CHONDROMALACIA OF THE PATELLA: PATHOLOGY AND PRINCIPLES
The term chondromalacia of the patella was first introduced into the orthopedic literature in 1928 by Aleman to describe the specific macroscopic and microscopic degeneration of the articular cartilage of the patella. Over the decades, a significant nosological error has permeated clinical practice, wherein "chondromalacia" has erroneously become synonymous with anterior knee pain.
Numerous umbrella terms have been proposed to describe this clinical presentation, including patellofemoral syndrome, patellofemoral arthralgia, extensor mechanism dysplasia, and anterior knee pain syndrome. However, these clinical descriptors do not accurately reflect the underlying histopathological condition. It is imperative for the orthopedic surgeon to recognize that some patients with minimal articular surface changes experience debilitating patellofemoral joint symptoms, while conversely, patients with profound articular degradation may remain entirely asymptomatic. Therefore, chondromalacia of the patella must be strictly reserved to describe a pathological condition of the cartilage, not a generalized clinical syndrome.
Pathophysiology and Histological Cascade
The fundamental pathological lesion in chondromalacia of the patella diverges significantly from that of primary osteoarthritis or traumatic arthritis. In osteoarthritis, the initial pathological changes manifest on the superficial surface of the cartilage, characterized by a loss of continuity of the transverse collagen fibers, followed by gross fibrillation.
Conversely, in chondromalacia of the patella, the initial lesion is a biochemical and structural alteration of the ground substance and collagen fibers within the deep, basal levels of the cartilage. It is fundamentally a disorder of the deep layers; the superficial articular surface is involved only in the late stages of its development. Early changes are tangible (softening) but not visually apparent, as the surface remains smooth and intact.
Clinical Pearl: The hallmark of early chondromalacia is a palpable softening or "blistering" of the cartilage with an intact superficial layer. Probing during arthroscopy is essential, as visual inspection alone will miss Grade I lesions.
This deep-layer degradation is attributed to a localized decrease in sulfated mucopolysaccharides within the ground substance. Histologically, this is demonstrated by a distinct loss of basophilia on hematoxylin and eosin (H&E) preparations.
These degenerative changes most commonly initiate at one of two specific anatomical sites in the deep cartilage layer:
1. An area approximately 1 cm in diameter astride the longitudinal ridge that separates the lateral facet from the medial facet.
2. An area straddling the inferior portion of the central ridge.
These two epicenters are anatomically proximate and frequently become confluent as the disease progresses.
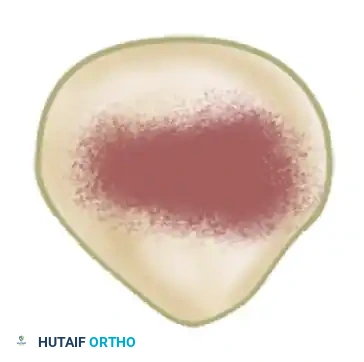
If these specific noncontact areas were never subjected to the mechanical shear and compressive stresses of articulation, the localized chondromalacia might remain clinically silent. However, when these softened areas articulate against the femoral trochlea—usually at extreme ranges of knee flexion—the structurally compromised cartilage is mechanically inadequate to support the collagen framework. The complex ultrastructure begins to fragment, leading to the next phase of degeneration: fibrillation.
Stages of Basal Degeneration
The progression of basal degeneration in the articular cartilage of the patella follows a predictable histological and morphological sequence:
Normal Cartilage Architecture:
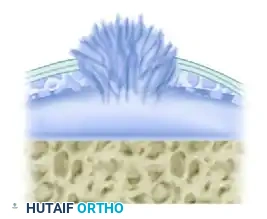
Fasciculation Stage 1:
Initial breakdown of the deep collagen network with early ground substance depletion.

Fasciculation Stage 2:
Progressive disorganization of the intermediate zone of collagen fibers.
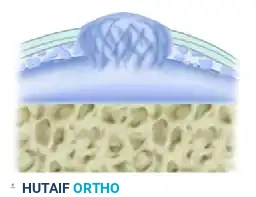
"Blister" Stage:
The deep structural failure leads to a macroscopic swelling or "blistering" of the cartilage, rendering it mechanically incompetent under shear stress.
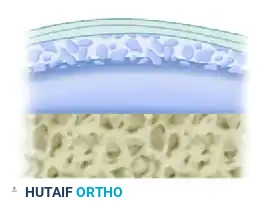
These changes may deepen progressively until all layers of the cartilage are affected, extending down to the subchondral bone plate.
The Genesis of Patellofemoral Pain
Because articular cartilage is entirely devoid of nociceptive nerve endings, the cartilage itself cannot be the direct source of pain. The pain associated with chondromalacia originates primarily from two sources:
1. Subchondral Bone: The disorganization of the fibrous structure in the intermediate zone of collagen fibers compromises the cartilage's energy-absorbing function. This subjects the highly innervated subjacent subchondral bone plates to abnormal pressure variations and microtrauma, acting as a direct pain stimulus.
2. Synovium: As the cartilage fibrillates, articular cartilage debris is shed into the joint space. This debris is phagocytized by the synovial lining, producing a secondary chemical synovitis, resulting in effusion, capsular distension, and pain.
CLASSIFICATION AND ETIOLOGY
Macroscopic Classification
Several classification systems exist, but the most universally accepted surgical grading is based on the macroscopic appearance of the cartilage during arthroscopy:
- Grade I: Minimal articular cartilage changes. The cartilage exhibits localized softening (chondromalacia) with minimal or no break in the surface. A blunt arthroscopic probe pressed onto the surface will sink into the cartilage, which may appear slightly discolored, swollen, or blistered.
- Grade II: Characterized by an area of superficial fibrillation or fissuring, creating an irregular, "velvet-like" surface. The defect is less than 1.25 cm in diameter.
- Grade III: Definite, deep fibrillation with fissuring extending down to, but not exposing, the subchondral bone. This presents the classic arthroscopic "crab meat" appearance. The defect is typically greater than 1.25 cm in diameter.

- Grade IV: Full-thickness cartilage loss. The articular cartilage has completely disappeared in focal areas, allowing exposure, eburnation, and erosion of the underlying subchondral bone.
Etiological Factors
The etiology of chondromalacia patellae is multifactorial and can be broadly categorized into biomechanical, biochemical, iatrogenic, and degenerative causes.
Biomechanical Causes (Acute):
* Dislocation of the patella with an associated chondral or osteochondral shear fracture.
* Direct trauma (e.g., a direct blow or fall onto the flexed knee, driving the patella into the trochlea).
* Fracture of the patella resulting in incongruous articular surfaces.
Biomechanical Causes (Chronic):
* Recurrent subluxation or dislocation of the patella (often secondary to trochlear dysplasia, patella alta, excessive femoral anteversion, or external tibial torsion).
* Increased Quadriceps angle (Q-angle).
* Quadriceps muscle imbalance (e.g., vastus medialis obliquus weakness or dysplasia).
* Posttraumatic malalignment following a malunited femoral shaft fracture.
* Excessive lateral pressure syndrome (ELPS).
* Meniscal injury leading to an alteration of the synchronous pattern of patellar tracking.
Biochemical and Systemic Causes:
* Rheumatoid arthritis and other inflammatory arthropathies.
* Recurrent hemarthrosis (e.g., hemophilia), where iron deposition degrades cartilage.
* Alkaptonuria (ochronosis).
* Sepsis and subsequent intraarticular adhesions.
Iatrogenic and Degenerative Causes:
* Repeated intraarticular corticosteroid injections, which can inhibit chondrocyte metabolism.
* Prolonged joint immobilization, leading to cartilage starvation (as cartilage relies on cyclical loading for synovial fluid imbibition).
* Primary osteoarthritis.
CLINICAL FINDINGS AND DIAGNOSIS
The signs and symptoms of chondromalacia of the patella are notoriously nonspecific; there is no single pathognomonic symptom.
Patients typically present with a dull, aching discomfort that is well-localized to the anterior aspect of the knee. This pain is classically exacerbated by prolonged sitting with the knees flexed—a phenomenon known as the "movie sign" or "theater sign."
Mechanical symptoms are common. Patients frequently describe a catching, grinding, or "giving way" sensation during dynamic activities. Both pain and the sensation of instability are markedly amplified when descending stairs or walking down an incline, as these activities exponentially increase the compressive forces across the patellofemoral joint (often reaching up to 7 times body weight).
Physical examination may reveal:
* Varying degrees of patellofemoral crepitation during active range of motion.
* Mild to moderate joint effusion, dependent on the severity of the secondary reactive synovitis.
* Pain with patellar compression (Clarke's test), though this is highly sensitive and poorly specific.
* Lateral tracking or a positive J-sign during terminal extension.
Surgical Warning: Arthroscopic examination of young adults with anterior knee pain confirms chondromalacia patellae in only about 45% of cases, with synovial plicae accounting for another 45%. Crucially, studies show no direct association between the severity of chondromalacia seen at arthroscopy and the clinical severity of anterior knee pain. Therefore, the mere presence of anterior knee pain syndrome should not be used as an isolated indication for diagnostic knee arthroscopy.
MANAGEMENT STRATEGIES
Treatment must be directed at the underlying cause of the articular surface changes (e.g., malalignment, instability) rather than merely addressing the result (the fibrillated cartilage).
Non-Operative Management
The cornerstone of initial treatment is non-operative. This includes:
* Pharmacotherapy: Nonsteroidal anti-inflammatory drugs (NSAIDs) to manage secondary synovitis.
* Physical Therapy: Aggressive quadriceps strengthening (specifically targeting the VMO), hamstring and iliotibial band stretching, and core stabilization.
* Activity Modification: Patients must avoid deep lunges, squats, and leg presses beyond 90 degrees of flexion. Training on uneven terrain should be strictly prohibited until symptoms resolve.
* Orthotics and Bracing: Patellar taping (McConnell taping) or dynamic patellar stabilizing braces may provide symptomatic relief by centralizing the patella within the trochlear groove.
Operative Management
Surgery is indicated only for chronic, recalcitrant patellofemoral pain after all attempts at non-operative management have been exhausted (typically a minimum of 6 months). Operative treatment is conceptually divided into two phases:
1. Treatment directed at malalignment and extensor mechanism abnormalities.
2. Treatment of the diseased cartilage itself.
Addressing Malalignment
Malalignment is the most common driver of excessive lateral pressure and subsequent chondromalacia.
* Lateral Retinacular Release: Recommended for excessive lateral pressure syndromes. It releases the strong tethering lateral structures. However, it is only effective if the preoperative diagnosis is accurate. Incongruity, lateral tilting, and lateral orientation must be confirmed on axial radiographs. Lateral release in patients with normal axial radiographs is highly unpredictable and generally contraindicated.
* Distal Realignment: For patients with patella alta or an excessive Q-angle, an open procedure that reestablishes a normal relationship between the patellar tendon and the patella is required.
Anterior elevation of the tibial tuberosity (the Maquet procedure) mechanically decompresses the patellofemoral joint by increasing the lever arm of the extensor mechanism, thereby reducing joint reactive forces.
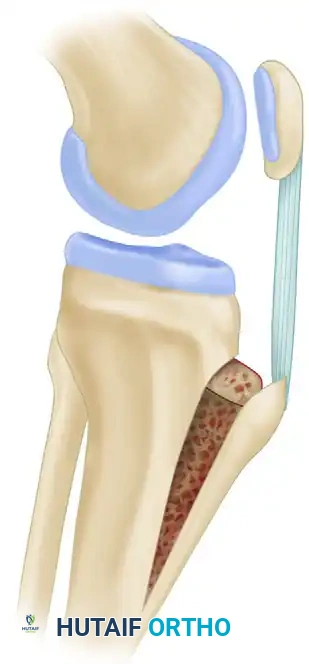
Alternatively, anteromedialization of the tibial tubercle (Fulkerson osteotomy) can simultaneously decompress the joint and correct lateral maltracking.
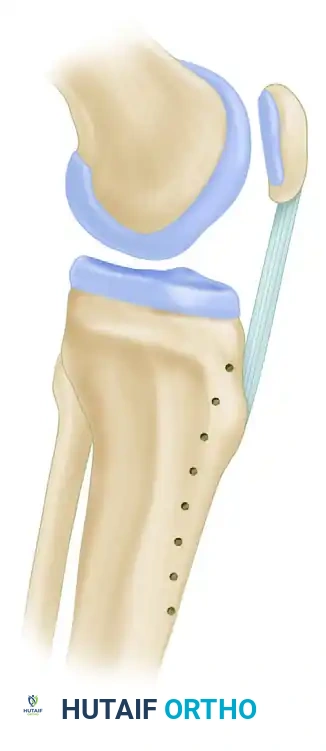
Addressing the Diseased Cartilage
Cartilage is an avascular tissue with severely limited intrinsic healing capacity. Any surgical intervention directed at the articular surface must account for this. Defects that breach the subchondral bone heal with fibrocartilage (predominantly Type I collagen), which is mechanically inferior to native hyaline cartilage (Type II collagen) and prone to eventual deterioration.
Procedures include:
1. Arthroscopic Patellar Shaving (Chondroplasty): Historically overutilized. Its primary benefit is the removal of fibrillated cartilage flaps, which decreases the shedding of breakdown products into the joint, thereby reducing painful chemical synovitis. It does not regenerate cartilage.
2. Local Excision and Subchondral Drilling/Microfracture: The area of chondromalacia is sharply debrided to stable vertical margins. The exposed subchondral bone is then perforated with multiple drill holes or an awl to release marrow elements, stimulating a fibrocartilaginous healing response.
3. Facetectomy: Excision of a severely diseased patellar facet (usually the lateral facet).
4. Patellofemoral Arthroplasty: Reserved for severe, unicompartmental patellofemoral arthritis in older patients.
SURGICAL TECHNIQUE: SUBPERIOSTEAL RELEASE OF THE LATERAL QUADRICEPS MECHANISM (OGATA)
When lateral patellar compression syndrome is identified, and conservative measures fail, a lateral release may be indicated. The Ogata technique of subperiosteal release of the lateral quadriceps mechanism offers a precise, tissue-sparing approach to decompress the lateral facet while maintaining the structural integrity of the vastus lateralis expansion.
Indications
- Documented excessive lateral pressure syndrome with lateral patellar tilt on axial radiographs.
- Focal chondromalacia of the lateral patellar facet.
- Failure of a minimum of 6 months of targeted physical therapy.
- Presence of a painful, unstable lateral patellar fragment or traction osteophyte.
Patient Positioning and Preparation
- The patient is placed supine on the operating table.
- A well-padded high thigh tourniquet is applied.
- The surgical limb is prepped and draped in a standard sterile fashion, allowing for full, unencumbered range of motion of the knee.
Step-by-Step Surgical Approach
Step 1: Arthroscopic Evaluation
* Begin with a comprehensive diagnostic arthroscopy.
* Evaluate the patellofemoral tracking dynamically. Assess the severity and exact location of the chondromalacia.
* Perform an arthroscopic chondroplasty (shaving) of unstable fibrillated cartilage flaps if Grade II or III changes are present. Flush the joint copiously to remove enzymatic debris.
Step 2: Surgical Incision
* Identify the distal portion of the vastus lateralis tendon and the lateral border of the patella.
* Make an oblique skin incision directly over the distal portion of the vastus lateralis tendon.
* Extend the incision just distal to the midportion of the separated or painful area of the lateral patella.
Step 3: Tendon Splitting and Subperiosteal Detachment
* Identify the vastus lateralis tendon, which in chronic cases is usually pathologically thickened and fibrotic.
* Carefully split the tendon longitudinally along the direction of its middle fibers.
* Identify the insertion of the tendon onto the painful lateral patellar fragment.
* Using a periosteal elevator or a sharp scalpel, detach this insertion subperiosteally.
* Critical Step: You must meticulously preserve the continuity of the superficial tendinous expansion that passes over the patella to the main extensor mass. This prevents catastrophic lateral tracking or iatrogenic medial subluxation postoperatively.
Step 4: Denuding the Fragment
* Dissect the tendon-periosteum complex—which consists of the tendinous expansion and the periosteal tissues—away from the bone.
* Sharply and completely denude the lateral patellar fragment.
Step 5: Dynamic Assessment of Fragment Stability
* Once the smaller portion of the lateral patella (or traction spur) is exposed, apply a dynamic bending stress to the fragment.
* Grasp the fragment firmly between the thumb and index finger and attempt to mobilize it.
* Observe the area of separation. In general, if the patellar fragment is mechanically unstable, a groove at the area of separation will visibly widen under this bending stress.
Step 6: Fragment Management (The Ogata Criteria)
* Grossly Mobile: If the fragment demonstrates gross mobility and widening at the groove, Ogata recommends complete surgical excision of the fragment, as it acts as a mechanical irritant.
* Stable: If the fragment shows little to no mobility with bending stress, it should be left in situ. The subperiosteal release itself is sufficient to decompress the lateral facet.
Step 7: Closure
* Thoroughly irrigate the wound to remove any bone debris.
* Release the tourniquet and achieve meticulous hemostasis to prevent postoperative hemarthrosis, which can severely inhibit rehabilitation.
* Repair the longitudinal splitting incision in the vastus lateralis tendon using #0 absorbable sutures (e.g., Vicryl). Ensure the repair is tension-free.
* Close the subcutaneous tissue and skin in a standard layered fashion.
Postoperative Care and Rehabilitation
The postoperative management protocol remains identical regardless of whether the lateral patellar fragment was excised or left in situ.
- Weight Bearing: The patient is allowed immediate full weight-bearing as tolerated (WBAT) with the knee locked in extension using a drop-lock brace or knee immobilizer for the first few days to protect against sudden buckling.
- Range of Motion: Immediate progression of active and active-assisted range-of-motion (ROM) exercises is initiated. Early motion is critical to prevent capsular adhesions and to provide nutritional imbibition to the articular cartilage.
- Strengthening: Isometric quadriceps sets and straight-leg raises are begun in the recovery room. Progression to closed-kinetic-chain exercises is permitted as pain and swelling subside.
- Return to Play: Sports activities and higher-impact loading are gradually reintroduced between 2 to 4 weeks postoperatively, strictly contingent upon the patient remaining asymptomatic and demonstrating symmetrical quadriceps control.
