Anterior Cervical Corpectomy and Fusion: A Masterclass in Decompression and Stabilization
Key Takeaway
This masterclass guides you through an anterior cervical corpectomy and fusion, a critical procedure for spinal cord compression. We'll meticulously cover patient selection, comprehensive preoperative planning, intricate surgical anatomy, and detailed intraoperative execution from incision to instrumentation. Learn to manage neurovascular risks, optimize graft placement, and prevent complications, ensuring thorough decompression and stable fusion for myelopathic patients.
Comprehensive Introduction and Patho-Epidemiology
The anterior cervical corpectomy and fusion (ACCF) represents a cornerstone in the armamentarium of the complex spine surgeon. This procedure is not merely an exercise in bone resection; it is an exacting biomechanical and neurological reconstruction. When a patient presents with significant cervical myelopathy secondary to retrovertebral compression, achieving meticulous decompression, restoring physiological sagittal balance, and securing a robust arthrodesis are paramount to arresting neurological decline. The cervical spine, with its dense concentration of critical neurovascular structures and demanding dynamic loads, requires absolute precision. This chapter will serve as an exhaustive masterclass on the nuances of ACCF, providing the academic orthopedic surgeon and neurosurgeon with a definitive framework for executing this complex procedure safely and effectively.
The Pathogenesis of Cervical Spondylotic Myelopathy
Before making an incision, the surgeon must possess a profound understanding of the underlying pathology. Cervical spondylotic myelopathy (CSM) is a progressive, insidious constellation of signs and symptoms arising from the mechanical compression and subsequent ischemic injury of the cervical spinal cord. The pathophysiology is multifactorial, involving direct static compression from osteophytes, disc herniations, and hypertrophied ligaments, compounded by dynamic injury during physiological neck flexion and extension. This repetitive microtrauma leads to endothelial damage within the microvasculature of the spinal cord, inducing ischemia, neuronal apoptosis, and ultimately, irreversible myelomalacia.

The most ubiquitous culprits driving this compression are spondylotic changes. However, we must heavily weigh the presence of Ossification of the Posterior Longitudinal Ligament (OPLL). OPLL is a challenging entity that presents as discrete or continuous ossification of the ligamentous complex, often densely adherent to, or even penetrating, the underlying dura mater. Furthermore, kyphotic deformity—whether primary degenerative or iatrogenic following a previous laminectomy—can severely drape the spinal cord over anterior osteophytic ridges, multiplying the compressive forces. Finally, a congenitally narrowed cervical canal (defined as a Torg-Pavlov ratio of the canal diameter to the vertebral body diameter of 0.8 or less) drastically lowers the threshold at which acquired degenerative changes become symptomatic. In these patients, the functional reserve of the subarachnoid space is non-existent.
Clinical Presentation and Diagnostic Nuances
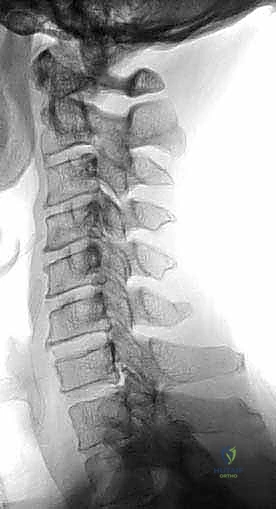
Patients afflicted with CSM often present with a subtle, non-specific clinical picture that is tragically misattributed to the normal aging process or peripheral neuropathies. Upper extremity complaints are hallmark early indicators. Patients frequently report a generalized feeling of clumsiness, an inability to manipulate fine objects such as coins or buttons, deteriorating handwriting, and diffuse, non-dermatomal, glovelike numbness. It is critical to recognize that patients may vehemently deny significant motor weakness early in the disease course, a factor that can mislead the unwary clinician.
Lower extremity symptoms typically manifest as gait instability, a profound sense of imbalance, and a widened base of support. Family members may note that the patient walks "as if intoxicated." The tandem gait test is highly sensitive for revealing this instability, though it is non-specific for localization. Lhermitte’s phenomenon—a sudden, electric shock-like sensation radiating down the spine or into the extremities upon neck flexion—is highly suggestive of posterior column irritation secondary to cord compression. Interestingly, a significant cohort of myelopathic patients will report absolutely no axial neck pain despite harboring advanced spondylosis, underscoring the danger of relying on pain as a primary diagnostic or triaging criterion. Bowel and bladder dysfunction are typically late-stage manifestations, portending a severe and often irreversible degree of cord damage.
Advanced Physical Examination Findings
The physical examination must be a rigorous interrogation of the upper motor neuron pathways. The presence of a Babinski sign (extension of the great toe with fanning of the lesser toes) is a classic indicator of corticospinal tract compromise. The Hoffman sign, elicited by flicking the nail of the middle finger and observing involuntary flexion of the index finger and thumb, is similarly indicative of cervical cord irritability.
The Inverted Radial Reflex is a particularly localizing sign; when tapping the brachioradialis tendon elicits paradoxical flexion of the digits rather than the expected elbow flexion, it strongly suggests a lesion at the C5-C6 level with concurrent cord compression. The Scapulohumeral reflex and the Finger Escape Sign (inability to maintain the ulnar digits in an extended and adducted position over 30 seconds) further corroborate the diagnosis. Crucially, the surgeon must test the Jaw Jerk reflex. A hyperactive jaw jerk implicates pathology cranial to the foramen magnum (e.g., ALS, intracranial mass), serving as a vital differentiator in the diagnostic workup.
Detailed Surgical Anatomy and Biomechanics
Mastery of anterior cervical surgery is predicated upon an intimate, three-dimensional understanding of the fascial planes and the critical neurovascular structures that traverse the anterior neck. The Smith-Robinson approach, utilized for ACCF, exploits the avascular internervous plane between the carotid sheath laterally and the visceral midline structures medially.
Superficial and Deep Cervical Dissection Planes
The superficial anatomy begins with the skin and subcutaneous tissue, followed immediately by the platysma muscle, which is enveloped by the superficial cervical fascia. The platysma can be divided transversely in line with a skin crease incision, or split longitudinally for extensile exposures. Deep to the platysma lies the investing layer of the deep cervical fascia, which encapsulates the sternocleidomastoid (SCM) and trapezius muscles.
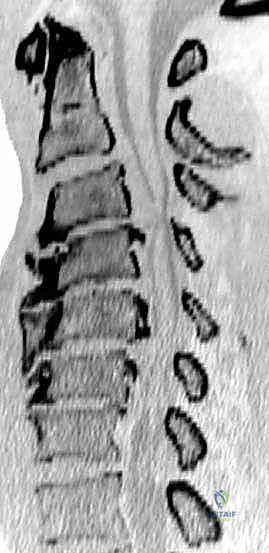
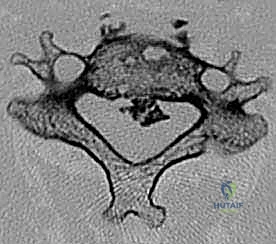
Entering the deep cervical anatomy requires meticulous navigation. The surgeon dissects medial to the SCM, encountering the pretracheal fascia which envelops the strap muscles (sternohyoid, sternothyroid, omohyoid) and the visceral axis (trachea, esophagus, thyroid gland). These visceral structures are gently mobilized and retracted medially. The prevertebral fascia, the final layer, covers the longus colli muscles and the anterior longitudinal ligament. The longus colli muscles are elevated subperiosteally from medial to lateral to expose the vertebral bodies and uncover the uncinate processes, which serve as the definitive lateral landmarks for the corpectomy defect.
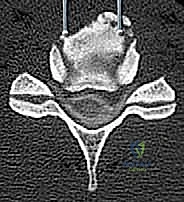
The Danger Zones: Neurovascular Structures
The anterior cervical approach is flanked by "danger zones" that harbor structures entirely intolerant to surgical trauma. Laterally, the carotid sheath contains the common carotid artery, internal jugular vein, and the vagus nerve (CN X). Retraction here must be firm but cautious; excessive pressure can induce bradycardia via the carotid sinus reflex, compromise cerebral perfusion, or cause direct intimal injury to the artery.
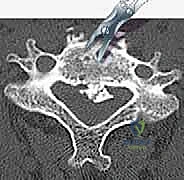
The recurrent laryngeal nerve (RLN), a branch of the vagus, is notoriously susceptible to traction neurapraxia. On the right, it loops beneath the subclavian artery and ascends obliquely into the tracheoesophageal groove. On the left, it loops under the aortic arch and ascends more vertically and medially, generally considered safer during a left-sided approach. However, regardless of the side, minimizing the pressure and duration of medial blade retraction within the tracheoesophageal groove is mandatory to prevent postoperative vocal cord paralysis and dysphagia. Furthermore, the sympathetic chain rests upon the longus colli muscle; dissection extending too far laterally over the muscle belly risks inducing a permanent Horner's syndrome.
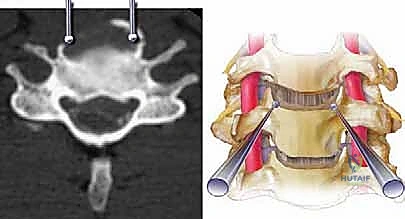
Biomechanical Considerations of the Corpectomy Defect
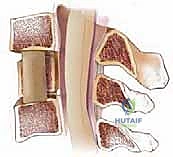
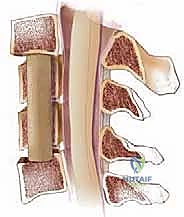
The creation of a corpectomy defect drastically alters the biomechanics of the cervical spine. By resecting the anterior column (the anterior longitudinal ligament, the anterior two-thirds of the vertebral body, and the adjacent intervertebral discs), the surgeon destabilizes the segment in flexion, extension, and rotation. The reconstruction must therefore act as a robust load-sharing device.
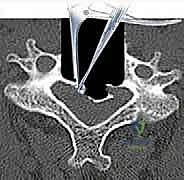
The interpositional strut graft—whether fibular allograft, iliac crest autograft, or a titanium cage—must be meticulously sized. An undersized graft will fail to restore physiological lordosis and is prone to catastrophic dislodgement ("kick-out"). Conversely, an oversized graft subjected to excessive distraction will over-tension the posterior elements, potentially causing an iatrogenic neurological deficit, and dramatically increases the risk of graft subsidence into the adjacent vertebral endplates. The anterior cervical plate functions primarily as a tension-band in extension and a buttress against anterior graft migration, but it cannot overcome poor graft-to-endplate apposition. The endplates must be meticulously prepared—decorticated enough to expose bleeding cancellous bone for osteogenesis, but preserved enough to maintain structural integrity and resist subsidence.
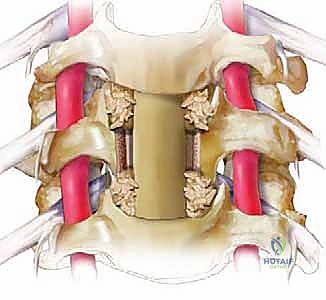
Exhaustive Indications and Contraindications
Patient selection is the ultimate determinant of surgical success. While ACDF is sufficient for pathology confined to the disc space, ACCF is specifically indicated when the compressive pathology is retrovertebral, or when attempting a multi-level ACDF would result in an unacceptably high number of non-union interfaces.
Patient Selection Criteria
The primary indication for ACCF is progressive cervical spondylotic myelopathy or myeloradiculopathy where the anterior column is the primary source of neural compression. This includes massive, broad-based central disc herniations that have migrated behind the vertebral body, severe retrovertebral osteophytosis, and symptomatic Ossification of the Posterior Longitudinal Ligament (OPLL). ACCF is also highly effective for addressing fixed cervical kyphosis, allowing the surgeon to resect the anterior tether and restore lordosis via a carefully contoured strut graft and plate.

In the setting of trauma, ACCF is the procedure of choice for burst fractures of the cervical spine with retropulsed bone fragments causing anterior cord compression. Furthermore, ACCF is indicated for the resection of primary or metastatic tumors involving the vertebral body, as well as for the debridement of vertebral osteomyelitis/discitis where the structural integrity of the vertebral body has been compromised.
Relative and Absolute Contraindications
Absolute contraindications for an isolated anterior corpectomy include severe, uncorrectable osteoporosis, which precludes adequate screw purchase and virtually guarantees graft subsidence. Additionally, if the primary compressive pathology is entirely posterior (e.g., ligamentum flavum hypertrophy) in a patient with preserved cervical lordosis, a posterior approach (laminectomy/laminoplasty) is more appropriate.
Relative contraindications include a history of extensive anterior neck irradiation or prior radical neck dissection, which obliterates the normal tissue planes and dramatically increases the risk of esophageal or neurovascular injury. In patients with more than three levels of involvement (e.g., a 4-level corpectomy), an isolated anterior approach carries an unacceptably high rate of hardware failure and pseudoarthrosis; these patients generally require a combined anterior-posterior (360-degree) reconstruction.
Summary of Surgical Approaches by Pathology
| Pathology / Anatomical Consideration | Preferred Approach (ACDF) | Preferred Approach (ACCF) | Preferred Approach (Posterior) |
|---|---|---|---|
| Soft Disc Herniation (Single Level) | Primary Choice | Contraindicated (Overkill) | Rarely indicated |
| Retrovertebral Osteophytes / OPLL | High risk of incomplete decompression | Primary Choice | Viable if lordosis is preserved |
| Fixed Cervical Kyphosis | Limited correction potential | Primary Choice (Restores alignment) | Contraindicated as standalone |
| Multilevel Stenosis (Lordotic spine) | Viable, but higher non-union risk | Viable for 1-2 bodies | Primary Choice (Laminoplasty) |
| Burst Fracture with Retropulsion | Insufficient | Primary Choice | Supplemental only |
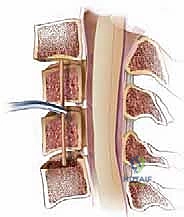
Pre-Operative Planning, Templating, and Patient Positioning
The success of an ACCF is forged in the preoperative planning phase. A meticulous review of advanced imaging dictates the boundaries of the bony resection and the dimensions of the reconstructive hardware.
Advanced Imaging and Modality Selection
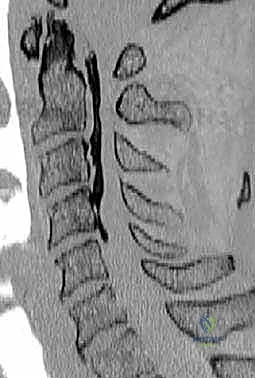
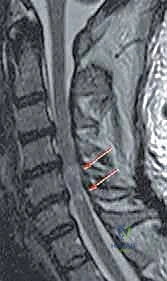
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the soft tissues, delineating the exact level and severity of spinal cord compression, and assessing for intramedullary signal changes. T2-weighted hyperintensity within the cord suggests edema or myelomalacia, while T1-weighted hypointensity is a grim prognostic indicator signifying cystic necrosis and irreversible damage.
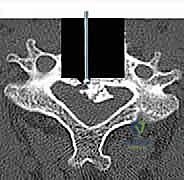
However, MRI alone is insufficient for ACCF planning. A high-resolution, fine-cut Computed Tomography (CT) scan is absolutely mandatory. The CT scan defines the bony architecture, confirming whether a compressive lesion is a soft disc or a calcified osteophyte. Crucially, the CT scan maps the course of the vertebral arteries within the transverse foramina. The surgeon must actively search for anatomical anomalies, such as a medialized vertebral artery or an anomalous entry point into the cervical spine. Failing to identify a medialized vertebral artery preoperatively can lead to catastrophic, life-threatening hemorrhage during the lateral decompression of the corpectomy.
Graft and Hardware Templating
Templating the reconstruction is performed prior to entering the operating theater. For a single-level corpectomy, an iliac crest autograft provides the gold standard for osteoinductivity, though it carries donor site morbidity. For multi-level corpectomies, the structural demands necessitate a fibular allograft strut or a titanium mesh cage packed with local autograft and allograft extenders.
The surgeon must estimate the length of the required strut graft and the corresponding anterior cervical plate. The plate must be long enough to secure adequate bicortical or robust unicortical purchase in the intact vertebral bodies above and below the defect, while avoiding encroachment on the adjacent, healthy intervertebral discs to prevent adjacent segment disease. The plate should be pre-contoured to match the desired cervical lordosis.
Anesthesia, Neuromonitoring, and Optimal Positioning
The procedure is performed under general endotracheal anesthesia. Maintaining adequate mean arterial pressure (MAP > 85 mmHg) is critical throughout the case to ensure spinal cord perfusion, particularly during the decompression phase when reperfusion injury can occur. Continuous intraoperative neuromonitoring, specifically Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs), is mandatory. Baseline signals are obtained prior to positioning and monitored continuously to detect any ischemic or mechanical insult to the spinal cord.
Positioning is a critical vulnerability for the myelopathic patient. The patient is placed supine. The head is secured in Gardner-Wells tongs with 15 to 30 pounds of inline traction, depending on the number of levels involved. This traction stabilizes the head and provides controlled, uniform distraction across the cervical segments, which is invaluable during graft insertion.
SURGICAL WARNING: The neck must be positioned in a neutral to slightly extended posture. The surgeon must review preoperative dynamic radiographs to determine the maximum safe extension; exceeding this limit can buckle the ligamentum flavum into the already compromised canal, causing catastrophic cord injury before the incision is even made. The shoulders are taped caudally to clear the radiographic field for intraoperative fluoroscopy.
Step-by-Step Surgical Approach and Fixation Technique
The execution of an ACCF demands a rhythmic, highly regimented workflow. Every instrument pass must be deliberate, respecting the microscopic margins of error surrounding the dura and the vertebral arteries.
Incision, Exposure, and Preparation
Following fluoroscopic localization, a transverse skin crease incision (for 1-2 level pathology) or an oblique incision along the anterior border of the SCM (for extensile >2 level pathology) is made. The platysma is divided, and the superficial cervical fascia is breached. Blunt dissection utilizing peanuts and Kittner sponges develops the plane between the carotid sheath laterally and the visceral structures medially.
Once the prevertebral fascia is reached, it is incised longitudinally over the midline of the vertebral bodies. The medial borders of the longus colli muscles are identified. Utilizing needle-tip electrocautery, the longus colli muscles are elevated subperiosteally from medial to lateral. This exposure must extend laterally to the uncovertebral joints (joints of Luschka), which serve as the absolute lateral boundaries of our safe working zone. Self-retaining retractors (e.g., Caspar or TrimLine) are placed, ensuring the smooth blades rest medially against the visceral structures and the toothed blades anchor securely under the elevated longus colli muscles laterally.
Discectomies, Corpectomy, and Meticulous Decompression
The procedure begins with thorough discectomies at the cranial and caudal extents of the planned corpectomy. Caspar distraction pins are placed in the vertebral bodies above and below the construct to gently open the disc spaces. The anterior annulus is incised, and the disc material is removed using pituitary rongeurs and curettes down to the posterior longitudinal ligament (PLL).
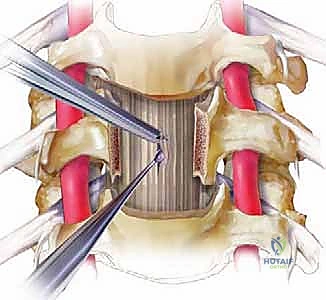
With the cranial and caudal boundaries established, the corpectomy commences. A high-speed burr (e.g., a 4mm or 5mm matchstick or round cutting burr) is utilized to resect the anterior two-thirds of the intervening vertebral body. The surgeon works systematically from anterior to posterior, creating a trough that is approximately 15 to 16 millimeters wide.
SURGICAL WARNING: The width of the corpectomy must not exceed the medial borders of the uncinate processes. Venturing lateral to the uncinate processes places the vertebral arteries at imminent risk of laceration.
As the posterior cortex of the vertebral body is approached, the cutting burr is exchanged for a diamond burr to thin the bone down to an eggshell thickness. This thin cortical layer is then carefully elevated away from the underlying PLL using micro-curettes and Kerrison rongeurs (1mm or 2mm). The PLL is then evaluated. If the PLL is hypertrophied, ossified, or if there is a sequestered disc fragment deep to it, it must be resected. Resection is performed by creating a micro-plane between the dura and the PLL using a nerve hook, followed by sharp division. In cases of severe OPLL where the ligament is fused to the dura, the "floating technique" may be employed—thinning the ossified mass until it floats freely away from the cord, avoiding a dural tear.
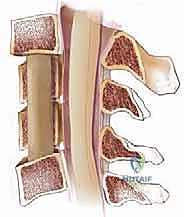
Graft Sizing, Placement, and Anterior Plate Fixation
Following complete decompression, the endplates of the superior and inferior intact vertebral bodies are prepared. The cartilaginous endplate is removed to expose bleeding subchondral bone, but the structural integrity of the bony endplate must be meticulously preserved to prevent graft subsidence.
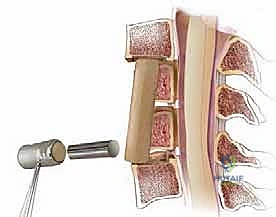
The defect is measured using calipers. The chosen strut graft (fibula allograft or titanium cage) is cut to size, typically adding 1-2 millimeters to the measured defect to ensure a tight interference fit. The traction weight on the Gardner-Wells tongs is temporarily increased, and the cranial and caudal vertebral bodies are distracted using the Caspar pins. The graft is gently tamped into place, ensuring it is recessed 1-2 millimeters deep to the anterior cortex of the adjacent vertebral bodies. The traction is then released, allowing the segment to compress and lock the graft into place under physiological load.
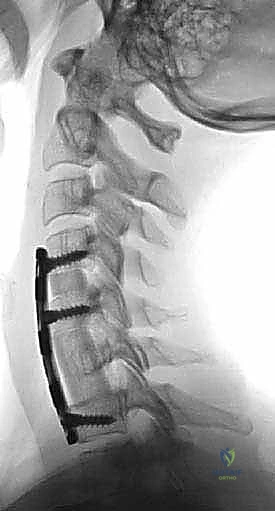
An appropriately sized anterior cervical plate is selected and contoured to maintain cervical lordosis. The plate is positioned symmetrically over the midline and secured using screws. In a dynamic plating system, the screws are placed to allow for micro-subsidence and continuous load-sharing through the graft. Intraoperative fluoroscopy (AP and lateral) is utilized to confirm ideal hardware placement, graft positioning, and restoration of sagittal alignment prior to meticulous hemostasis and layered closure.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, ACCF is associated with a specific profile of severe complications. The surgeon must be prepared to identify and manage these intraoperative and postoperative challenges decisively.
Intraoperative Neurovascular Complications
The most devastating intraoperative complication is a laceration of the vertebral artery, occurring in approximately 0.2% to 0.5% of cases. This typically occurs due to spatial disorientation or an anomalous medialized artery. If arterial bleeding is encountered, the immediate priority is packing the defect with hemostatic agents (e.g., Surgicel, Gelfoam) and applying direct, firm pressure. Blind clamping or cautery is absolutely contraindicated as it will exacerbate the injury and risk compromising the sympathetic chain or exiting nerve roots. If bleeding cannot be controlled with tamponade, emergent endovascular embolization is required.
Dural tears occur more frequently in ACCF than ACDF, particularly in the setting of OPLL (incidence up to 10%). Primary repair of an anterior dural tear is technically exceptionally difficult due
