Anterior Cervical Approaches: An Intraoperative Masterclass for Spinal Fellows
Key Takeaway
This masterclass guides fellows through anterior cervical approaches, including Smith-Robinson and Whitesides techniques. We cover comprehensive surgical anatomy, meticulous patient positioning, and granular intraoperative steps from incision to hardware application. Critical pearls, potential pitfalls, and strategies for complication management and postoperative rehabilitation are emphasized, ensuring a thorough understanding of these vital spinal procedures.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues and fellows. As we transition from the didactic realm into the operative theater, it is imperative to deeply understand the nuances of anterior cervical approaches. Today, we are going to transform your theoretical knowledge into practical, hands-on surgical wisdom. The anterior cervical spine offers a direct, elegant, and highly effective route to ventral pathology, but it demands an uncompromising respect for the intricate regional anatomy. Our goal as spinal surgeons is not merely to perform the procedure, but to master it—understanding every anatomical decision point, anticipating every potential pitfall, and executing the dissection with meticulous precision.
The patho-epidemiology of the cervical spine dictates that the vast majority of compressive lesions—be it spondylotic osteophytes, extruded soft disc herniations, or ossification of the posterior longitudinal ligament (OPLL)—arise anterior to the neural elements. Consequently, the anterior approach, pioneered by Smith and Robinson, and concurrently by Cloward in the 1950s, has become the workhorse of cervical spine surgery. By approaching the spine ventrally, we achieve direct decompression of the neural elements without manipulating the delicate spinal cord, all while preserving the posterior tension band. This biomechanical preservation is paramount for maintaining sagittal balance and preventing post-laminectomy kyphosis, a complication historically rampant before the widespread adoption of anterior techniques.
Epidemiologically, the incidence of anterior cervical procedures, particularly the Anterior Cervical Discectomy and Fusion (ACDF), has seen exponential growth over the last three decades. This surge is driven by an aging demographic, improved advanced imaging modalities (MRI), and the refinement of segmental instrumentation. Pathologies ranging from acute traumatic burst fractures to chronic, insidious cervical spondylotic myelopathy (CSM) are now routinely and safely addressed via this corridor. However, this high volume of surgery must not breed complacency. The anterior neck is a densely packed conduit of vital neurovascular and visceral structures.
As you progress through your fellowship, you will realize that the true mastery of this approach lies in the preoperative planning and the initial exposure. The actual bony decompression and instrumentation, while technically demanding, are often straightforward if the exposure is flawless. Conversely, a poorly planned or hastily executed approach will fight you for the entire duration of the case, increasing the risk of catastrophic complications such as esophageal perforation, major vascular injury, or permanent vocal cord paralysis. Let us delve into the architectural framework that makes this approach both brilliant and perilous.
Detailed Surgical Anatomy and Biomechanics
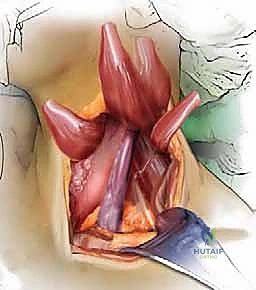
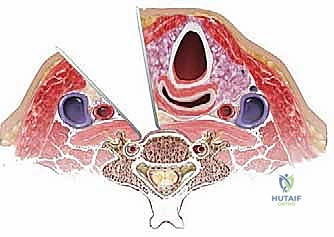
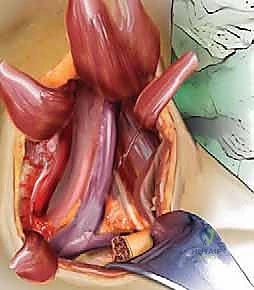
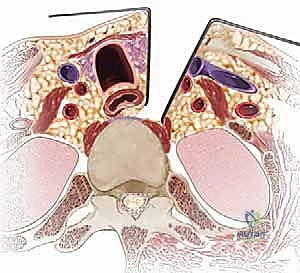
To navigate the anterior cervical spine safely, one must possess a three-dimensional, almost instinctual understanding of the fascial layers and the structures they envelop. The dissection proceeds through a series of distinct anatomical planes, acting as surgical highways. Beneath the skin and subcutaneous fat lies the platysma muscle, innervated by the cervical branch of the facial nerve. Deep to the platysma is the superficial layer of the deep cervical fascia, which encloses the sternocleidomastoid (SCM) and trapezius muscles. The primary surgical corridor is developed between the visceral fascia (enclosing the trachea, esophagus, and thyroid gland) medially, and the carotid sheath (enclosing the common carotid artery, internal jugular vein, and vagus nerve) laterally.

The neurovascular anatomy in this region is notoriously unforgiving. The recurrent laryngeal nerve (RLN) is of paramount concern. There has been considerable historical debate regarding whether approaching from the right or left affects the rate of postoperative RLN palsy. The literature suggests a slightly higher incidence of injury with right-sided approaches. This is due to the variable course of the right RLN, which branches off the vagus nerve higher in the neck, looping under the right subclavian artery, and traversing the surgical field obliquely. On the left, the RLN follows a more consistent, protected path within the tracheoesophageal groove after looping under the aortic arch. Additionally, the superior laryngeal nerve (SLN), which provides motor innervation to the cricothyroid muscle (affecting voice pitch), is at risk during superior exposures (C3-C4) and must be protected by avoiding aggressive superior retraction of the superior thyroid artery and vein.
The vertebral artery represents another critical structure, particularly during lateral decompression, corpectomy, or the Whitesides approach. It typically enters the transverse foramen at C6 (though anomalies entering at C7, C5, or even C4 exist) and ascends vertically (the V2 segment). Medial aberrancy of the vertebral artery, where it loops into the vertebral body, is a rare but potentially fatal anatomical variant that must be identified on preoperative axial CT or MRI. Furthermore, the cervical sympathetic chain lies on the ventral surface of the longus colli muscle. Dissecting too laterally over the longus colli, or placing retractor blades aggressively into the muscle belly, can result in Horner's syndrome (ptosis, miosis, anhidrosis).
Biomechanically, the anterior column of the cervical spine is the primary load-bearing structure, supporting approximately 80% of axial compressive forces. The intervertebral discs, the anterior longitudinal ligament (ALL), and the uncovertebral joints (joints of Luschka) work in concert to provide stability while allowing physiological motion. When we perform an ACDF or corpectomy, we are deliberately destabilizing this load-sharing axis. Therefore, our reconstruction—whether via structural allograft, autograft, or interbody cages supplemented by anterior cervical plating—must meticulously restore the disc height, recreate physiological cervical lordosis, and provide immediate rigid fixation to facilitate arthrodesis. Failure to respect these biomechanical principles will inevitably lead to graft subsidence, pseudoarthrosis, or hardware failure.
Exhaustive Indications and Contraindications
The decision to utilize an anterior cervical approach must be dictated by a careful correlation of the patient's clinical presentation, neurological examination, and advanced imaging findings. The anterior corridor is the optimal choice for direct decompression of ventral pathology.
Clinical Indications for Anterior Approaches
The most frequent indication is cervical radiculopathy secondary to a soft disc herniation or uncovertebral osteophytosis that has failed conservative management. In these cases, a focal ACDF provides excellent outcomes with high fusion rates. Cervical spondylotic myelopathy (CSM) caused by ventral spinal cord compression is another primary indication. When the compression spans multiple levels, the surgeon must decide between a multi-level ACDF, a single or multi-level cervical corpectomy and strut grafting, or a hybrid approach. The anterior approach is also indicated for the stabilization of unstable traumatic injuries, such as flexion-teardrop fractures or burst fractures, where the anterior column is severely compromised. Furthermore, infectious processes (discitis/osteomyelitis) and primary or metastatic neoplastic lesions of the vertebral body are frequently best addressed via anterior debridement, resection, and structural reconstruction.
Absolute and Relative Contraindications
Despite its versatility, the anterior approach is not universally applicable. Absolute contraindications include the presence of an active, untreated systemic infection or a localized anterior neck infection (e.g., deep space neck abscess) that is not the primary target of the surgery. Severe multi-level posterior compression (e.g., massive ligamentum flavum hypertrophy) in the absence of significant ventral pathology is better addressed via a posterior laminectomy or laminoplasty. A critical relative contraindication is continuous, severe Ossification of the Posterior Longitudinal Ligament (OPLL). In cases where the OPLL mass is intimately fused with the dura (the "double-layer" or "tram-track" sign on CT), attempting an anterior resection carries a prohibitively high risk of a massive, unrepairable dural tear and subsequent cerebrospinal fluid (CSF) fistula. In such OPLL cases, a posterior indirect decompression is often safer.
Considerations for Prior Neck Surgery
A history of prior anterior neck surgery demands extreme caution and alters our decision-making algorithm. If a patient has had a previous ACDF and requires an adjacent segment surgery, approaching from the opposite side is tempting to avoid scar tissue. However, it is an absolute medicolegal and clinical requirement to perform a preoperative indirect laryngoscopy (usually via an ENT consultation) to evaluate vocal cord function. If the patient has an asymptomatic, pre-existing unilateral RLN palsy from the first surgery, you must approach the spine from the side of the existing injury. Approaching from the virgin side risks injuring the only functioning RLN, leading to bilateral vocal cord paralysis—a catastrophic complication requiring an emergent, permanent tracheostomy.
| Category | Specific Conditions / Factors | Clinical Rationale & Management |
|---|---|---|
| Primary Indications | Ventral soft disc herniation, Uncovertebral osteophytes | Direct decompression of the exiting nerve root or spinal cord. |
| Primary Indications | Cervical Spondylotic Myelopathy (Ventral compression) | Restoration of CSF space and cord perfusion via ACDF or Corpectomy. |
| Trauma Indications | Burst fractures, Flexion-teardrop fractures | Anterior column reconstruction and stabilization of the tension band. |
| Contraindications | Severe dorsal compression (Ligamentum flavum hypertrophy) | Requires posterior approach (laminectomy/laminoplasty) for adequate decompression. |
| Contraindications | Severe OPLL with dural ossification | High risk of catastrophic dural tear; posterior approach preferred. |
| Surgical Warnings | Prior anterior neck surgery | Mandatory ENT vocal cord check. Must approach from the side of any existing RLN palsy to avoid bilateral paralysis. |
Pre-Operative Planning, Templating, and Patient Positioning
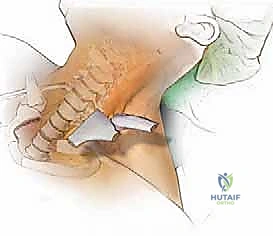
Fellows, I cannot overstate this: the success of your operation is largely determined before you ever scrub in. Preoperative planning is the foundation upon which surgical execution is built. You must meticulously evaluate plain radiographs, MRI, and often a fine-cut CT scan. On the lateral radiograph, assess the patient's anatomy relative to the target levels. For high cervical lesions (C2-C3), evaluate the position of the mandible. If the mandible obscures the target disc space, nasal intubation is highly recommended. This allows the anesthesiologist to keep the mouth closed, permitting the mandible to be retracted superiorly and out of your surgical trajectory. Conversely, for cervicothoracic junction pathology (C7-T1), evaluate the sternal notch. In patients with a short, stocky neck, the sternum may completely block your access to C7-T1, necessitating a manubrial splitting approach.
General Positioning Principles
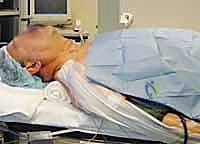
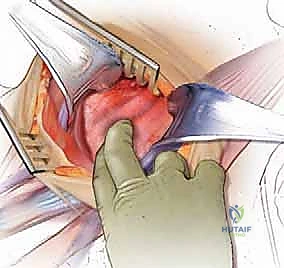
Proper patient positioning dictates the ease of your surgical exposure and patient safety. All anterior cervical approaches begin with the patient in a supine position on a radiolucent operating table. We aim for slight extension of the neck to straighten the cervical lordosis, thereby bringing the anterior vertebral bodies into a more superficial and accessible plane. We achieve this by placing a rolled sheet or a dedicated gel bump horizontally under the patient's scapulae. This not only extends the neck but allows the shoulders to drop posteriorly.
Crucially, you must assess the amount of extension the patient tolerates preoperatively while awake. In patients with severe myelopathy or critical stenosis, excessive neck extension can compress the spinal cord, worsening neurological symptoms or causing irreversible ischemic injury. Never exceed the preoperatively assessed tolerable extension during positioning. Once positioned, the shoulders are gently taped down toward the foot of the bed to facilitate intraoperative lateral fluoroscopy. Exercise extreme caution here: overly aggressive downward traction on the shoulders can stretch the brachial plexus, leading to devastating postoperative upper extremity paresis. Secure the head with a foam doughnut to prevent occipital alopecia or pressure necrosis during prolonged cases.
Neuromonitoring and Final Checks
The use of intraoperative spinal cord monitoring, specifically Somatosensory Evoked Potentials (SSEP) and Motor Evoked Potentials (MEP), is strongly recommended, especially in myelopathic patients or complex reconstructions. Baseline signals must be obtained after positioning but before the incision. If signals degrade upon positioning, the neck extension must be immediately reduced. Remember, however, that neuromonitoring is an adjunct, not a guarantee. It is not completely sensitive to isolated nerve root injuries. Finally, ensure that the fluoroscopy C-arm can easily swing in for a true lateral image without compromising the sterile field. Only when you have perfect imaging, stable neuromonitoring, and optimal positioning should you ask for the scalpel.
Step-by-Step Surgical Approach and Fixation Technique
The execution of the surgical approach must be a fluid, deliberate process. We will cover the standard Smith-Robinson approach, as it is the foundation, followed by specialized approaches for the upper and lower extremes of the cervical spine.
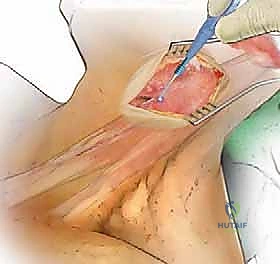
The Classic Smith-Robinson Approach
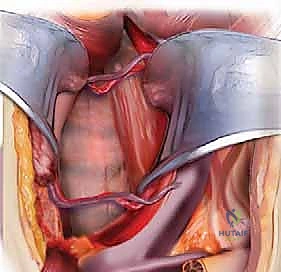
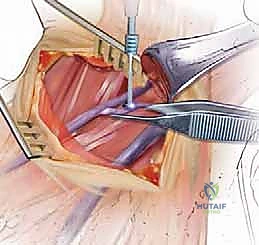
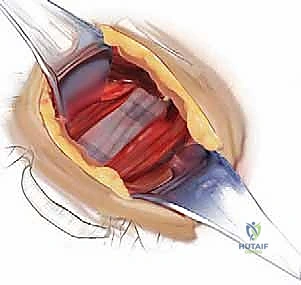
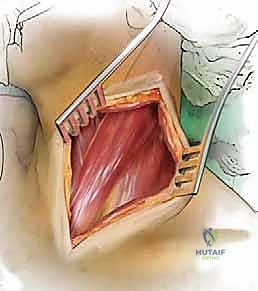
The classic Smith-Robinson approach provides excellent access from C2 down to T1 in most patients with a standard body habitus. An incision is planned using anatomical landmarks: the hyoid bone (C3), the thyroid cartilage (C4-C5), and the cricoid cartilage (C6). A transverse incision placed within a natural skin crease yields the most cosmetically acceptable scar, though a longitudinal incision along the anterior border of the SCM may be required for multi-level corpectomies exceeding three levels. After incising the skin and subcutaneous tissue, the platysma is identified. It is divided in line with the skin incision. Subplatysmal flaps are then elevated superiorly and inferiorly to mobilize the tissue.
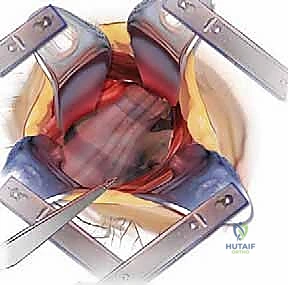
The deep dissection relies on blunt, finger dissection. The anterior border of the SCM is identified, and the deep cervical fascia is incised. Using your index finger, develop the avascular plane between the carotid sheath laterally (feeling the carotid pulse) and the visceral axis medially (trachea and esophagus). The omohyoid muscle may cross the field obliquely; for upper cervical levels, it is retracted inferiorly, and for lower levels, it is retracted superiorly or divided if necessary. Deep in this plane, the prevertebral fascia overlying the longus colli muscles and the anterior longitudinal ligament (ALL) is encountered.
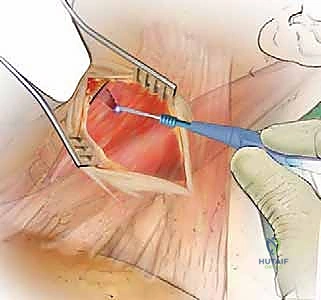
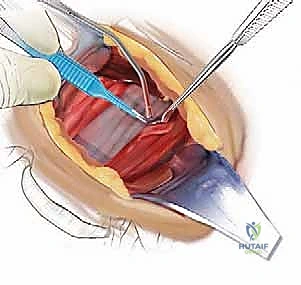
Needle localization is performed to confirm the correct level. Once confirmed, the medial borders of the longus colli muscles are meticulously elevated off the vertebral bodies using Bovie electrocautery and a periosteal elevator. This step is critical: the sympathetic chain lies on the lateral aspect of the longus colli. By staying strictly subperiosteal and medial, you protect the sympathetic chain and avoid Horner's syndrome. Self-retaining retractors (e.g., Caspar or TrimLine) are placed under the elevated longus colli muscles. Ensure the medial blade is securely seated under the muscle to prevent it from slipping and lacerating the esophagus.
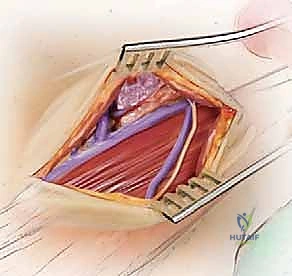
The Lateral Retropharyngeal Approach (Whitesides)
The Whitesides approach is a highly specialized corridor reserved for anterior access to the upper cervical spine (C1-C3). It is indicated for high cervical tumors, infections, or unstable fractures where a posterior approach is contraindicated. The patient is positioned supine with the head turned away from the side of the approach. Nasotracheal intubation on the contralateral side is essential to allow the jaw to be fully closed and retracted.
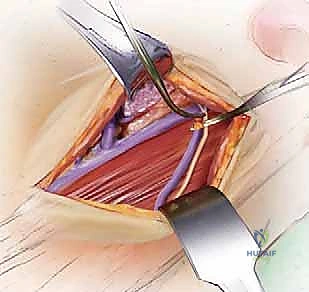
The incision begins at the mastoid process and extends inferiorly along the anterior border of the SCM. The platysma is divided, and the SCM is retracted laterally. The dissection must carefully identify and protect the spinal accessory nerve (CN XI), which enters the SCM superiorly. The internal jugular vein and carotid artery are mobilized and retracted laterally, while the hypoglossal nerve (CN XII) and the superior laryngeal nerve are identified and protected. The retropharyngeal space is entered, providing a direct view of the anterior arches of C1, C2, and C3. This approach carries a high risk of retropharyngeal swelling, necessitating vigilant postoperative airway management, often requiring the patient to remain intubated for 24-48 hours.
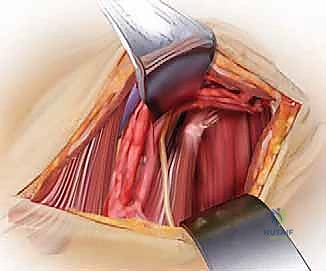
Approaches to the Cervicothoracic Junction
When pathology extends to C7, T1, or T2, standard approaches may be obstructed by the sternum and clavicles. For these levels, we utilize the transmanubrial-transclavicular or the sternal-splitting (median sternotomy) approach. The deep dissection principles remain similar, but the initial bony exposure requires the assistance of a thoracic surgeon.

A median sternotomy provides extensile distal access. The dissection must proceed in a cranial-to-caudal direction. This strategy is vital to identify and protect major crossing vessels, specifically the left brachiocephalic (innominate) vein, which crosses left to right just behind the manubrium. Furthermore, if performing a left-sided approach to this region, the surgeon must be acutely aware of the thoracic duct. It ascends into the neck and arches laterally to empty into the junction of the left internal jugular and subclavian veins. Injury to the thoracic duct results in a chyle leak, a notoriously difficult complication to manage, often requiring specialized diets, prolonged drainage, or reoperation for ligation.
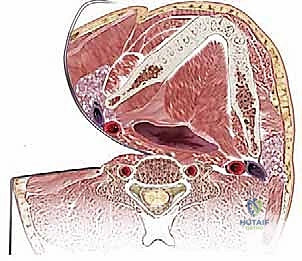
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, complications in anterior cervical surgery do occur. As a fellow, you must not only know how to perform the surgery but how to recognize and manage these adverse events immediately. Dysphagia is the most common postoperative complaint, occurring in up to 70% of patients transiently, and persisting in about 5-10% chronically. It is primarily caused by prolonged retraction of the esophagus, local edema, and hematoma formation. To minimize dysphagia, periodically release the self-retaining retractors during long cases to restore mucosal perfusion, utilize the lowest profile plates possible, and meticulously deflate the endotracheal tube cuff temporarily after retractor placement to allow the esophagus to settle.
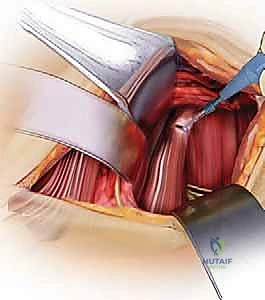
Recurrent Laryngeal Nerve (RLN) palsy is a feared complication, presenting as postoperative hoarseness, vocal fatigue, or in severe cases, aspiration. The incidence ranges from 1% to 11%. If a patient exhibits significant hoarseness postoperatively, an immediate ENT consultation for flexible laryngoscopy is mandatory to document vocal cord mobility. Most neuropraxic injuries recover within 6 to 12 weeks. However, if the nerve was transected, permanent paralysis ensues, which may require vocal cord injection (medialization laryngoplasty) to improve voice quality and prevent aspiration pneumonia.
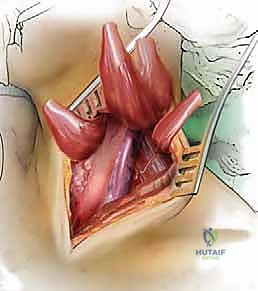
Vertebral artery injury is a catastrophic intraoperative event, with an incidence of approximately 0.3% to 0.5%, usually occurring during aggressive lateral decompression of the uncovertebral joints or during corpectomy. If massive arterial bleeding is encountered from the lateral gutter, immediate action is required. Do not blindly place cautery or instruments into the bleeding site. First, achieve hemostasis with direct, firm tamponade using hemostatic agents (e.g., Gelfoam, Surgicel) and cottonoid patties. Once controlled, obtain an emergent intraoperative angiogram. Depending on the collateral circulation (assessed via the contralateral artery), the injured vessel may require endovascular embolization or, rarely, direct primary repair.
Esophageal perforation is rare (less than 0.5%) but carries a high mortality rate if unrecognized. It can occur from sharp retractor blades slipping, aggressive drilling, or hardware prominence eroding the tissue over time. Intraoperatively, if a tear is suspected, it can be tested by filling the wound with saline and having anesthesia inject air via an orogastric tube (the "tire leak" test). Small intraoperative tears must be repaired primarily in two layers by an ENT or general surgeon, followed by strict NPO status and broad-spectrum antibiotics. Delayed perforations present with fever, neck swelling, and subcutaneous emphysema, requiring emergent surgical exploration and drainage.
| Complication | Estimated Incidence | Primary Prevention & Salvage Management |
|---|---|---|
| Dysphagia | 30-70% (Transient), 5% (Chronic) | Release retractors periodically. Deflate ET cuff. Treatment: Diet modification, speech therapy. |
| RLN Palsy | 1 - 11% | Avoid over-retraction. Identify anatomy. Treatment: ENT consult, vocal cord medialization if permanent. |
| Vertebral Artery Injury | 0.3 - 0.5% | Pre-op CT for anomalies. Careful lateral dissection. Salvage: Tamponade, endovascular embolization. |
| Esophageal Perforation | < 0.5% | Secure retractor placement. Low-profile hardware. Salvage: Primary two-layer repair, NPO, IV antibiotics. |
| Horner's Syndrome | 0.1 - 1% | Subperiosteal dissection of longus colli. Avoid lateral cautery. Treatment: Usually transient, observation. |
Phased Post-Operative Rehabilitation Protocols
The surgery does not end when the skin is closed; it ends when the patient has achieved a solid arthrodesis and returned to functional baseline. Postoperative care must be phased and logically structured. In the immediate postoperative phase (0-2 weeks), the primary focus is on airway observation and hematoma prevention. A surgical drain is typically left in place for 24 hours to prevent retropharyngeal hematoma, which can cause acute airway obstruction. Patients are usually placed in a soft cervical collar for comfort, though a rigid collar (e.g., Aspen or Miami J) may be utilized for multi-level corpectomies or patients with poor bone quality. A dysphagia diet (soft foods, thickened liquids) is initiated and advanced as tolerated.

During the early rehabilitation phase (2-6 weeks), the goal is to promote healing while preventing stiffness. Patients are encouraged to perform gentle, isometric neck exercises and engage in progressive daily walking. Heavy lifting (greater than 10 pounds), overhead activities, and extreme ranges of motion (especially extension) are strictly prohibited. The use of NSAIDs is generally avoided during this phase, as they have been shown to inhibit osteoblastic activity and may increase the risk of pseudoarthrosis, though this remains a topic of debate in the literature.
The intermediate phase (6-12 weeks) is defined by radiographic assessment. At the 6-week mark, upright AP, lateral, and flexion-extension radiographs are obtained to assess hardware integrity and the early stages of bony incorporation. If the hardware is stable and there is no significant lucency around the grafts, the cervical collar is progressively weaned. Patients are then transitioned to formal physical therapy, focusing on restoring cervical range of motion, scapular stabilization, and upper extremity strengthening.
Return to full activity, including contact sports or heavy manual labor, is typically considered between 3 to 6 months postoperatively. This clearance is contingent upon radiographic evidence of solid bony fusion (bridging trabecular bone across the disc space) and a complete resolution of preoperative neurological deficits. In patients who
Clinical & Radiographic Imaging Archive