Introduction & Epidemiology
Central Cord Syndrome (CCS) represents the most common incomplete spinal cord injury pattern, characterized by a disproportionately greater motor weakness in the upper extremities compared to the lower extremities, often accompanied by varying degrees of sensory loss and bladder dysfunction. This unique clinical presentation is a direct consequence of the somatotopic organization of the motor and sensory tracts within the cervical spinal cord, where upper extremity fibers are located more centrally.
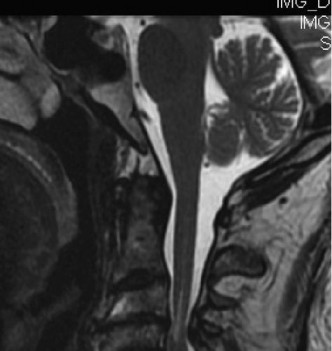
Typically, CCS occurs in older adults with pre-existing cervical spondylosis and spinal canal stenosis who sustain a hyperextension injury, often a fall causing a chin strike or direct head impact. Less commonly, it can result from direct trauma with fracture-dislocation. The mechanism involves a "pincer" effect where the spinal cord is compressed anteriorly by osteophytes or a bulging disc and posteriorly by infolding of the ligamentum flavum. This compression, combined with associated vascular compromise (ischemia to the central gray matter), leads to the characteristic neurological deficit.
Epidemiologically, CCS accounts for approximately 9% to 15% of all traumatic spinal cord injuries. The incidence is higher in males and increases with age due to the prevalence of degenerative cervical spine changes. Prognosis for CCS is generally favorable compared to other incomplete cord syndromes, with recovery often following a pattern: lower extremities first, followed by proximal upper extremities, and finally, distal upper extremity function. However, fine motor control of the hands can remain significantly impaired, posing long-term challenges for patients. The initial severity, age, and MRI findings (e.g., presence and extent of intramedullary hemorrhage/edema) are key prognostic indicators.
Surgical Anatomy & Biomechanics
Understanding the intricate anatomy and biomechanics of the cervical spine is paramount in appreciating the pathophysiology and guiding the management of CCS.
Cervical Spine Anatomy
The cervical spine comprises seven vertebrae (C1-C7), providing support and mobility to the head. Key anatomical structures include:
*
Vertebral Bodies:
Anterior load-bearing structures.
*
Intervertebral Discs:
Act as shock absorbers and provide flexibility.
*
Neural Arch:
Formed by pedicles and laminae, enclosing the spinal canal.
*
Facet Joints:
Paired synovial joints providing stability and guiding motion.
*
Ligaments:
*
Anterior Longitudinal Ligament (ALL):
Reinforces the anterior vertebral bodies.
*
Posterior Longitudinal Ligament (PLL):
Reinforces the posterior vertebral bodies, extending into the spinal canal.
*
Ligamentum Flavum:
Elastic ligament connecting adjacent laminae, thickens with age, and can buckle into the canal during hyperextension.
*
Interspinous and Supraspinous Ligaments:
Connect spinous processes.
*
Nuchal Ligament:
Extension of the supraspinous ligament in the cervical region.
Spinal Cord Anatomy
The cervical spinal cord is critical. Its internal architecture dictates the clinical presentation of CCS.
*
Gray Matter:
Butterfly-shaped, located centrally. Contains neuronal cell bodies, including motor neurons in the anterior horn and sensory neurons in the posterior horn. The central location of the gray matter makes it particularly vulnerable to compressive and ischemic forces.
*
White Matter Tracts:
Surrounding the gray matter, containing ascending sensory and descending motor pathways.
*
Lateral Corticospinal Tracts:
Primarily responsible for voluntary motor control. Fibers for the upper extremities are more medially located within these tracts, while lower extremity fibers are more peripheral. This somatotopic organization is the anatomical basis for the disproportionate weakness seen in CCS.
*
Anterior Corticospinal Tracts:
Contribute to axial and proximal limb muscle control.
*
Spinothalamic Tracts:
Carry pain and temperature sensation. Located anterolaterally.
*
Dorsal Columns:
Medially located, carrying proprioception, vibration, and fine touch.
Vascular Supply
The spinal cord's vascular supply is crucial.
*
Anterior Spinal Artery:
Formed by branches of the vertebral arteries, supplies the anterior two-thirds of the spinal cord (ventral gray matter, anterior and lateral white matter columns).
*
Posterior Spinal Arteries:
Paired arteries supplying the posterior one-third (dorsal columns, posterior horns).
*
Radicular Arteries:
Segmental branches from the vertebral, subclavian, intercostal, and lumbar arteries, reinforcing the anterior and posterior spinal arteries.
Ischemia in the central gray matter and surrounding white matter, particularly affecting the anterior spinal artery territory, is a major component of CCS pathophysiology.
Biomechanics of Injury
The classic mechanism for CCS is a hyperextension injury in a patient with pre-existing cervical spondylosis and spinal stenosis.
*
Spondylosis:
Degenerative changes (osteophytes, disc bulging, ligamentum flavum hypertrophy) narrow the spinal canal.
*
Hyperextension:
During forced hyperextension, two phenomena occur:
1.
Anterior Compression:
The spinal cord is compressed by anterior osteophytes and disc material.
2.
Posterior Compression:
The ligamentum flavum buckles and infolds into the posterior aspect of the spinal canal, further compressing the cord.
* This "pincer" effect, combined with the stretching of the spinal cord over the anterior vertebral bodies, leads to direct mechanical trauma and ischemic injury to the centrally located gray matter and adjacent white matter tracts. The upper extremity motor fibers, being most central, bear the brunt of this injury, resulting in their characteristic weakness.
Indications & Contraindications
The management of Central Cord Syndrome is often a nuanced decision, balancing conservative measures with surgical intervention.
Non-Operative Indications
Initial management for the majority of CCS cases is conservative. This approach is favored when:
*
Neurological Status:
The patient demonstrates spontaneous, early neurological improvement. Many patients with CCS show significant recovery, particularly in the lower extremities, within the first 24-72 hours.
*
Spinal Stability:
There is no evidence of fracture, dislocation, or ligamentous instability on imaging. Most classic CCS cases present with stable spines.
*
Imaging:
MRI shows only cord edema without significant structural compression by a herniated disc or fracture fragment, or without cord compression that requires urgent decompression.
*
Patient Comorbidities:
Significant medical comorbidities may preclude the patient from tolerating general anesthesia and surgery.
Non-operative management typically involves:
*
Spinal Immobilization:
A rigid cervical collar is often used initially to limit neck motion and prevent further injury, typically for 6-12 weeks.
*
Medical Management:
*
Blood Pressure Control:
Maintaining adequate mean arterial pressure (MAP) to optimize spinal cord perfusion, typically MAP > 85 mmHg.
*
Oxygenation:
Ensuring adequate oxygen delivery.
*
Fluid Management:
Maintaining euvolemia.
*
Steroids:
High-dose methylprednisolone, as per NASCIS II and III protocols, is controversial and not routinely recommended by many guidelines due to unclear benefit and increased risk of complications (e.g., GI bleeding, infection). Current AANS/CNS guidelines generally advise against routine use.
*
Rehabilitation:
Early initiation of physical and occupational therapy is crucial to maximize functional recovery.
*
Close Neurological Monitoring:
Serial neurological exams are essential to detect any progression or deterioration.
Operative Indications
Surgical intervention is generally considered for a subset of CCS patients or when conservative measures fail. The timing of surgery for CCS without instability is often debated, with a trend towards early decompression for improving neurological outcomes, though some still advocate for delayed surgery after initial stabilization and observation.
*
Progressive Neurological Deficit:
Any documented worsening of motor or sensory function after the initial injury.
*
Spinal Instability:
Presence of an unstable fracture, dislocation, or significant ligamentous injury that directly compromises spinal cord integrity or risks further neurological injury.
*
Persistent Significant Spinal Cord Compression:
Imaging (MRI) demonstrating persistent severe anterior or posterior spinal cord compression by a disc herniation, osteophyte, or infolded ligamentum flavum, without significant neurological improvement over a period of conservative management (typically 6-12 weeks for chronic myelopathy, but potentially earlier if no signs of recovery are observed).
*
Early Decompression for Specific Subgroups:
Increasing evidence suggests that early surgical decompression (within 24-72 hours) for patients with traumatic CCS and persistent spinal cord compression may improve neurological recovery, particularly for those with severe deficits or specific MRI findings (e.g., ongoing compression, limited intramedullary hemorrhage). This remains an area of ongoing research and clinical discussion.
Contraindications
- Rapid Neurological Recovery: Patients who exhibit significant and rapid neurological improvement are typically managed non-operatively, as the risks of surgery may outweigh potential benefits.
- Severe Comorbidities: Patients with severe medical conditions that make them poor surgical candidates for general anesthesia or prolonged procedures.
- Diffuse Intramedullary Cord Edema/Hemorrhage without Structural Compression: If imaging reveals extensive cord edema or hemorrhage without a clearly identifiable compressive lesion amenable to surgical decompression, the role of surgery is diminished.
Table: Operative vs. Non-Operative Indications for Central Cord Syndrome
| Feature / Indication | Non-Operative Management | Operative Management |
|---|---|---|
| Neurological Status | Improving or stable neurological deficit | Progressive neurological deficit |
| Minor initial deficit or rapid recovery observed | Persistent severe deficit with minimal or no recovery | |
| Spinal Stability | Stable cervical spine, no fracture/dislocation | Unstable fracture, dislocation, or significant ligamentous injury |
| Imaging (MRI) | Cord edema only, no significant persistent compression | Significant persistent spinal cord compression (disc, osteophyte, buckle) |
| Absence of critical spinal canal stenosis | Critical spinal canal stenosis predisposing to ongoing cord injury | |
| Timing | Initial phase for most stable CCS patients | Early (within 24-72h) for select cases with persistent compression and severe deficit |
| Delayed (weeks-months) for chronic compression without recovery | ||
| Patient Factors | Significant medical comorbidities precluding surgery | Medically optimized and able to tolerate surgery |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential to optimize outcomes and minimize risks associated with surgical intervention for Central Cord Syndrome.
Pre-Operative Planning
-
Diagnostic Imaging Review:
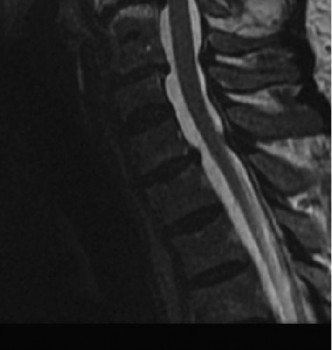
- MRI: The gold standard to assess spinal cord pathology (edema, hemorrhage, compression, contusion) and evaluate soft tissue compressive lesions (disc herniation, ligamentum flavum hypertrophy). Crucially, it delineates the extent and location of cord compression.
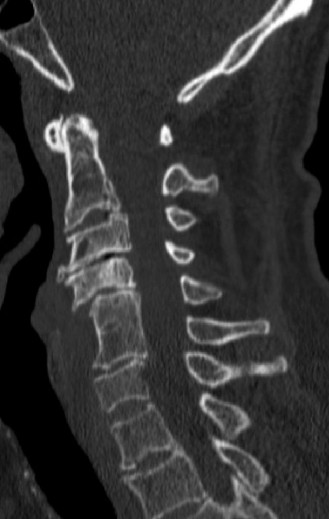
- CT Scan: Provides detailed bony anatomy, identifies fractures, osteophytes, and can assess spinal canal dimensions. CT angiogram may be considered if vertebral artery injury is suspected, particularly in fracture-dislocations.
- Dynamic X-rays: Flexion/extension views may be indicated if instability is suspected, though caution is advised in acute trauma.
- Neurological Assessment: A detailed baseline neurological examination must be documented, including motor strength (MRC scale), sensory examination (pinprick, light touch, proprioception), and reflex assessment. This serves as a critical benchmark for post-operative evaluation.
- Medical Optimization: A comprehensive pre-operative medical workup is performed to assess and optimize the patient's cardiorespiratory status, coagulation profile, nutritional status, and glycemic control. Consultation with internal medicine, cardiology, or pulmonology may be required.
-
Surgical Approach Selection:
The choice between an anterior, posterior, or combined approach depends on the primary source and location of spinal cord compression, the number of levels involved, sagittal alignment, and surgeon preference.
- Anterior Approach (ACDF/ACCF): Indicated for primary anterior compression (disc herniation, osteophytes), often effective for 1-3 levels.
- Posterior Approach (Laminectomy/Laminoplasty): Indicated for multi-level posterior compression (ligamentum flavum hypertrophy) or foraminotomy for radiculopathy. Laminoplasty is favored over laminectomy for multi-level decompression to reduce the risk of post-laminectomy kyphosis.
- Combined Approach: Rarely indicated for CCS unless complex multi-directional compression or severe kyphosis requiring realignment.
- Informed Consent: Detailed discussion with the patient or legal proxy regarding the nature of the procedure, expected outcomes, and potential complications (e.g., neurological worsening, infection, bleeding, pseudarthrosis, dysphagia, C5 palsy, hardware failure).
- Blood Management: Type and cross-match blood products are typically ordered.
Patient Positioning
Proper patient positioning is critical to ensure surgical access, spinal alignment, patient safety, and optimal neuromonitoring signal acquisition.
-
General Considerations:
- Anesthesia: General endotracheal anesthesia is standard.
- Neuromonitoring: Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs) are highly recommended to monitor spinal cord function throughout the procedure, especially during decompression and instrumentation.
- Prophylactic Antibiotics: Administered pre-incision.
- DVT Prophylaxis: Sequential compression devices (SCDs) are applied.
-
Anterior Approach (Supine Positioning):
- The patient is placed supine on the operating table.
- A roll or bolster is often placed transversely between the scapulae to facilitate neck extension and allow shoulders to drop posteriorly, exposing the neck.
- The head is secured in a neutral or slightly extended position, typically in a donut headrest or gentle traction if deemed safe and necessary. Avoid excessive extension, particularly in patients with pre-existing stenosis, to prevent cord compromise.
- Arms are tucked at the sides.
- Prepping and draping: From the chin to the nipples, and ear-to-ear, including the iliac crest if autograft harvest is planned.
-
Posterior Approach (Prone Positioning):
- The patient is carefully log-rolled from supine to prone position after induction of anesthesia, ensuring the cervical spine remains in a neutral, aligned position.
- Head Fixation: A Mayfield® skull clamp or Gardner-Wells tongs are applied for rigid head fixation, allowing precise control of cervical alignment and preventing inadvertent movement. The head is maintained in a neutral position or slight flexion depending on the pathology and desired lordosis. Excessive flexion can cause cord stretch, while excessive extension can exacerbate impingement in a stenotic canal.
- Chest and Pelvis Supports: Padded chest rolls and pelvic rolls are used to offload the abdomen, minimizing intra-abdominal pressure and consequently reducing epidural venous bleeding. This also allows the shoulders to drop, aiding visualization.
- Arm Positioning: Arms are usually abducted and externally rotated ("superman" position) with elbows flexed, supported on arm boards, ensuring no brachial plexus compression.
- Prepping and Draping: From the occiput down to the mid-thoracic region, and laterally to the deltoids.
- Careful padding of all pressure points is crucial to prevent peripheral nerve palsies or skin breakdown.
Detailed Surgical Approach / Technique
The choice of surgical approach for Central Cord Syndrome is dictated by the primary location of compression (anterior vs. posterior), the number of levels involved, and the sagittal alignment of the cervical spine. Decompression of the neural elements and stabilization of the spinal column are the overarching goals.
Anterior Cervical Decompression and Fusion (ACDF) / Anterior Cervical Corpectomy and Fusion (ACCF)
Indications: Primarily for anterior spinal cord compression caused by disc herniation, osteophytes, or ossification of the posterior longitudinal ligament (OPLL), typically involving 1-3 levels.
Approach (Right-sided common, left also used):
1.
Incision:
A transverse skin incision is made along a skin crease, typically at the C5-C6 level for mid-cervical pathology, or a longitudinal incision along the anterior border of the sternocleidomastoid (SCM) for multi-level exposure.
2.
Dissection:
*
Platysma:
Incised or split longitudinally.
*
Internervous Plane:
The plane between the sternocleidomastoid muscle (innervated by CN XI) and the strap muscles (sternohyoid, sternothyroid, omohyoid – innervated by ansa cervicalis) is entered. The SCM and carotid sheath contents (carotid artery, internal jugular vein, vagus nerve) are retracted laterally. The trachea and esophagus are retracted medially. The recurrent laryngeal nerve should be identified and protected, especially on the left side where its course is more variable and susceptible to injury.
*
Prevertebral Fascia:
Incised longitudinally.
*
Longus Colli Muscles:
Subperiosteal dissection of the longus colli muscles laterally at the target level(s) allows exposure of the anterior vertebral bodies and disc spaces.
3.
Localization:
Intraoperative fluoroscopy or X-ray confirms the correct spinal level(s).
4.
Discectomy (ACDF):
* The anterior annulus fibrosus is incised, and a partial or complete discectomy is performed.
* Using pituitary rongeurs, curettes, and high-speed burrs, the posterior annulus and posterior osteophytes are meticulously removed.
* Under microscopic visualization or loupes, the posterior longitudinal ligament (PLL) is carefully resected to achieve direct decompression of the dura and spinal cord. Protruding osteophytes are burred down.
* Paddles or nerve root retractors are used to protect the nerve roots and cord during decompression.
5.
Corpectomy (ACCF):
For multi-level anterior compression involving vertebral body pathology or extensive OPLL.
* After discectomy at adjacent levels, the involved vertebral body is removed using a high-speed burr and curettes.
* Careful decompression is performed, typically leaving a thin shell of posterior cortex initially, then gently removing it to expose the dura and spinal cord.
6.
Fusion:
*
ACDF:
An appropriately sized interbody cage (PEEK, titanium) packed with autograft (e.g., iliac crest) or allograft is inserted into the disc space.
*
ACCF:
A reconstructive cage or strut graft (titanium, fibular allograft, autograft) is placed to span the corpectomy defect.
* An anterior cervical plate is then applied across the superior and inferior vertebral bodies (or reconstructive cage) and secured with screws to provide immediate stability and facilitate fusion.
7.
Closure:
Layered closure after hemostasis, including platysma, deep fascia, and skin. A drain may be placed.
Posterior Cervical Decompression (Laminoplasty / Laminectomy)
Indications: Primarily for multi-level cervical stenosis causing posterior cord compression (e.g., by infolding ligamentum flavum, hypertrophy of laminae) or when an anterior approach is deemed insufficient or too risky (e.g., rigid kyphosis).
Approach:
1.
Incision:
A midline longitudinal incision is made from the occiput to the appropriate caudal level, typically C2 or C3 to C7.
2.
Dissection:
*
Ligamentum Nuchae:
Incised in the midline.
*
Subperiosteal Dissection:
Paraspinal muscles (trapezius, splenius capitis, semispinalis cervicis) are carefully dissected subperiosteally off the spinous processes and laminae using electrocautery and Cobb elevators. Great care is taken to preserve the muscle attachments at C2 and C7 to minimize axial neck pain and maintain post-operative sagittal balance. The muscle groups are retracted laterally.
*
Localization:
Intraoperative fluoroscopy confirms levels.
3.
Laminoplasty (Preferred for multi-level stenosis to preserve stability and reduce kyphosis risk):
*
Open-door Laminoplasty:
* A hinge side is created on one side (e.g., left) by thinning the lamina with a high-speed burr, preserving the outer cortex.
* The contralateral side (e.g., right) undergoes complete osteotomy at the lamina-facet junction.
* The laminae are then "opened" or swung laterally like a door, expanding the spinal canal diameter.
* The expanded laminae are secured in the open position using mini-plates and screws, or sutures to the lateral mass.
*
French-door Laminoplasty:
* A midline sagittal osteotomy of the spinous processes and laminae is performed.
* Hinges are created laterally on both sides (lamina-facet junction).
* The laminae are spread apart, and the space is maintained with bone grafts or mini-plates.
* The ligamentum flavum is typically resected during this process.
4.
Laminectomy (Reserved for specific cases due to potential for post-laminectomy kyphosis):
* Complete removal of the spinous processes and laminae at the affected levels using rongeurs and a high-speed burr.
* The ligamentum flavum is resected to decompress the spinal cord.
* This provides wide decompression but may destabilize the posterior column, potentially leading to kyphosis, especially if more than 3 levels are involved or if there's pre-existing kyphosis.
5.
Foraminotomy:
If radiculopathy is present, a foraminotomy (enlargement of the neuroforamen) can be performed by resecting the medial portion of the facet joint.
6.
Fusion (if necessary):
If instability is present or anticipated (e.g., extensive laminectomy with pre-existing kyphosis), posterior instrumentation (lateral mass screws/rods or pedicle screws/rods) and fusion with bone graft may be performed concurrently.
Key Surgical Principles:
*
Adequate Decompression:
The primary goal is to relieve all sources of spinal cord compression.
*
Preservation of Stability:
Avoid iatrogenic instability. If stability is compromised, fusion is mandatory.
*
Protection of Neural Structures:
Meticulous technique, proper retraction, and continuous neuromonitoring are crucial to prevent iatrogenic cord or nerve root injury.
*
Hemostasis:
Careful hemostasis to prevent post-operative hematoma formation, which can compress the cord.
Complications & Management
Despite meticulous surgical technique, complications can arise following surgery for Central Cord Syndrome. Vigilant post-operative monitoring and prompt intervention are critical for optimal patient outcomes.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (Approximate) | Salvage Strategies |
|---|---|---|
| Intraoperative Neurological Worsening / Spinal Cord Injury | < 1% | Immediate cessation of surgical manipulation, check neuromonitoring signals, verify alignment, remove/adjust implants, consider fluid bolus/pressors. If due to hematoma, evacuate immediately. |
| Dural Tear | 1-5% | Primary repair with suture (e.g., 6-0 Prolene) if possible. Reinforce with dural sealant (fibrin glue), muscle patch, fat graft. Lumbar drain for 3-5 days to reduce CSF pressure and promote healing. Strict bed rest. |
| C5 Palsy (Post-Posterior Decompression) | 5-20% (transient) | Often spontaneous recovery within 3-6 months. Physical therapy. EMG/NCS if persistent. Rarely requires nerve exploration or transfer. Multifactorial etiology (reperfusion injury, tethering, root manipulation). |
| Dysphagia (Anterior Approach) | 5-70% (transient), <5% (persistent) | Post-operative voice rest, soft diet, swallowing therapy. Steroids (dexamethasone) for acute edema. Evaluation by speech-language pathologist. ENT consult for vocal cord assessment (recurrent laryngeal nerve injury). Usually resolves within weeks to months. |
| Recurrent Laryngeal Nerve Palsy (Anterior Approach) | < 1-5% | Typically transient. Voice rest, speech therapy. ENT consultation for vocal cord assessment. Surgical repair rarely indicated for acute injury; may be considered for persistent deficit. |
| Epidural Hematoma | < 1% | Urgent MRI/CT for diagnosis. Prompt surgical evacuation of hematoma if causing neurological deficit (within 6-12 hours if possible to maximize recovery). Close post-op drain monitoring. |
| Surgical Site Infection (SSI) | 1-5% | Superficial: Wound care, oral antibiotics. Deep: Surgical debridement, IV antibiotics (culture-guided), wound vacuum-assisted closure (VAC) or flap coverage. Hardware removal may be required if persistent or chronic. |
| Pseudarthrosis / Non-union | 5-15% (fusion rates vary) | Conservative management for asymptomatic cases. If symptomatic (pain, instability, hardware failure), revision surgery with additional bone graft, optimized fixation, or different fusion technique (e.g., 360-degree fusion for previous anterior-only). |
| Hardware Failure | 2-10% | Removal of failed hardware. Revision surgery with stronger constructs, additional fixation points, or different plate/screw system. Addressing underlying pseudarthrosis is critical. |
| Adjacent Segment Disease (ASD) | 2-4% per patient-year | Initial conservative management (NSAIDs, physical therapy, injections). If progressive or unremitting myelopathy/radiculopathy, revision surgery involving decompression and fusion at the adjacent segment(s). |
| Post-Laminectomy Kyphosis | 5-30% (variable, higher in children, extensive laminectomy) | Anterior cervical fusion with instrumentation, posterior instrumentation and fusion, or combined approach to restore sagittal balance and stabilize the spine. |
| Cerebrospinal Fluid (CSF) Leak | 1-5% | Bed rest, head of bed elevation, wound care, +/- lumbar drain, +/- surgical revision of dural repair. |
| Peripheral Nerve Injury | < 1% | Prevention with careful positioning and padding. If evident, nerve conduction studies/EMG. Physical therapy. Neurologist consultation. |
General Management Principles
- Vigilant Post-operative Monitoring: Close observation for changes in neurological status, vital signs, drain output (for hematoma), and signs of infection.
- Pain Management: Multimodal analgesia to optimize patient comfort and facilitate early mobilization.
- Early Mobilization: As soon as deemed safe by the surgeon, to prevent complications such as DVT, pneumonia, and deconditioning.
- Neurological Assessment: Serial neurological exams are crucial to detect any new or worsening deficits.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a cornerstone of recovery for patients undergoing surgery for Central Cord Syndrome, aiming to restore strength, function, and independence. Protocols are typically phased and individualized based on the surgical procedure, neurological recovery, and patient-specific factors.
Phase 1: Acute Post-Operative (Weeks 0-6)
Goals:
Pain control, wound healing, protecting the surgical construct, preventing secondary complications, initiating early mobility.
*
Bracing:
A rigid cervical collar (e.g., Aspen, Miami J) is often used for 6-12 weeks following fusion procedures to provide external support and promote fusion. For laminoplasty without fusion, a soft collar may be used for comfort, or no collar depending on surgeon preference and stability.
*
Pain Management:
Multimodal approach including opioids, NSAIDs (if no contraindications for fusion), muscle relaxants, and neuropathic pain medications.
*
Wound Care:
Meticulous wound care to prevent infection.
*
Early Mobilization:
*
Bed Mobility:
Log-rolling for turning, encouraging independence with transfers.
*
Ambulation:
Progressive ambulation with assistance as tolerated.
*
Gentle Range of Motion (ROM):
*
Passive ROM:
For peripheral joints (shoulders, elbows, wrists, hands, hips, knees, ankles) to prevent contractures and stiffness.
*
Cervical ROM:
Strict avoidance of active or passive cervical flexion, extension, or rotation in the braced period, particularly after fusion. Gentle isometric contractions of the cervical paraspinals may be initiated cautiously in later stages for pain relief, but not for ROM.
*
Respiratory Management:
Deep breathing exercises, incentive spirometry to prevent atelectasis and pneumonia.
*
Activities of Daily Living (ADLs):
Training in modified ADLs (e.g., dressing, hygiene) with assistance, respecting cervical precautions.
*
Monitoring:
Continued neurological checks for any changes.
Phase 2: Subacute / Intermediate (Weeks 6-12)
Goals:
Gradually increase cervical ROM, initiate strengthening, improve endurance, address residual neurological deficits.
*
Brace Weaning:
If a rigid collar was used for fusion, gradual weaning typically begins around 6-8 weeks based on radiographic evidence of healing and surgeon assessment.
*
Cervical ROM:
*
Active-assisted ROM:
Gentle, pain-free active-assisted cervical flexion, extension, lateral bending, and rotation.
*
Active ROM:
Progress to full active ROM as tolerated, ensuring no pain or neurological symptoms.
*
Strengthening:
*
Cervical:
Gentle isometric neck strengthening exercises (e.g., against hand resistance). Progress to light resistance exercises for neck musculature.
*
Scapular Stabilizers:
Strengthening exercises (e.g., rows, prone Ys, Ts) to support shoulder girdle.
*
Upper Extremity:
Progressive resistive exercises for shoulders, elbows, wrists, and especially hands. Focus on grip strength, fine motor coordination, and dexterity which are often most affected in CCS.
*
Core Strengthening:
Crucial for overall spinal stability and posture.
*
Endurance:
Increase walking distance and duration.
*
Proprioception and Balance:
Exercises to improve balance and coordination.
*
Occupational Therapy:
Continued focus on fine motor skills, adaptive equipment, and strategies for performing ADLs.
Phase 3: Chronic / Advanced (Weeks 12+)
Goals:
Maximize functional recovery, return to work and recreational activities, long-term maintenance.
*
Full Cervical ROM and Strength:
Achieve full, pain-free cervical ROM and adequate strength.
*
Advanced Strengthening:
Progress to higher-level resistance exercises for all muscle groups, including sport-specific or work-specific training.
*
Neuromuscular Re-education:
Focus on advanced motor control, coordination, and proprioceptive drills.
*
Ergonomics:
Education on proper posture, lifting techniques, and workstation ergonomics to prevent recurrence or exacerbation of symptoms.
*
Return to Activity:
Gradual return to work, recreational activities, and sports as deemed appropriate by the surgeon and therapist, respecting spinal precautions. Avoid contact sports.
*
Long-Term Maintenance:
Encourage lifelong commitment to exercise, core strengthening, and good posture.
*
Pain Management:
Transition to non-pharmacological pain management strategies.
Throughout all phases, a multidisciplinary approach involving physical therapists, occupational therapists, pain management specialists, and neurologists is essential to address the complex needs of CCS patients. Regular follow-up with the orthopedic surgeon is crucial to monitor fusion progression, hardware integrity, and overall clinical status.
Summary of Key Literature / Guidelines
The management of Central Cord Syndrome has evolved significantly, with ongoing research refining best practices, particularly regarding the timing and necessity of surgical intervention.
Role of Steroids
- The NASCIS (National Acute Spinal Cord Injury Study) trials (II and III) , conducted in the 1990s, explored the use of high-dose methylprednisolone (MPSS) for acute spinal cord injury. While NASCIS II suggested a modest benefit if MPSS was initiated within 8 hours of injury, NASCIS III extended this to 24-48 hours with a longer duration.
- Current Consensus: Despite these findings, the routine use of high-dose MPSS for acute spinal cord injury, including CCS, is no longer universally recommended. Major organizations like the American Association of Neurological Surgeons (AANS) and Congress of Neurological Surgeons (CNS) in their most recent guidelines (2013, reaffirmed 2019) state that MPSS "may be considered as an option" within 8 hours, but caution about the increased risk of complications (e.g., pneumonia, GI hemorrhage, wound infection). They recommend against its use beyond 8 hours post-injury. This shift reflects a cautious approach, balancing potential minor neurological benefits against significant adverse event risks.
Timing of Surgery for Traumatic Central Cord Syndrome
-
Early vs. Delayed Decompression:
This remains a critical area of debate.
- Historically: Most stable CCS cases were managed non-operatively, with surgery considered only for progressive neurological deficit or persistent compression after a period of observation, often weeks to months (delayed decompression).
- Recent Evidence (AANS/CNS 2013/2019 Guidelines): Strong recommendation for early surgical decompression (within 24 hours) for patients with traumatic cervical spinal cord injury (including CCS) and persistent spinal cord compression. Level II evidence suggests that early surgery is associated with improved neurological outcome. This is attributed to minimizing secondary injury mechanisms such as ischemia, excitotoxicity, and inflammation.
- Subgroup Analysis: While early decompression seems beneficial, its precise application to all CCS patients is still refined. Patients with severe neurological deficits and clear evidence of persistent cord compression on MRI are often prioritized for early intervention. Those with mild deficits or rapid early recovery are still often managed conservatively.
- Controversy: The evidence for early surgery specifically for CCS is primarily derived from studies on broader acute traumatic SCI populations. Some still argue for initial conservative management in stable CCS patients, given its generally favorable prognosis and the potential for spontaneous recovery. However, the trend is towards earlier decompression when significant structural compression persists.
Surgical Approaches for Cervical Myelopathy
-
Anterior vs. Posterior:
The choice of surgical approach for chronic cervical myelopathy (the underlying pathology in many CCS cases) depends on the specific anatomical compression and sagittal alignment.
- Anterior Decompression (ACDF/ACCF): Effective for anterior compression (discs, osteophytes) and can restore lordosis. Typically used for 1-3 levels.
- Posterior Decompression (Laminoplasty/Laminectomy): Indicated for multi-level posterior compression or when kyphosis precludes an anterior approach. Laminoplasty is generally favored over laminectomy for multi-level decompression due to a lower risk of post-operative kyphosis and better preservation of cervical motion. Several studies have shown comparable neurological outcomes between anterior and posterior approaches when appropriately applied, but laminoplasty has demonstrated lower rates of adjacent segment disease compared to fusion.
- Sagittal Balance: Maintaining or restoring cervical lordosis is paramount in any surgical intervention, as kyphosis can exacerbate cord tension and lead to poorer outcomes and increased risk of adjacent segment disease.
Prognostic Factors for Recovery
- Initial Severity: Greater initial neurological deficit typically correlates with less complete recovery.
- Age: Younger patients generally have better recovery potential.
- MRI Findings: The presence and extent of intramedullary hemorrhage (suggesting more severe cord damage) on MRI is a negative prognostic indicator, while pure edema is associated with better outcomes. The degree of spinal cord compression is also crucial.
- Time to Decompression: As noted, earlier decompression in selected cases is increasingly linked to better neurological outcomes.
Key Literature Sources
- AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves: Publishes evidence-based guidelines on acute spinal cord injury and cervical myelopathy.
- AO Spine Foundation: Provides educational resources and guidelines for spine trauma and degenerative conditions.
- North American Spine Society (NASS): Offers clinical guidelines and position statements on various spine conditions.
In summary, while conservative management remains the initial approach for many stable CCS patients with early neurological improvement, the paradigm for persistent structural compression, especially in severe deficits, is shifting towards early surgical decompression. The choice of surgical technique is highly individualized, based on precise anatomical considerations and aiming for effective decompression and stable spinal reconstruction.

