INTRODUCTION AND HISTORICAL CONTEXT
The restoration of elbow flexion is the primary functional priority in the reconstruction of upper trunk (C5-C6) and extended upper trunk (C5-C7) brachial plexus injuries. Historically, nerve grafting from ruptured spinal nerves or extra-plexal donors (such as the intercostal or spinal accessory nerves) yielded inconsistent results due to the prolonged distance regenerating axons had to travel to reach the target motor endplates.
In 1994, Christophe Oberlin et al. revolutionized the management of brachial plexus avulsions by introducing the concept of the intra-plexal, extra-lesional nerve transfer. The "Oberlin Transfer" involves the transposition of redundant motor fascicles from the intact ulnar nerve to the motor branch of the biceps brachii muscle. Because the coaptation is performed in the mid-arm, mere centimeters from the neuromuscular junction, the time required for axonal regeneration is drastically reduced, preventing irreversible motor endplate degradation.
This guide provides an exhaustive, textbook-level analysis of the single fascicular transfer (Oberlin I) and the subsequent double fascicular transfer (Oberlin II / Mackinnon modification), detailing the surgical anatomy, operative technique, and postoperative rehabilitation protocols.
INDICATIONS AND CONTRAINDICATIONS
Indications
- Upper Trunk Brachial Plexus Injuries (C5-C6): Erb-Duchenne palsy resulting in loss of shoulder abduction and elbow flexion, with preserved lower trunk (C8-T1) hand function.
- Extended Upper Trunk Injuries (C5-C7): Provided that ulnar nerve function (C8-T1) remains robust.
- Timing: Optimal surgical intervention is between 3 to 6 months post-injury. Interventions performed up to 9–12 months may still yield functional recovery, but outcomes degrade significantly after 12 months due to irreversible muscle atrophy and motor endplate fibrosis.
Contraindications
- Lower Trunk Involvement: Any clinical or electrodiagnostic evidence of ulnar nerve impairment (C8-T1) strictly contraindicates its use as a donor.
- Late Presentation: Injuries older than 12–18 months with profound biceps atrophy (consider free functioning muscle transfer instead).
- Pre-existing Ulnar Neuropathy: Severe cubital tunnel syndrome or polyneuropathy that compromises the donor fascicles.
Clinical Pearl: Always perform a rigorous preoperative clinical examination and electromyography (EMG) to confirm that the ulnar nerve has Medical Research Council (MRC) Grade 5 strength and normal conduction velocities before selecting it as a donor.
SURGICAL ANATOMY AND BIOMECHANICS
A profound understanding of the cross-sectional and internal topographical anatomy of the peripheral nerves in the arm is mandatory for successful fascicular dissection.
The Recipient: Musculocutaneous Nerve
The musculocutaneous nerve arises from the lateral cord (C5-C7). It pierces the coracobrachialis muscle and descends obliquely between the biceps brachii (anteriorly) and the brachialis (posteriorly).
* Biceps Motor Branch: Typically branches off the main trunk of the musculocutaneous nerve approximately 13 to 15 cm distal to the acromion. It often presents as one or two distinct branches entering the deep surface of the biceps muscle.
* Brachialis Motor Branch: Arises slightly more distally and deeply to innervate the brachialis muscle.
The Donor: Ulnar Nerve
The ulnar nerve arises from the medial cord (C8-T1). In the upper arm, it travels in the anterior compartment, medial to the brachial artery, before piercing the medial intermuscular septum at the Arcade of Struthers to enter the posterior compartment.
* Fascicular Topography: At the mid-humeral level, the ulnar nerve is highly fasciculated. The motor fascicles destined for the flexor carpi ulnaris (FCU) and the intrinsic muscles of the hand are topographically distinct. The FCU fascicles are generally located in the anterior and medial aspect of the ulnar nerve cross-section.
* Redundancy: The ulnar nerve contains significant redundancy in its innervation of the FCU and intrinsic hand muscles. Harvesting 10% to 20% of the cross-sectional area (specifically targeting the FCU fascicle) does not result in clinically significant donor site morbidity.
PREOPERATIVE PREPARATION AND POSITIONING
Anesthesia
- General anesthesia is required.
- CRITICAL: Long-acting neuromuscular blocking agents (paralytics) must be strictly avoided after induction. Short-acting agents may be used for intubation, but full neuromuscular recovery is mandatory prior to the commencement of the dissection to allow for intraoperative nerve stimulation.
Patient Positioning
- The patient is placed in the supine position.
- The affected upper extremity is abducted to 90 degrees and externally rotated on a radiolucent hand table.
- A sterile tourniquet may be placed high on the arm, though many microsurgeons prefer to operate without a tourniquet to allow for continuous assessment of the vasa nervorum and to avoid ischemic neurapraxia, which can confound electrical stimulation.
Figure A: Patient positioning and planned skin incision on the medial aspect of the arm.
STEP-BY-STEP SURGICAL TECHNIQUE: THE OBERLIN TRANSFER
1. Incision and Superficial Exposure
- Identify the humeral insertion of the pectoralis major tendon.
- Measure approximately 4 cm distal to this insertion on the medial aspect of the arm.
- Outline the origin of the branch of the musculocutaneous nerve to the biceps muscle on the skin.
- Make an 8 to 10 cm longitudinal incision straddling this point, following the medial bicipital groove.
- Incise the superficial fascia and the deep brachial fascia over the biceps muscle.
2. Identification of the Recipient Nerve (Biceps Branch)
- Retract the biceps muscle belly laterally and anteriorly.
- Identify the musculocutaneous nerve situated in the interval between the biceps and the coracobrachialis muscle.
- Carefully dissect the epineurium of the musculocutaneous nerve to identify the specific motor branch(es) entering the biceps muscle.
- Anatomical Variation: Be aware that there are numerous variations in the origin and distribution of this nerve. The biceps branch may arise proximally or present as multiple smaller branches.
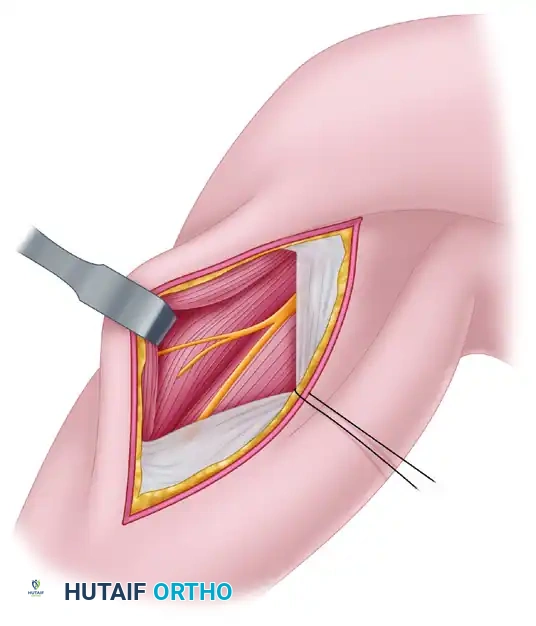
Figure B: Approach to the musculocutaneous nerve. The biceps muscle is retracted laterally to expose the nerve of the biceps muscle.
3. Preparation of the Recipient Nerve
- Once the biceps branch is definitively identified, trace it proximally along the main trunk of the musculocutaneous nerve for approximately 2 cm.
- Split the branch from the main trunk using micro-scissors.
- Transect the biceps branch as proximally as possible to ensure adequate length for a tension-free coaptation with the ulnar nerve fascicle.
Surgical Warning: Ensure that the transection of the biceps branch is clean and perpendicular. Crush injury to the nerve stump will result in disorganized axonal regeneration and neuroma formation.
4. Identification and Dissection of the Donor Nerve (Ulnar Nerve)
- Shift the focus medially to the neurovascular bundle.
- Identify the ulnar nerve, which lies medial to the brachial artery and median nerve.
- Confirm the identity of the ulnar nerve using a handheld electrical nerve stimulator (set at 0.5 to 2.0 mA). Stimulation should elicit robust wrist flexion (FCU) and intrinsic hand muscle contraction.
- Bring the operating microscope into the surgical field.
5. Intraneural Dissection and Fascicle Selection
- Under microscopic magnification (typically 10x to 15x), perform a longitudinal epineurotomy on the anterior aspect of the ulnar nerve over a distance of 3 to 4 cm.
- Carefully separate the internal fascicular groups. The vascular pedicle of the nerve usually has a transverse orientation and can generally be preserved during this longitudinal dissection.
- Using the nerve stimulator at a low threshold (0.5 mA), systematically stimulate the isolated fascicles.
- Identify a fascicle that produces strong, isolated contraction of the flexor carpi ulnaris (FCU) without significant intrinsic hand muscle activation. This is typically located anteromedially.
- Once identified, dissect this motor fascicle distally to gain sufficient length, and transect it distally.
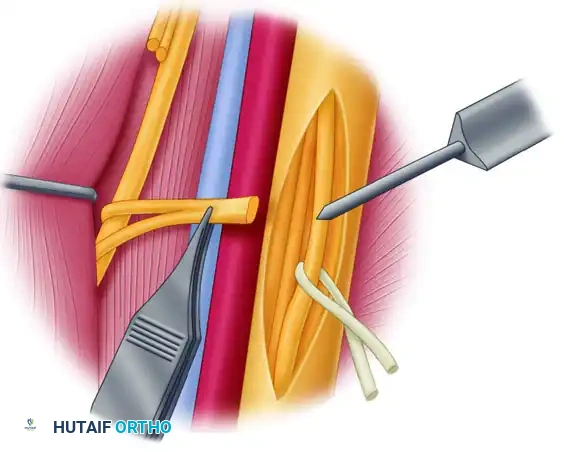
Figure C: Microscopic intraneural dissection of the ulnar nerve. Identification of the selected motor fascicle is confirmed by means of electrical stimulation.
6. Nerve Coaptation
- Pivot the distally transected ulnar nerve fascicle toward the proximally transected biceps motor branch.
- Ensure that the coaptation site is completely tension-free throughout a full range of elbow motion. If tension exists, further proximal dissection of the biceps branch or distal dissection of the ulnar fascicle is required.
- Perform an end-to-end epineurial/perineurial repair using three to four interrupted 9-0 or 10-0 nylon sutures under the microscope.
- Supplement the repair with a thin layer of fibrin glue to seal the coaptation site and prevent axonal escape.
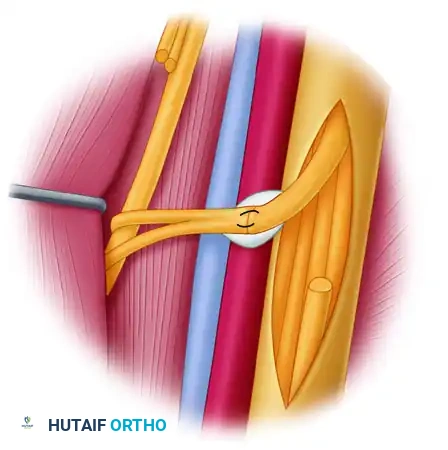
Figure D: The completed fascicle transfer. The distal end of the ulnar nerve fascicle is coapted to the nerve of the biceps muscle using nylon sutures and fibrin glue.
THE DOUBLE FASCICULAR TRANSFER (MACKINNON MODIFICATION)
While the classic Oberlin transfer effectively restores biceps function, true elbow flexion biomechanics rely heavily on the brachialis muscle, which is the primary workhorse for elbow flexion across all forearm positions (pronation, neutral, and supination).
To optimize strength and reliability, Mackinnon and colleagues introduced the double fascicular transfer. This technique utilizes redundant fascicles from both the ulnar and median nerves to reinnervate the biceps and brachialis muscles, respectively.
Technique for Double Transfer
- Ulnar to Biceps: Performed exactly as described above. A redundant FCU fascicle from the ulnar nerve is transferred to the biceps branch of the musculocutaneous nerve.
- Median to Brachialis:
- Identify the median nerve, which lies lateral to the ulnar nerve and anterior to the brachial artery.
- Identify the brachialis motor branch of the musculocutaneous nerve, which lies deeper and slightly distal to the biceps branch.
- Perform an epineurotomy on the median nerve.
- Using electrical stimulation, identify a redundant motor fascicle innervating the flexor carpi radialis (FCR) or pronator teres (PT).
- Transect this fascicle distally and coapt it to the proximally transected brachialis motor branch using 9-0 nylon and fibrin glue.
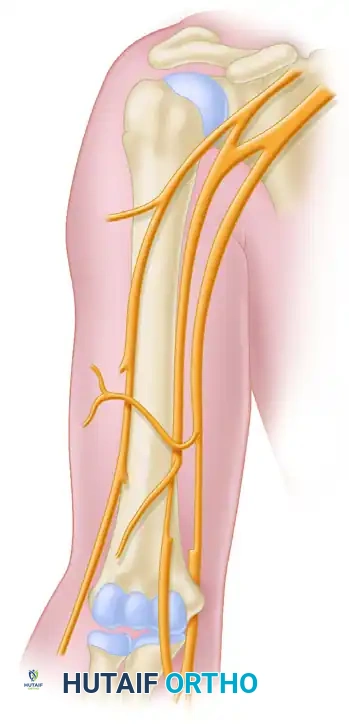
Figure 62-23A: Schematic representation of the double fascicular transfer. The redundant flexor carpi ulnaris (FCU) fascicle from the ulnar nerve is transferred to the biceps brachii branch.
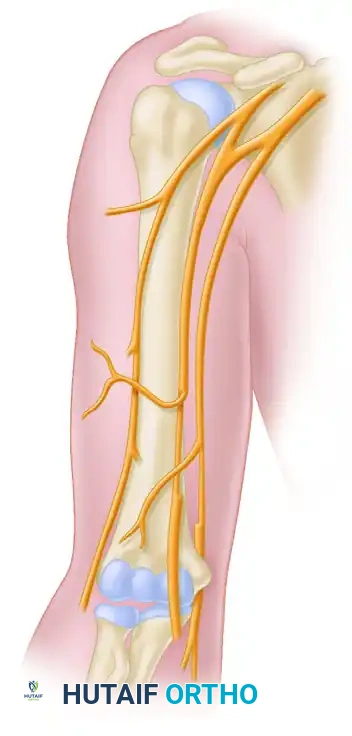
Figure 62-23B: The redundant flexor carpi radialis (FCR) fascicle from the median nerve is transferred to the brachialis branch, completing the double fascicular transfer.
Clinical Pearl: The double fascicular transfer is now considered the gold standard for restoring elbow flexion in C5-C6 avulsion injuries. It provides superior pinch strength and greater overall elbow flexion torque compared to the single Oberlin transfer.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a nerve transfer relies as much on meticulous postoperative rehabilitation and cortical relearning as it does on the microsurgical repair.
Phase I: Protection and Immobilization (Weeks 0-3)
- The upper extremity is immobilized in a long-arm splint or sling with the elbow flexed to 90 degrees and the forearm in neutral rotation.
- Strict avoidance of elbow extension and shoulder abduction to prevent tension on the microscopic coaptation sites.
- Passive range of motion (ROM) of the wrist and digits is encouraged immediately to prevent stiffness and maintain gliding of the donor nerve tendons (FCU/FCR).
Phase II: Early Mobilization (Weeks 3-8)
- The splint is discontinued.
- Gentle, gravity-eliminated active-assisted range of motion (AAROM) of the elbow is initiated.
- Avoid aggressive passive stretching into full elbow extension.
Phase III: Motor Relearning and Cortical Plasticity (Months 3-12)
- Axonal regeneration occurs at a rate of approximately 1 mm per day. Given the proximity of the transfer, early signs of reinnervation (EMG potentials or flicker of movement) may be seen as early as 3 to 5 months.
- Biofeedback and Donor Activation: This is the most critical phase. The patient must be taught to activate the donor muscle to fire the recipient muscle. For the Oberlin transfer, the patient is instructed to forcefully flex the wrist (activating the FCU) while simultaneously attempting to flex the elbow.
- Over time, through neuroplasticity, the cerebral cortex remaps this pathway. The patient will eventually be able to flex the elbow independently without needing to consciously flex the wrist.
Phase IV: Strengthening (Months 12+)
- Once independent elbow flexion reaches MRC Grade 3 (anti-gravity), progressive resistance training is initiated.
- Maximum functional recovery may take up to 24 months.
COMPLICATIONS AND PITFALLS
- Failure of Reinnervation: Can occur due to tension at the coaptation site, poor fascicle selection (e.g., selecting a sensory fascicle instead of a motor fascicle), or delayed surgical intervention (>12 months).
- Donor Site Morbidity: Transient weakness or paresthesias in the ulnar or median nerve distributions may occur due to intraoperative manipulation. Permanent clinically significant weakness of the FCU or FCR is exceedingly rare due to the synergistic innervation of the wrist flexors.
- Neuroma Formation: Results from inadequate coaptation or escaping axons. Meticulous microsurgical technique and the use of fibrin glue minimize this risk.
- Co-contraction: Incomplete cortical remapping may result in simultaneous contraction of the wrist flexors and elbow flexors, though this rarely limits functional utility.
CONCLUSION
The transfer of ulnar nerve fascicles to the biceps motor branch remains a cornerstone in the surgical management of upper brachial plexus injuries. By adhering to strict microsurgical principles, utilizing intraoperative electrical stimulation for precise fascicular mapping, and employing rigorous postoperative rehabilitation, orthopedic surgeons can reliably restore life-altering elbow flexion to patients suffering from devastating paralytic injuries. The evolution to the double fascicular transfer further cements this technique as the gold standard in modern peripheral nerve reconstruction.
