapsular fat planes, or frank lateral subluxation of the femoral head due to severe effusion. In neonates, a painful, dislocated hip on plain film is a highly worrisome sign of advanced sepsis.
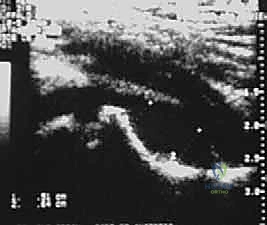
Ultrasound is the workhorse imaging modality for the pediatric irritable hip. It is highly sensitive, rapid, non-invasive, and does not require sedation. An ultrasound can definitively confirm the presence of an intra-articular effusion, measure the degree of capsular distension (typically comparing the anterior capsule-to-bone distance with the contralateral asymptomatic hip), and guide a diagnostic needle aspiration. However, ultrasound cannot reliably differentiate between sterile reactive fluid, purulent exudate, or hemarthrosis based solely on echogenicity.
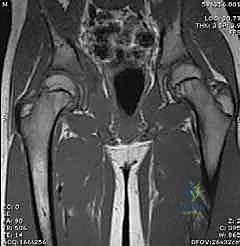
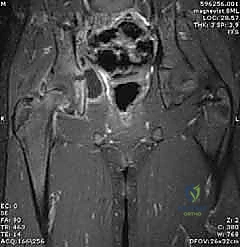
Magnetic Resonance Imaging (MRI) is the definitive, gold-standard imaging modality, utilized when the diagnosis remains equivocal after initial workup, or when concomitant extra-articular pathology is suspected. MRI provides exquisite soft-tissue contrast, capable of identifying early marrow edema indicative of proximal femoral or acetabular osteomyelitis, adjacent pyomyositis (e.g., psoas abscess), and the extent of capsular hypertrophy. T1-weighted, T2-weighted with fat suppression (STIR), and gadolinium-enhanced sequences are routinely employed. However, the requirement for general anesthesia in young children and logistical delays in obtaining an emergent MRI mean it should not postpone definitive surgical drainage if the clinical picture and ultrasound/aspiration findings already mandate intervention.
Patient positioning in the operating theater must facilitate both adequate surgical exposure and the potential need for intraoperative fluoroscopy. The patient is placed supine on a radiolucent operating table. A small, radiolucent bump (such as a folded towel) is placed under the ipsilateral hemipelvis to slightly elevate the affected hip, bringing the anterior structures into better relief. The entire hemipelvis, lower extremity, and ipsilateral abdomen are prepped and draped free, allowing for unrestricted manipulation of the hip joint during the procedure. This mobility is essential for dynamically altering tissue tension to aid deep dissection and for thoroughly irrigating the joint cavity.
Step-by-Step Surgical Approach and Drainage Technique
The surgical execution of the modified Smith-Peterson approach demands precision, anatomical respect, and thoroughness. Under general anesthesia, prior to the skin incision, a final diagnostic aspiration can be performed if not previously done, sending fluid immediately for cell count, Gram stain, and aerobic/anaerobic cultures.
The skin incision can be tailored to the patient's anatomy and the surgeon's preference. A longitudinal incision starting just distal to the ASIS and extending distally along the line of the sartorius provides excellent, extensible exposure. Alternatively, a transverse "bikini line" incision, centered over the lower abdomen/groin crease just distal to the ASIS, offers a superior cosmetic result while still allowing adequate mobilization of the superficial fascial windows.
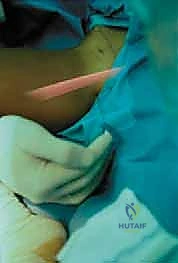
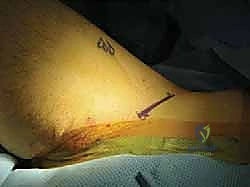
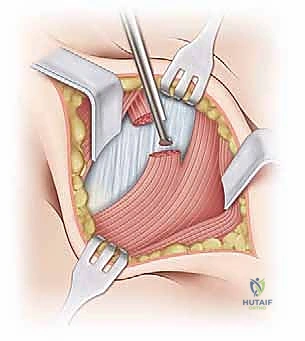
Following skin incision and meticulous hemostasis of the subcutaneous tissues, the deep investing fascia of the thigh (fascia lata) is identified. The interval between the sartorius (medial) and the TFL (lateral) is bluntly developed. The surgeon must remain acutely aware of the Lateral Femoral Cutaneous Nerve (LFCN) during this stage. The nerve is typically found in the medial aspect of the TFL fascia or crossing the interval; it must be carefully mobilized and gently retracted, usually medially with the sartorius, to prevent neurapraxia.
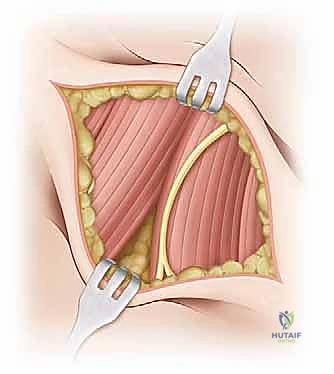
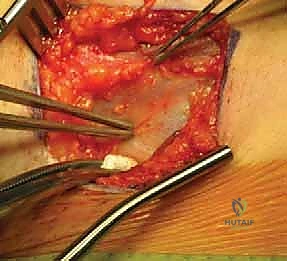
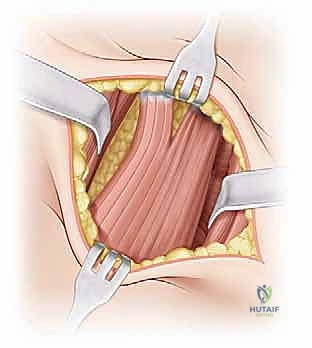
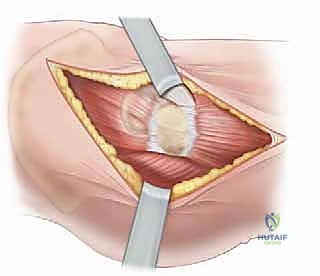
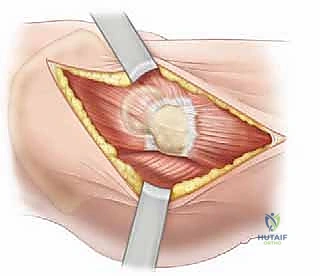
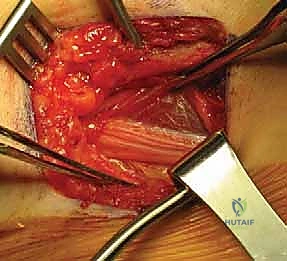
Deepening the dissection, the rectus femoris is identified. The plane is developed along the lateral border of the rectus femoris. The straight head of the rectus is traced proximally to its origin on the AIIS. At this juncture, the ascending branches of the lateral femoral circumflex artery and vein will be encountered crossing the operative field transversely. These vessels must be definitively isolated, clamped, and ligated or electrocoagulated; failure to do so will result in brisk bleeding that obscures the deep fascial planes. The rectus femoris is then retracted medially (or laterally, depending on specific exposure needs), revealing the underlying iliopsoas muscle and the anterior capsule of the hip joint. The iliopsoas is gently elevated and retracted medially, taking immense care not to place excessive traction on the femoral nerve lying within its substance.