Peripheral Nerve Suturing: Advanced Surgical Techniques and Repair Protocols

Key Takeaway
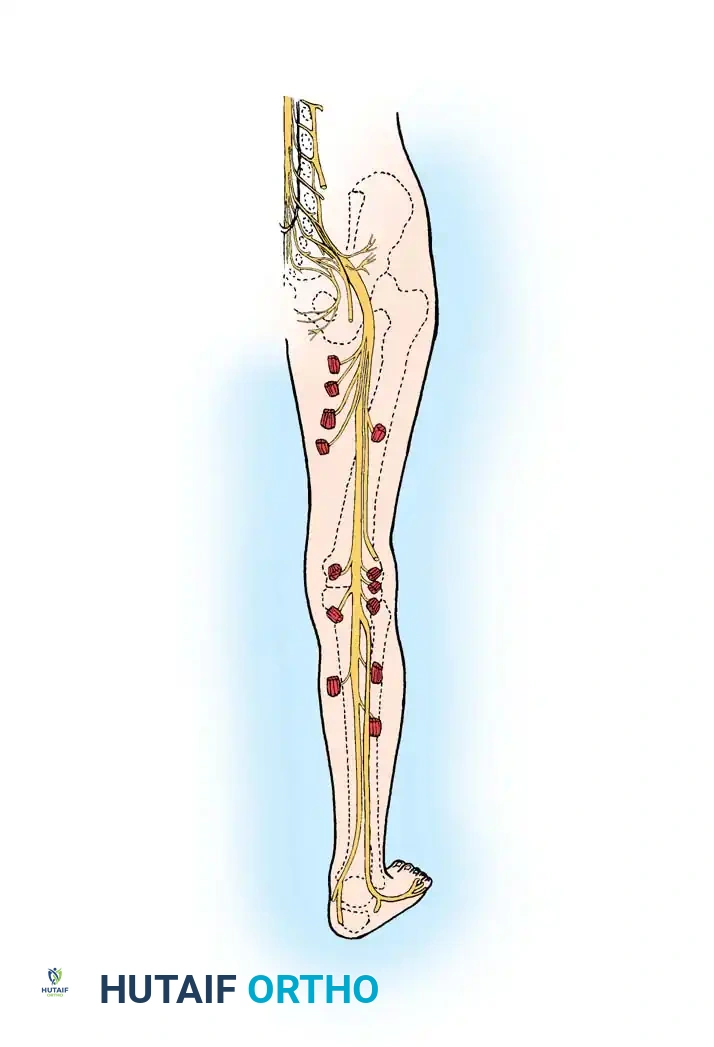
Peripheral nerve suturing requires meticulous microsurgical technique and a profound understanding of intraneural topography. Successful neurorrhaphy depends on tension-free apposition, accurate fascicular alignment, and appropriate timing of repair. This guide details the indications, surgical approaches, and postoperative protocols for primary and secondary nerve repairs, emphasizing the critical role of magnification and precise suture placement in restoring motor and sensory function.
INTRODUCTION TO PERIPHERAL NERVE SUTURING
The restoration of peripheral nerve continuity is one of the most exacting disciplines within operative orthopaedics and hand surgery. The principles that govern the suturing of major peripheral nerves apply equally to the delicate peripheral nerves of the hand and digits. Successful neurorrhaphy is not merely a mechanical approximation of tissue; it is a biological facilitation of axonal regeneration.
To achieve optimal functional recovery, the orthopaedic surgeon must synthesize a deep understanding of intraneural topography with flawless microsurgical technique. Critical considerations in nerve repair include distinguishing between mixed versus pure motor or sensory nerves, understanding the internal fascicular arrangement, selecting the appropriate surgical incisions, achieving tension-free apposition through strategic mobilization, and utilizing appropriate magnification and suture materials.
Careful, tissue-respecting technique is paramount. The ultimate goal is to provide the best possible anatomical restoration, thereby minimizing axonal escape and maximizing end-organ reinnervation.
INTRANEURAL TOPOGRAPHY AND FASCICULAR ANATOMY
A profound comprehension of intraneural anatomy is the foundation of successful nerve repair. Peripheral nerves are not homogenous cables; they possess a complex, dynamic internal architecture that changes along their longitudinal course. The internal arrangement of the nerve dictates the type of repair (epineurial vs. perineurial) that will yield the best outcome.
As described by Millesi, peripheral nerves can be classified into four distinct fascicular patterns. Recognizing these patterns is essential for accurate fascicular alignment during neurorrhaphy.
The Four Fascicular Patterns
1. Monofascicular Pattern
A single, large fascicle comprises the entire nerve. This pattern is relatively rare in major nerve trunks but can be seen in terminal branches.

Figure 68-5A: Monofascicular arrangement, characterized by a single large fascicle enveloped by epineurium.
2. Oligofascicular Pattern
The nerve is composed of a few (typically two to ten) relatively large fascicles. This arrangement is highly characteristic of the common digital and proper digital nerves in the palm and digits. Because of this simple internal arrangement, their pure sensory function, and the short distance to the end organ, the prognosis after repair of digital nerves is generally excellent.
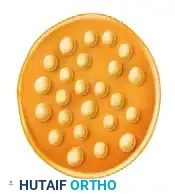
Figure 68-5B: Oligofascicular arrangement, typical of digital nerves.
3. Polyfascicular Pattern with Group Arrangement
The nerve contains numerous fascicles that are clustered into distinct groups or bundles, separated by abundant interfascicular epineurium. This pattern is classically found in the median and ulnar nerves at the level of the wrist. Grouped fascicular repair is often indicated here to ensure motor and sensory pathways are correctly matched.

Figure 68-5C: Polyfascicular with group arrangement, commonly seen in major mixed nerves at the wrist.
4. Polyfascicular Pattern without Group Arrangement
The nerve consists of many small fascicles scattered randomly throughout the epineurial connective tissue without distinct grouping. This pattern presents the greatest challenge for accurate alignment, as matching individual fascicles is nearly impossible.

Figure 68-5D: Polyfascicular without group arrangement, presenting significant challenges for topographical alignment.
Clinical Pearl: The longitudinal epineurial blood vessels are the most reliable macroscopic guide to rotational alignment. Always map these vessels before resecting the neuromatous ends to ensure the nerve is not inadvertently rotated during repair.
INDICATIONS AND TIMING OF NERVE REPAIR
The timing of nerve repair is a critical determinant of functional outcome. While primary repair (within 48 hours of injury) is generally preferred for sharp, clean lacerations, numerous clinical scenarios dictate a delayed or secondary approach.
Primary Repair
Primary repair is indicated for sharp transections with minimal crush or avulsion components, provided the wound is clean and the patient is stable. Immediate repair prevents retraction of the nerve ends and utilizes the optimal biological window before significant scarring occurs.
Indications for Delayed or Secondary Repair
Secondary repair (performed 3 weeks or more after injury) is often necessary and, in many cases, biologically advantageous. Delaying the repair allows for Wallerian degeneration to clear the distal stump and for the extent of intraneural scarring to demarcate clearly.
Secondary repair is strictly indicated under the following conditions:
- Extensive Soft Tissue Injury: If there is massive soft tissue loss or avulsion, nerve repair is secondary. It is indicated only after stable, well-vascularized skin coverage has been obtained.
- Wound Contamination: An extensively contaminated wound (e.g., agricultural injuries, bite wounds) requires delay. Infection will destroy a nerve repair and compromise wound closure. Serial débridements must be completed before neurorrhaphy is attempted.
- Polytrauma and Life-Threatening Injuries: In the presence of multiple limb or systemic injuries, aggressive management of life-threatening conditions, vascular repair, and bone stabilization take absolute precedence over nerve repair.
- Crush Injuries: Crush mechanisms cause unpredictable zones of intraneural damage that cannot be accurately assessed acutely. Waiting 3 to 6 weeks allows clear demarcation between healthy nerve and fibrotic scar tissue.
- Traction Injuries: Similar to crush injuries, traction causes extensive, longitudinal intraneural fibrosis. Acute exploration often reveals intact epineurium with ruptured fascicles (axonotmesis or neurotmesis in continuity). Delayed exploration is mandatory.
- Unknown Prior Surgical Intervention: If a patient's initial care was managed by another surgeon and the extent of the nerve injury or the quality of the repair is unknown, secondary exploration is warranted. This ensures that a skillful, tension-free repair has been achieved.
Surgical Warning: Never attempt a primary repair on a nerve that has sustained a severe stretch or crush injury. The zone of injury extends far beyond the macroscopic damage. Suturing traumatized nerve ends will inevitably result in massive neuroma formation and complete failure of regeneration.
The "Window of Opportunity" for Secondary Repair
Although historical data from World War II suggested that useful sensation could occasionally return after repairs performed up to 2 years post-injury, this is the exception, not the rule. The best results for secondary repair occur if the neurorrhaphy is performed within the first 3 months of injury. Return of motor function after excessive delay is highly unpredictable due to irreversible motor endplate degradation and muscle atrophy.
PREOPERATIVE PLANNING AND SURGICAL APPROACH
Patient Positioning and Equipment
The procedure must be performed under tourniquet control to ensure a bloodless field, which is absolute for microsurgical visualization.
* Magnification: Magnification is non-negotiable. In the palm and fingers, 2.5× to 4.5× magnifying loupes are usually sufficient for accurate repair of oligofascicular nerves. More proximally (wrist and forearm), or when repairing terminal branches distal to the distal interphalangeal (DIP) joint, an operating microscope is highly recommended to achieve satisfactory anatomical repair.
* Instrumentation: Specialized microsurgical instruments, including jeweler's forceps, microscissors, and microneedle holders, are required.
Incisions and Exposure
Incisions to expose and mobilize the nerve must adhere strictly to the proven principles of hand surgery:
1. Avoid Flexion Creases: Incisions should never cross flexion creases at right angles to prevent debilitating flexion contractures. Bruner zigzag incisions or mid-lateral incisions are preferred in the digits.
2. Preserve Vascularity: Skin flaps must be elevated with full thickness to avoid devascularization.
3. Extensile Approach: The incision must be extensile, allowing adequate proximal and distal exposure without creating additional neurovascular injury.
Nerve Mobilization and Ischemia
To achieve a tension-free repair, the nerve must be mobilized proximally and distally. The exact extent to which a peripheral nerve can be mobilized without creating critical ischemia is debated; however, the longitudinal intrinsic blood supply of the nerve is robust. Generally, within the digits, palm, and wrist, extensive mobilization of the nerve from its surrounding mesoneurium is safe and insufficient to cause ischemic harm, provided the segmental vessels are respected where possible.
SURGICAL TECHNIQUE: STEP-BY-STEP NEURORRHAPHY
1. Preparation of the Nerve Ends
The most critical step in delayed repair is the preparation of the nerve stumps. The neuroma (proximal stump) and glioma (distal stump) must be resected back to healthy, viable fascicles.
Using a fresh scalpel blade or specialized nerve cutting forceps, serial slices of the nerve end are taken until normal fascicular architecture "pouts" or "mushrooms" out from the epineurium. The presence of healthy, bleeding epineurial vessels and distinct, unscarred fascicles confirms adequate resection.
2. Achieving Tension-Free Apposition
Tension is the primary enemy of nerve regeneration. Tension causes ischemia, stimulates fibroblastic proliferation, and leads to scarring at the repair site.
* Mobilization of the nerve ends is the first line of defense against tension.
* Joint positioning (e.g., mild wrist flexion for median nerve repair) can be utilized, but extreme positioning should be avoided as it will lead to traction injury when the joint is eventually mobilized.
* If tension-free apposition cannot be achieved with an 8-0 suture, a nerve graft (e.g., sural nerve) or nerve conduit is strictly indicated.
3. Suture Material Selection
Suture materials reflect the delicate nature of the tissue and the minimal tension required.
* In the forearm, wrist, and hand, 8-0 and 9-0 monofilament nylon sutures are the standard of care.
* Monofilament nylon is preferred due to its high tensile strength, smooth passage through tissue, and minimal inflammatory tissue reactivity.
4. Types of Nerve Repair
The choice of repair technique depends on the fascicular anatomy of the nerve at the level of injury.
Epiperineurial (Grouped Fascicular) Repair
For polyfascicular nerves with group arrangements (e.g., median and ulnar nerves at the wrist), a combination epiperineurial-perineurial neurorrhaphy is preferred.
1. The external epineurium is retracted slightly.
2. Matching groups of fascicles (motor to motor, sensory to sensory) are identified based on size, topography, and surface vessels.
3. Sutures (9-0 or 10-0) are placed through the interfascicular epineurium and the perineurium of the grouped fascicles to coapt them directly.
4. The external epineurium is then loosely approximated with 8-0 sutures to relieve tension on the internal repair.
Perineurial (Fascicular) Repair
For oligofascicular nerves (e.g., digital nerves), a pure perineurial or epineurial repair is sufficient.
1. The needle is passed through the epineurium, catching a small bite of the perineurium of the underlying fascicle.
2. Typically, 2 to 4 sutures are sufficient for a digital nerve.
3. Care must be taken not to pass the needle through the center of the fascicle, which would damage the endoneurial contents and axons.
Pitfall: Over-suturing the nerve creates a foreign body reaction and mechanical barrier to axonal sprouting. Use the minimum number of sutures necessary to achieve precise alignment and prevent fascicular buckling.
POSTOPERATIVE MANAGEMENT AND REHABILITATION
The surgical repair is only the first phase of treatment; meticulous postoperative management is essential for a successful outcome.
Immobilization Phase
Immediately postoperatively, the limb is immobilized in a bulky, non-compressive splint. The joints are positioned to minimize tension on the repair site (e.g., wrist in 20-30 degrees of flexion for volar wrist repairs).
* Immobilization is typically maintained for 3 to 4 weeks. This allows the epineurial healing to gain sufficient tensile strength to withstand physiological gliding.
Mobilization and Gliding Phase
At 3 to 4 weeks, the splint is removed, and a carefully supervised rehabilitation program begins.
* Protected Mobilization: Active and passive range of motion exercises are initiated to prevent joint stiffness and tendon adhesions.
* Nerve Gliding: Specific nerve gliding exercises are introduced to prevent the nerve from adhering to the surrounding scar bed. A tethered nerve will undergo traction micro-trauma during normal limb movement, severely compromising regeneration.
Sensory Re-education and Motor Retraining
As regenerating axons reach the distal end organs (advancing at approximately 1 mm per day, or 1 inch per month), the patient will experience altered sensation (Tinel's sign will advance distally).
* Sensory Re-education: Once protective sensation begins to return, a formal sensory re-education program is critical. This involves training the brain to interpret the new, often distorted, afferent signals. Techniques include texture discrimination, object identification, and localization exercises.
* Motor Retraining: For mixed or motor nerves, electrical stimulation may be used early to maintain muscle bulk, followed by biofeedback and progressive resistance exercises once voluntary motor unit recruitment is detected.
CONCLUSION
The suturing of peripheral nerves demands a rigorous adherence to microsurgical principles, a deep respect for intraneural anatomy, and precise clinical judgment regarding the timing of intervention. By ensuring tension-free apposition, accurate fascicular alignment, and appropriate postoperative rehabilitation, the orthopaedic surgeon can optimize the biological environment for axonal regeneration, thereby maximizing the restoration of critical motor and sensory functions in the injured extremity.
📚 Medical References
- peripheral nerve suturing in monkeys, Plast Reconstr Surg 46:31, 1970.
- Greene TL, Steichen JB: Digital nerve grafting using the dorsal sensory branch of the ulnar nerve, J Hand Surg 10B:37, 1985.
- Guelinckx PJ, Carlson BM, Faulkner JA: Parallel-fi bered muscles transplanted with neurovascular repair to bipennate muscle sites in rabbits, Plast Reconstr Surg 89:290, 1992.
- Hakstian RW: Funicular orientation by direct stimulation: an aid to peripheral nerve repair, J Bone Joint Surg 50A:1178, 1968.
- Hakstian RW: Perineural
You Might Also Like