Introduction & Epidemiology
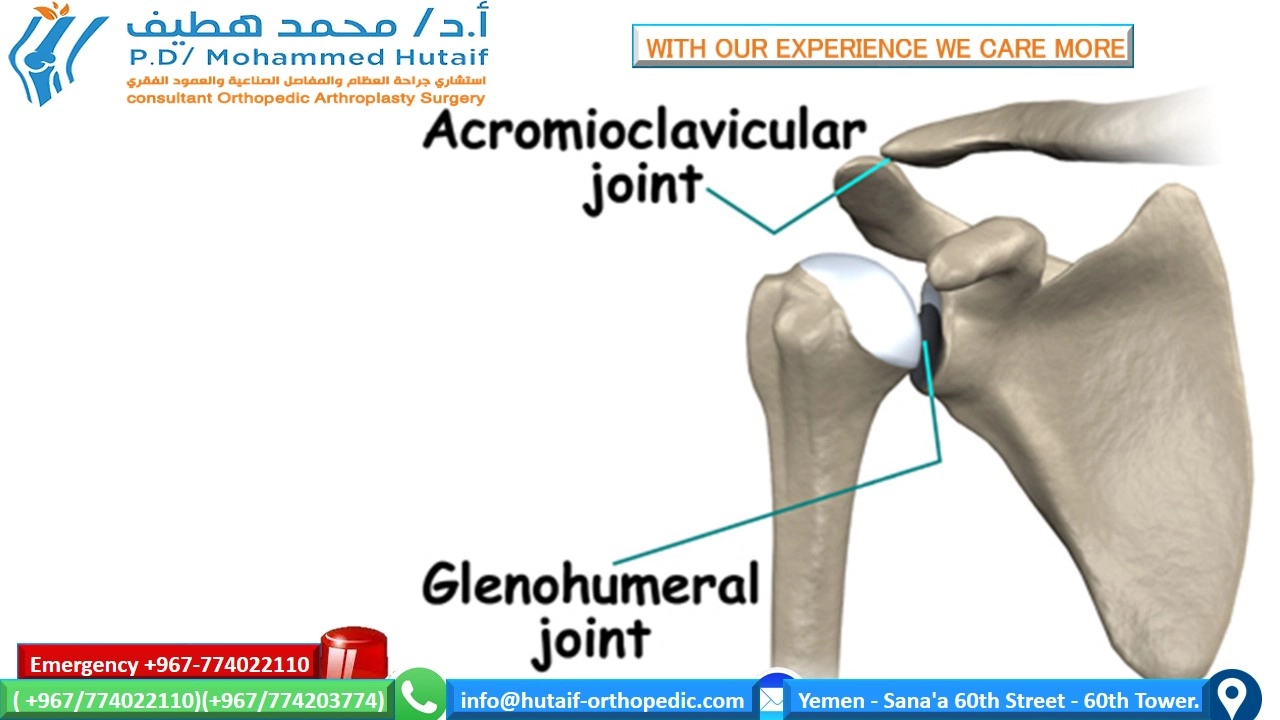
Shoulder osteoarthritis (OA) is a progressive degenerative joint disease characterized by the breakdown of articular cartilage, subchondral bone remodeling, and synovial inflammation. While the term "shoulder osteoarthritis" can broadly encompass degenerative changes in both the acromioclavicular (AC) joint and the glenohumeral (GH) joint, clinical practice often uses it as a shorthand for glenohumeral osteoarthritis (GHOA) due to its greater clinical significance and impact on global shoulder function. This review will primarily focus on GHOA, with relevant discussions of concomitant AC joint pathology where appropriate.
GHOA represents a significant burden on healthcare systems and patient quality of life. It is distinct from other forms of arthritis in its primary mechanical etiology, although inflammatory components contribute to its progression. The disease manifests as a gradual loss of the protective articular cartilage, leading to increased friction, bone-on-bone articulation, and subsequent pain, stiffness, and functional limitation.

The image above illustrates the fundamental pathology of osteoarthritis: the erosion of articular cartilage, leading to a diminished joint space and subchondral bone changes, which are characteristic findings in advanced disease.
Epidemiology:
GHOA affects millions worldwide, with its prevalence increasing with age. Radiographic evidence of GHOA is found in approximately 20-30% of individuals over 60 years of age, though only a subset of these are symptomatic. Symptomatic GHOA affects around 3% of the population over 60.
Classification and Etiology:
GHOA can be broadly classified into primary (idiopathic) and secondary forms:
*
Primary GHOA:
The most common form, with no identifiable predisposing factor. It is thought to be multifactorial, involving genetic predisposition, age-related chondrocyte senescence, and cumulative microtrauma.
*
Secondary GHOA:
Occurs due to an identifiable cause, including:
*
Post-traumatic Arthritis:
Following fractures of the humeral head or glenoid, dislocations, or other significant joint trauma. Altered joint congruity, cartilage damage, and instability contribute.
*
Rotator Cuff Tear Arthropathy (CTA):
A distinct and severe form of GHOA characterized by massive, irreparable rotator cuff tears leading to superior migration of the humeral head, erosion of the glenoid and acromion, and subsequent arthritic changes. The loss of the superior stabilizing effect of the rotator cuff leads to abnormal biomechanics and accelerated cartilage wear.
*
Inflammatory Arthropathy:
Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and other systemic inflammatory conditions can lead to erosive changes in the glenohumeral joint.
*
Osteonecrosis (Avascular Necrosis - AVN):
Impaired blood supply to the humeral head leads to collapse of the subchondral bone, causing secondary GHOA. Common etiologies include corticosteroid use, alcohol abuse, sickle cell disease, and trauma.
*
Dysplasia:
Congenital abnormalities in glenoid or humeral head morphology can predispose to early degenerative changes.
*
Infection:
Septic arthritis can rapidly destroy articular cartilage, leading to post-infectious GHOA.
*
Metabolic Disorders:
Hemochromatosis, ochronosis, and gout can also contribute.
Pathophysiology:
The hallmark of GHOA is the progressive degradation of articular cartilage. Chondrocytes, the sole cells within cartilage, lose their ability to maintain the extracellular matrix (ECM), composed primarily of collagen type II and proteoglycans (aggrecan). This imbalance between synthesis and degradation, often driven by matrix metalloproteinases (MMPs) and aggrecanases (ADAMTS enzymes), leads to:
*
Cartilage softening and fibrillation:
Early changes characterized by disruption of the superficial collagen network.
*
Fissuring and erosion:
Deepening defects extending into the subchondral bone.
*
Subchondral bone changes:
Sclerosis, cyst formation, and osteophyte development at the joint margins.
*
Synovitis:
Secondary inflammation of the synovial membrane, contributing to pain and further cartilage degradation through cytokine release.
*
Capsular thickening and contracture:
Leading to loss of range of motion.
Clinical Presentation:
Patients typically present with:
*
Pain:
insidious onset, deep, aching, often localized to the posterior shoulder or lateral deltoid insertion. Aggravated by activity, relieved by rest, but can become nocturnal in advanced stages.
*
Stiffness:
Especially noticeable in the morning or after periods of inactivity, often described as a "grinding" sensation.
*
Crepitus:
Audible or palpable grinding, clicking, or popping during shoulder movement.
*
Loss of Motion:
Progressive decrease in active and passive range of motion, particularly external rotation and elevation.
*
Functional Limitations:
Difficulty with activities of daily living (ADLs) such as reaching overhead, dressing, personal hygiene, and lifting.
Diagnosis:
*
History:
Detailed account of pain characteristics, functional limitations, prior trauma, medical comorbidities, and previous treatments.
*
Physical Examination:
* Inspection: Atrophy (deltoid, rotator cuff), swelling, asymmetry.
* Palpation: Tenderness over joint line, AC joint, rotator cuff insertions.
* Range of Motion: Assessment of active and passive forward flexion, abduction, external rotation, and internal rotation. Often, a capsular pattern (loss of external rotation > abduction > internal rotation) is observed.
* Strength Testing: To assess rotator cuff and deltoid integrity (especially important for rTSA considerations).
* Special Tests: Impingement signs (Neer, Hawkins), instability tests (apprehension, relocation), AC joint tests (cross-body adduction) to rule out concomitant pathologies.
*
Imaging:
*
Plain Radiographs:
Essential initial imaging. Standard views include true anteroposterior (AP) (Grashey view), scapular Y-view, and axillary lateral view. Key findings include joint space narrowing, subchondral sclerosis, osteophytes, and subchondral cysts. Walch classification is a common system to categorize glenoid morphology and retroversion based on axillary views for surgical planning.
*
Computed Tomography (CT) Scan:
Indicated for detailed assessment of bone morphology, glenoid version, bone loss, and humeral head defects, which are crucial for preoperative planning, especially for shoulder arthroplasty. 3D reconstructions are invaluable.
*
Magnetic Resonance Imaging (MRI):
Less critical for primary GHOA diagnosis but useful for assessing soft tissue pathology such as rotator cuff integrity (e.g., in CTA), labral tears, and capsular status, or to rule out other causes of shoulder pain.
Surgical Anatomy & Biomechanics
A thorough understanding of the surgical anatomy and biomechanics of the glenohumeral joint is paramount for successful management of GHOA, particularly in the context of arthroplasty.
Glenohumeral Joint:
This is a diarthrodial, multiaxial ball-and-socket joint, renowned for its extensive range of motion but inherently unstable due to the small glenoid fossa articulating with a large humeral head (glenoid-to-humeral head ratio typically 1:4).
-
Bony Anatomy:
- Humeral Head: Approximately one-third of a sphere, typically retroverted by 20-30 degrees relative to the epicondylar axis and inclined superiorly by 130-140 degrees relative to the humeral shaft. The articular surface is bordered by the anatomic neck, with the surgical neck distal to the greater and lesser tuberosities.
- Glenoid: A shallow, pear-shaped fossa on the lateral aspect of the scapula. Its articular surface is typically retroverted by 0-7 degrees relative to the scapular body and inclined superiorly by 5-10 degrees. GHOA often leads to progressive posterior glenoid erosion and retroversion, requiring careful management during arthroplasty. The Walch classification for glenoid morphology (A1, A2, B1, B2, C) is critical for surgical planning, particularly B2 (biconcave, posterior erosion) and C (severely retroverted) glenoids, which pose significant challenges.
- Scapula: Provides the stable base for glenoid articulation. Its position and movement (scapulothoracic rhythm) are crucial for optimal shoulder function.
- Clavicle: Articulates with the acromion (AC joint) and sternum. While the AC joint is separate, its pathology (e.g., distal clavicle osteolysis, severe AC OA) can contribute to overall shoulder pain and may warrant concomitant resection during shoulder arthroplasty.
-
Soft Tissue Stabilizers:
- Labrum: A fibrocartilaginous ring that deepens the glenoid fossa, increasing its concavity and contact area. Its integrity is vital for joint stability.
- Joint Capsule: A fibrous sac surrounding the joint, reinforced by ligaments. It is redundant inferiorly to allow for abduction.
- Glenohumeral Ligaments (SGHL, MGHL, IGHL complex - anterior and posterior bands): Thickened bands within the capsule that provide static stability, particularly at the extremes of motion. The IGHL complex is the primary anterior stabilizer in abduction and external rotation.
-
Rotator Cuff:
The dynamic stabilizers of the shoulder, comprising four muscles:
- Supraspinatus: Initiates abduction, depresses humeral head.
- Infraspinatus & Teres Minor: External rotators, depress humeral head.
- Subscapularis: Internal rotator, depresses humeral head.
- Collectively, the rotator cuff muscles create a "concavity-compression" effect, centralizing the humeral head within the glenoid fossa and resisting translation. Their integrity is fundamental for successful conventional TSA. In CTA, their failure leads to superior migration and eccentric loading.
- Deltoid Muscle: The primary abductor of the shoulder, critical for elevating the arm. Its integrity is essential for function after both TSA and rTSA.
Biomechanics of GHOA:
The normal glenohumeral joint relies on a delicate balance of static and dynamic stabilizers. In GHOA:
*
Cartilage Degradation:
Increases friction and alters load distribution, leading to pain and accelerated wear.
*
Osteophyte Formation:
Restricts motion, particularly external rotation and abduction.
*
Capsular Contracture:
Further limits range of motion.
*
Altered Joint Kinematics:
The loss of the smooth articular surface and changes in glenoid/humeral head morphology (e.g., posterior glenoid erosion, humeral head subluxation) disrupt the normal rolling and sliding mechanisms, leading to aberrant motion patterns and increased joint contact stresses.
*
Rotator Cuff Dysfunction (in CTA):
In massive rotator cuff tears, the superior stabilizing force is lost, allowing the deltoid to cause superior migration of the humeral head. This shifts the center of rotation superiorly and medially, leading to "femoralization" of the acromion and "acetabularization" of the proximal humerus, and progressive erosion. This profound biomechanical alteration is what makes rTSA the preferred treatment for CTA, as it restores the deltoid lever arm and medializes/distalizes the center of rotation.
Neurovascular Structures of Concern (relevant to surgical approaches):
*
Axillary Nerve:
Arises from the posterior cord (C5-C6). Supplies the deltoid and teres minor, and sensory innervation to the lateral deltoid region. Highly vulnerable during inferior capsular dissection and deltoid retraction, particularly with inferior glenoid exposure.
*
Musculocutaneous Nerve:
Arises from the lateral cord (C5-C7). Supplies the biceps and coracobrachialis. Vulnerable during pectoralis major mobilization, lying typically 5-8 cm distal to the coracoid process.
*
Brachial Plexus:
Proximal to the shoulder joint, can be injured by excessive traction or malpositioning.
*
Cephalic Vein:
Lies in the deltopectoral groove, a key landmark for the deltopectoral approach.
*
Axillary Artery and Vein:
Lie deep and medial to the glenohumeral joint. At risk with aggressive glenoid reaming or screw placement, especially with anterior-inferior osteophytes.
Indications & Contraindications
The decision to proceed with surgical intervention for GHOA is complex, requiring careful consideration of patient factors, disease severity, and the potential benefits versus risks of surgery. Initial management is almost universally non-operative.
Non-Operative Indications:
Conservative management is the first-line treatment for nearly all patients with GHOA, particularly those with mild-to-moderate symptoms or significant comorbidities.
*
Mild-to-moderate pain and functional impairment:
Symptoms are tolerable and do not significantly impact activities of daily living.
*
Failure to exhaust non-operative treatments:
Patients who have not adequately attempted or responded to a structured conservative regimen.
*
Patient preference:
Some patients may prefer to defer surgery due to personal choice, fear of surgery, or other life circumstances.
*
Significant medical comorbidities:
Conditions that increase surgical risk (e.g., uncontrolled cardiac disease, severe pulmonary dysfunction, bleeding disorders) may contraindicate or delay surgery.
*
Active infection elsewhere in the body.
*
Unrealistic patient expectations.
Operative Indications:
Surgical intervention is considered when conservative measures have failed to provide adequate pain relief and functional improvement, and the patient's quality of life is significantly compromised.
*
Severe, intractable pain:
Debilitating pain refractory to all non-operative modalities, including NSAIDs, physical therapy, activity modification, and intra-articular injections (corticosteroid, hyaluronic acid). Nocturnal pain is a common indicator of advanced disease.
*
Significant functional limitation:
Inability to perform activities of daily living (ADLs) such as dressing, bathing, reaching, or sleeping comfortably.
*
Radiographic evidence of advanced GHOA:
Typically characterized by severe joint space narrowing, subchondral sclerosis, extensive osteophyte formation, and potentially subchondral cysts (Samuelson or Outerbridge grades III-IV, or Walch B2/C glenoids).
*
Failure of other surgical interventions:
Prior arthroscopic debridement or osteotomy that has not provided lasting relief.
*
Younger patients with focal defects:
May be candidates for cartilage restoration procedures (e.g., osteochondral allograft, autologous chondrocyte implantation) if the lesion is small and contained, though this is less common for diffuse GHOA.
*
Patient desire:
A well-informed patient who understands the risks and benefits and is motivated for rehabilitation.
Contraindications for Arthroplasty (TSA or rTSA):
*
Absolute Contraindications:
*
Active infection (systemic or local):
Particularly periprosthetic joint infection (PJI) or septic arthritis. This necessitates a two-stage revision strategy.
*
Paralysis of the deltoid and rotator cuff:
For conventional TSA, absence of a functional rotator cuff (especially subscapularis) is a contraindication. For rTSA, absence of a functional deltoid is an absolute contraindication, as it is the primary motor.
*
Irreparable axillary nerve injury:
Contraindication for rTSA due to inability to power the deltoid.
*
Uncontrolled systemic inflammatory disease:
Which could lead to rapid implant loosening or infection.
*
Severe medical comorbidities:
That make the patient an unacceptable anesthetic or surgical risk.
*
Neuropathic arthropathy (Charcot joint):
Due to high failure rates.
*
Patient unwilling or unable to participate in a rigorous post-operative rehabilitation program.
*
Relative Contraindications:
*
Significant glenoid bone loss:
While challenging, can often be addressed with bone grafting or augmented glenoid components.
*
Compromised skin or soft tissue:
Over the surgical site, increasing infection risk.
*
Previous irradiation of the shoulder:
Impairs healing and increases infection risk.
*
Psychiatric illness or substance abuse:
May hinder compliance and rehabilitation.
*
Osteoporosis:
Increases risk of periprosthetic fracture and component loosening, though not an absolute contraindication.
Table: Operative vs. Non-Operative Indications for GHOA
| Feature | Non-Operative Indications | Operative Indications (TSA/rTSA) |
|---|---|---|
| Pain Severity | Mild to moderate, manageable with medication/activity mod. | Severe, intractable, debilitating pain, refractory to conservative Rx. |
| Functional Impact | Minimal to moderate impairment in ADLs. | Significant impairment in ADLs, inability to work/participate in leisure. |
| Radiographic Findings | Early to moderate joint space narrowing, mild osteophytes. | Advanced joint space narrowing, subchondral sclerosis, cysts, large osteophytes (Walch B2/C). |
| Response to PT/Injections | Good or acceptable improvement. | Failed all non-operative modalities (PT, NSAIDs, injections). |
| Patient Age/Activity | Younger or less active patients; comorbidities. | Active, motivated patient seeking pain relief and functional restoration. |
| Medical Status | Significant comorbidities precluding surgery. | Medically optimized for surgery. |
| Shoulder Integrity | Intact rotator cuff (for conventional TSA consideration). | Intact rotator cuff (TSA) OR massive, irreparable cuff tear with CTA/pseudoparalysis (rTSA). |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning and appropriate patient positioning are critical for optimizing outcomes and minimizing complications in shoulder arthroplasty.
Pre-Operative Planning:
1.
Patient Evaluation:
*
Detailed History & Physical:
Confirm diagnosis, assess severity of symptoms, range of motion, strength, neurovascular status. Document pre-existing comorbidities (cardiac, pulmonary, renal, metabolic, infectious) and current medications (anticoagulants, immunosuppressants).
*
Patient Expectations:
Crucial for managing post-operative satisfaction. Discuss realistic outcomes, rehabilitation demands, and potential activity restrictions.
2.
Imaging Review:
*
Standard Radiographs:
True AP (Grashey), scapular Y, and axillary views are essential for initial assessment of joint space narrowing, osteophytes, sclerosis, and subluxation.
*
Computed Tomography (CT) Scan with 3D Reconstruction:
The gold standard for assessing glenoid morphology (version, inclination, bone loss), humeral head retroversion, and humeral shaft anatomy. This is critical for templating the glenoid component (especially for Walch B2/C glenoids) and humeral stem.
*
Magnetic Resonance Imaging (MRI):
If rotator cuff integrity is uncertain or there are concerns about other soft tissue pathologies (e.g., AVN, labral pathology), MRI provides valuable information, especially when considering conventional TSA versus rTSA.
3.
Templating:
*
Humeral Component:
Determine appropriate humeral head size, stem diameter and length, and offset. Aim to restore the anatomical offset and version. Stemless components are increasingly used for primary GHOA, necessitating careful consideration of proximal humeral bone stock.
*
Glenoid Component (TSA):
Select appropriate size and reaming strategy to correct glenoid retroversion and achieve stable fixation. Consider eccentric reaming, posterior glenoid bone grafting, or augmented glenoids for Walch B2/C deformities.
*
Glenoid Component (rTSA):
Critically important. Determine baseplate size and position (typically inferiorly and centrally or slightly anteriorly to minimize scapular notching). Select appropriate glenosphere size and offset. Consider bone grafting for significant glenoid bone loss.
*
Pre-operative Software Planning:
Advanced 3D planning software allows virtual implantation and assessment of component positioning, bone coverage, and range of motion, which is particularly beneficial for complex cases.
4.
Blood Management:
Type and cross-match blood if significant blood loss is anticipated (less common in primary arthroplasty but relevant for revisions). Consider tranexamic acid.
5.
Infection Prophylaxis:
Standard intravenous antibiotics (e.g., cefazolin) within 60 minutes of incision.
6.
Anesthesia Consultation:
Assess patient's fitness for general anesthesia and consider regional nerve blocks (interscalene block) for post-operative pain management.
Patient Positioning:
The
beach chair position
is the most commonly utilized position for shoulder arthroplasty, offering excellent exposure and surgeon comfort. The
lateral decubitus position
is an alternative, more commonly used for arthroscopic procedures or complex revisions where gravity-assisted traction is desired.
Beach Chair Position (Key Considerations):
1.
Operating Table:
Standard operating table with a beanbag or specialized shoulder support.
2.
Torso Positioning:
* The patient is positioned semi-supine, with the torso elevated 30-70 degrees from the horizontal. A 45-degree angle is typical.
* The patient's buttock should be at the break in the operating table to allow for slight Trendelenburg if needed.
* Secure the torso with a beanbag or straps to prevent sliding during surgery.
3.
Head and Neck:
* The head is secured in a well-padded headrest (e.g., horseshoe headrest or specific shoulder chair headrest) to prevent cervical spine injury and allow neutral alignment.
* Ensure adequate padding to protect pressure points (occiput).
* The neck should be slightly flexed, and the head turned contralateral to the operative shoulder, ensuring the surgical field is clear.
* Confirm adequate clearance between the operative shoulder and the headrest to allow for full range of motion of the arm during surgery.
4.
Operative Arm:
* The operative arm is typically free-draped and positioned on a sterile Mayo stand or an adjustable arm holder.
* This allows for dynamic intra-operative manipulation (flexion, extension, rotation) by the surgical assistant to facilitate exposure and implant insertion.
* Ensure adequate padding in the axilla to prevent neurovascular compression.
5.
Non-Operative Arm:
* Secured and padded at the patient's side, ensuring no compression of the ulnar nerve at the elbow or brachial plexus in the axilla.
6.
Lower Extremities:
* Well-padded and secured, typically with knees slightly flexed to prevent nerve compression.
* Ensure proper circulation and avoid excessive pressure on calves/heels to prevent DVT.
7.
Fluoroscopy:
Ensure adequate access for intra-operative fluoroscopy (C-arm) if needed, especially for glenoid component placement in rTSA or complex revisions. The C-arm typically approaches from the contralateral side of the table.
8.
Sterile Preparation and Draping:
Wide sterile prep extending from the neck to the mid-chest, including the entire upper extremity. Draping must allow for full intra-operative manipulation of the arm.
Detailed Surgical Approach / Technique
The primary surgical solutions for GHOA are Total Shoulder Arthroplasty (TSA) and Reverse Total Shoulder Arthroplasty (rTSA) . Hemiarthroplasty (HA) is less commonly performed for primary GHOA due to inferior outcomes compared to TSA, mainly due to persistent glenoid pain, but may be used in younger patients with intact glenoid cartilage or specific fracture patterns. Resection arthroplasty and arthrodesis are salvage procedures. This section will focus on TSA and rTSA via the deltopectoral approach.
I. Deltopectoral Approach (Common for both TSA and rTSA)
This is the workhorse approach for most shoulder arthroplasties, offering reliable exposure with low risk to major neurovascular structures when performed correctly.
- Incision: A curvilinear incision typically 10-15 cm in length, starting near the coracoid process, extending distally along the deltopectoral groove, and curving slightly laterally over the proximal humerus.
-
Superficial Dissection:
- Sharp dissection through skin and subcutaneous tissue.
- Identify the cephalic vein within the deltopectoral groove. This is the key landmark. The vein is typically retracted medially with the pectoralis major, or laterally with the deltoid, or ligated if necessary (though generally avoided).
- The deltopectoral interval is then developed by blunt dissection, separating the anterior deltoid from the pectoralis major.
-
Deep Dissection:
- Retraction: Retract the deltoid laterally and the pectoralis major medially.
- Clavipectoral Fascia: Incise the clavipectoral fascia inferior to the coracoid to expose the conjoint tendon (coracobrachialis and short head of biceps) and the underlying subscapularis muscle.
- Coracoid Process: Palpate the coracoid. No detachments are typically needed, but in revision cases or for increased exposure, a partial detachment of the pectoralis minor insertion or even osteotomy of the coracoid can be performed (with careful attention to the musculocutaneous nerve).
- Internervous Plane: The deltopectoral interval is a true internervous plane. The deltoid is supplied by the axillary nerve, and the pectoralis major by the medial and lateral pectoral nerves.
II. Conventional Total Shoulder Arthroplasty (TSA)
Indications:
Primary GHOA with an intact, functional rotator cuff.
Goal:
Restore anatomical joint mechanics, achieve smooth articulation, and relieve pain while preserving rotator cuff function.
-
Exposure of the Glenohumeral Joint:
-
Subscapularis Management:
This is critical.
- Lesser Tuberosity Osteotomy: Most common and preferred technique. An osteotomy is performed at the insertion of the subscapularis onto the lesser tuberosity. The osteotomized fragment, with the attached subscapularis, is then retracted medially. This preserves the tendon length and bony insertion for later repair.
- Subscapularis Tenotomy: The tendon is sharply divided at its insertion. This may be preferred in cases of severe contracture or revision, but tendon lengthening and healing may be more challenging.
- Capsulotomy: A T-shaped or inverted T-shaped capsulotomy is performed to expose the humeral head and glenoid. Remove anterior osteophytes.
- Humeral Head Dislocation: The humeral head is gently dislocated anteriorly and delivered into the wound.
-
Subscapularis Management:
This is critical.
-
Humeral Head Preparation:
- Humeral Head Resection: Specialized cutting guides are used to resect the humeral head at the appropriate angle (typically 30-45 degrees of retroversion relative to the epicondylar axis and 130-140 degrees of inclination). The amount of resection aims to restore the joint line and allow proper soft tissue tensioning.
- Humeral Canal Preparation: Reamers and broaches are used sequentially to prepare the humeral medullary canal for the chosen humeral stem. Attention to humeral version is paramount.
- Trial Implants: Trial humeral stem and head are inserted to assess fit, offset, and version.
-
Glenoid Preparation:
- Exposure: The arm is internally rotated and adducted to maximize glenoid exposure. Careful removal of posterior osteophytes (the "comma sign") is important to allow proper glenoid exposure and to reduce posterior impingement. Protect the axillary nerve inferiorly.
- Cartilage Removal and Reaming: The remaining articular cartilage is removed. Specialized reamers are used to prepare the glenoid fossa. The goal is to correct glenoid retroversion (ideally to neutral or slight anteversion) and achieve stable fixation. Eccentric reaming (removing more posterior bone) or posterior bone grafting may be required for significant Walch B2/C retroversion.
- Drill Holes: Drill holes for the pegs/keel of the glenoid component.
- Trial Glenoid: Insert a trial glenoid component to assess stability, congruity, and coverage.
- Definitive Glenoid Implantation: The glenoid component (typically polyethylene, cemented) is then implanted. Meticulous cement technique is essential for longevity. Hybrid (metal-backed with poly liner) or uncemented glenoids are less common in primary TSA due to higher aseptic loosening rates.
-
Humeral Stem Implantation:
- Definitive Humeral Component: The definitive humeral stem (cemented or uncemented, stemmed or stemless) is implanted with the appropriate head component. Ensure correct version and offset.
- Reduction: The humeral head is reduced into the glenoid component.
- Soft Tissue Balancing and Stability: Assess the stability through a full range of motion. It should be stable in all positions, particularly external rotation. Evaluate soft tissue tension.
-
Closure:
- Subscapularis Repair: The lesser tuberosity osteotomy fragment (with subscapularis) is securely reattached to the humerus using non-absorbable sutures, typically through transosseous tunnels or suture anchors. This is the most critical step for functional recovery and preventing anterior instability.
- Capsular Closure: If a capsulotomy was performed, it is repaired.
- Deltopectoral Interval: Closed with absorbable sutures.
- Subcutaneous and Skin Closure: Layered closure. Drain may be placed, though increasingly omitted.
III. Reverse Total Shoulder Arthroplasty (rTSA)
Indications:
Cuff Tear Arthropathy (CTA), irreparable rotator cuff tears with pseudoparalysis, failed TSA with rotator cuff deficiency, complex proximal humeral fractures in elderly patients, revision surgery, certain tumors.
Goal:
Restore deltoid function, medializing and distalizing the center of rotation to bypass the non-functional rotator cuff, and improve pain and elevation.
- Exposure (Deltopectoral Approach): Identical to TSA, as described above. Subscapularis management may vary; it is often deficient or irreparable in CTA, so repair may not be feasible or necessary.
-
Humeral Head Resection:
- The humeral head is resected, often with a slightly more medial cut than TSA, depending on the chosen implant system, to accommodate the humeral tray.
- The cut typically aims to remove the articular surface and allow for the humeral tray and liner.
-
Humeral Canal Preparation:
- Reamers and broaches prepare the humeral canal for the humeral stem (cemented or uncemented).
- The humeral tray is implanted, and a trial polyethylene liner is placed.
-
Glenoid Preparation (Critical Steps):
- Exposure: Meticulous exposure is required, often necessitating removal of significant anterior and posterior osteophytes. Careful protection of the axillary nerve inferiorly is paramount.
- Glenoid Reaming: The glenoid is reamed to a flat, bleeding surface. The goal is to achieve a stable base for the glenoid baseplate.
-
Baseplate Positioning:
This is the most critical step for rTSA longevity and avoiding complications like scapular notching.
- The baseplate should be positioned inferiorly on the glenoid to allow maximum overhang of the glenosphere without impingement on the inferior glenoid neck (reducing scapular notching).
- It should also be well-seated on healthy bone, often slightly anteriorly on the glenoid to provide stability and tension.
- Screw Fixation: Multiple bicortical screws are used to fix the baseplate. At least two divergent screws (typically superior and inferior) are critical for robust fixation. Screw length and trajectory must be carefully controlled to avoid penetration into the chest cavity or neurovascular structures.
- Bone Grafting: For significant glenoid bone loss, structural bone grafts (autograft or allograft) may be necessary to restore bone stock and provide a stable platform for the baseplate.
- Glenosphere Implantation: The definitive glenosphere (typically cobalt-chrome or titanium) is then attached to the baseplate. Various sizes and offsets are available to optimize deltoid tension and range of motion.
-
Reduction and Tensioning:
- The humeral polyethylene liner is snapped onto the glenosphere.
- The joint is reduced.
- Stability and Tension: Assess stability through a full range of motion. The primary goal is to ensure adequate deltoid tension for active elevation without excessive tension that could lead to nerve injury or early loosening. The arm should achieve at least 90 degrees of abduction and flexion.
- Lateralization: Many modern rTSA systems incorporate various degrees of lateralization (offset glenospheres, lateralized baseplates, or augmented liners) to increase the deltoid moment arm and potentially reduce scapular notching.
-
Closure:
- Capsule and Subscapularis: The capsule and subscapularis (if present and repairable) may be loosely closed, but often they are deficient in rTSA cases.
- Deltopectoral Interval and Layers: Closed as in TSA.
Complications & Management
Shoulder arthroplasty, while highly effective, is associated with a range of potential complications, both general surgical and specific to the procedure type. Vigilant intraoperative technique and thorough postoperative surveillance are essential.
Table: Common Complications of Shoulder Arthroplasty and Management
| Complication | Incidence (Approx.) | Management Strategy |
|---|---|---|
| Infection (PJI) | 1-4% | Acute (<4 weeks): Irrigation & Debridement (I&D) with component retention (especially if stable, well-fixed components), systemic antibiotics (IV then oral). Subacute/Chronic (>4 weeks): Two-stage revision (component removal, debridement, antibiotic spacer, then reimplantation after infection eradication). Excision arthroplasty or arthrodesis for recalcitrant cases. |
| Glenoid Loosening (TSA) | 5-15% (at 10 yrs) | Aseptic: Revision glenoid component (with or without humeral component revision depending on status). May require bone grafting for glenoid defects. Consider rTSA as a revision option if rotator cuff is compromised or significant glenoid bone loss. Septic: As above for PJI. |
| Rotator Cuff Failure (TSA) | 5-10% (subscap.) | Subscapularis failure: Surgical repair (direct repair, tendon transfer), potentially with revision of humeral component or conversion to rTSA if cuff is irreparable and symptoms severe. Supraspinatus/Infraspinatus failure: Less common to cause revision in TSA unless leading to superior humeral migration; often managed conservatively. |
| Instability/Dislocation | 1-5% (TSA), 2-10% (rTSA) | TSA (often anterior): Closed reduction, sling immobilization. Recurrent dislocations may require revision for component malposition, soft tissue imbalance, or subscapularis failure. rTSA (often inferior-posterior, impingement): Closed reduction. If recurrent, evaluate component positioning (glenosphere size/offset, baseplate position), deltoid tension, scapular notching. May require revision of glenosphere/liner, soft tissue augmentation, or component repositioning. |
| Periprosthetic Fracture | 1-3% | Intraoperative: Depends on location and stability. Cerclage wiring, open reduction internal fixation (ORIF), or revision with a longer stem. Postoperative: Depends on location, displacement, and implant stability. ORIF, non-operative management (sling), or revision arthroplasty (especially for humeral component loosening). |
| Neuropathy (Axillary, Brachial Plexus) | 1-5% (higher in rTSA) | Axillary Nerve: Often transient. Non-operative management with observation, neurophysiological studies, physical therapy. Surgical exploration and neurolysis or repair if persistent or severe deficit (e.g., in rTSA, due to lengthening). Brachial Plexus: Usually due to traction. Remove inciting factor (e.g., arm traction), observe. |
| Scapular Notching (rTSA) | 50-90% (radiographic) | Often asymptomatic. If symptomatic (pain, limited ROM, audible clunking), assess severity. Strategies to prevent include inferior glenoid baseplate placement, lateralized glenospheres/liners, eccentric glenospheres. Management if symptomatic: conservative (PT), or revision with further lateralization, débridement of impinged bone, or conversion to a different rTSA design. |
| Acromial/Scapular Spine Fracture (rTSA) | 2-5% | Non-displaced/stable: Non-operative management with immobilization (sling) and protected range of motion. Displaced/unstable: Surgical fixation (ORIF) if bone stock permits, or non-operative if fixation is not feasible. May lead to deltoid dysfunction and poor outcomes. |
| Humeral Loosening (Aseptic) | <1% (TSA), 1-5% (rTSA) | TSA: Revision with a new humeral stem (longer, different fixation) or conversion to rTSA if glenoid is also loose or rotator cuff is compromised. rTSA: Revision of humeral stem. Requires careful bone preparation and sometimes bone grafting. |
| Stiffness/Arthrofibrosis | 5-10% | Conservative: Aggressive physical therapy, pain management, sometimes intra-articular injections. Surgical: Manipulation under anesthesia (MUA) if early and motion lost. Arthroscopic or open capsular release for recalcitrant cases. |
| Vascular Injury | <0.1% | Immediate surgical exploration, vascular repair (arterial/venous), or grafting. Requires rapid recognition and intervention. |
| Heterotopic Ossification | 10-20% (radiographic) | Often asymptomatic. Prophylaxis with NSAIDs (e.g., indomethacin) or radiation therapy in high-risk patients. If symptomatic and severe, surgical excision (after maturation) may be considered, but recurrence is high. |
General Principles of Complication Management:
*
Early Recognition:
Prompt identification of complications is key to effective management.
*
Systemic Evaluation:
Rule out systemic causes (e.g., infection) before focusing on local issues.
*
Imaging:
Utilize appropriate imaging (X-rays, CT, MRI, bone scan) to characterize the problem.
*
Multidisciplinary Approach:
Involve infectious disease specialists, neurologists, and other subspecialists as needed.
*
Patient Communication:
Maintain open and honest communication with the patient regarding the nature of the complication, treatment options, and expected outcomes.
*
Salvage Procedures:
In cases of severe, irreparable failure, salvage options like arthrodesis (fusion), resection arthroplasty (excision of components, typically for recalcitrant infection or elderly, low-demand patients), or amputation may be considered as last resorts.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an integral and critical component of successful shoulder arthroplasty, directly influencing functional outcomes, pain relief, and implant longevity. Protocols vary between surgeons, implant types (TSA vs. rTSA), and specific patient factors, but generally follow a phased approach designed to protect the healing tissues while progressively restoring motion and strength.
I. General Principles:
*
Patient Education:
Crucial for compliance. Patients must understand the goals of each phase, precautions, and anticipated timelines.
*
Pain Management:
Adequate pain control is essential to allow for rehabilitation participation.
*
Protection of Surgical Repair:
Especially the subscapularis in TSA and the deltoid in rTSA.
*
Gradual Progression:
Avoid overstressing healing tissues.
*
Individualization:
Protocols should be adapted to the patient's progress, pain level, and any intraoperative findings (e.g., quality of subscapularis repair).
*
Communication:
Close collaboration between the surgeon and physical therapist is vital.
II. Conventional Total Shoulder Arthroplasty (TSA) Protocol
Phase 1: Maximum Protection (Weeks 0-6)
*
Goals:
Protect surgical repair, minimize pain and swelling, initiate passive range of motion (PROM).
*
Immobilization:
Sling for 4-6 weeks continuously, only removed for exercises and personal hygiene.
*
Precautions:
*
NO active internal rotation, adduction, or extension against resistance (especially for subscapularis repair).
*
Avoid reaching behind the back.
*
No lifting, pushing, or pulling with the operative arm.
*
Avoid sudden movements.
*
Exercises (Daily):
*
Passive Range of Motion (PROM):
Pendulum exercises, supine passive forward flexion (up to 90-120 degrees depending on surgeon preference), supine passive external rotation (limited to 30-45 degrees depending on subscapularis repair integrity and implant stability). Assisted by therapist or non-operative arm using a stick.
*
Elbow, Wrist, Hand ROM:
Active exercises to prevent stiffness.
*
Scapular Retraction:
Gentle isometric exercises.
*
Cryotherapy:
For pain and swelling.
Phase 2: Moderate Protection / Early Strengthening (Weeks 6-12)
*
Goals:
Progress PROM, initiate active-assisted range of motion (AAROM) and active range of motion (AROM), begin gentle strengthening.
*
Immobilization:
Sling discontinued, or used for comfort/protection in crowds.
*
Precautions:
*
Continue to avoid heavy lifting, pushing, pulling.
*
No aggressive stretching, especially into end-range external rotation and extension.
*
Exercises:
*
AAROM/AROM:
Progress forward flexion, abduction, external/internal rotation within pain-free limits. Use pulleys, stick exercises.
*
Isometric Strengthening:
Gentle isometric exercises for deltoid and rotator cuff (supraspinatus, infraspinatus, subscapularis) in neutral positions.
*
Scapular Stabilization:
Progress exercises for periscapular muscles.
*
Light Functional Activities:
Begin with light ADLs.
Phase 3: Minimal Protection / Progressive Strengthening (Weeks 12-24)
*
Goals:
Restore full functional ROM, achieve good strength, prepare for return to light activities.
*
Precautions:
*
Avoid heavy impact sports, heavy overhead lifting.
*
Exercises:
*
Progressive Resistive Exercises (PREs):
Using resistance bands, light weights for all major shoulder muscle groups (rotator cuff, deltoid, biceps, triceps).
*
Proprioceptive Training:
Balance and coordination exercises.
*
Functional Training:
Simulate desired activities, gradually increase resistance and complexity.
Phase 4: Return to Activity / Maintenance (Months 6+ & Lifetime)
*
Goals:
Maximize strength and endurance, maintain ROM, return to appropriate activities.
*
Restrictions:
Permanent restrictions on high-impact sports, contact sports, heavy overhead lifting (e.g., >5-10 kg). Avoid activities with risk of falls or direct impact to the shoulder.
*
Maintenance:
Continue a home exercise program (HEP) to maintain strength and flexibility.
III. Reverse Total Shoulder Arthroplasty (rTSA) Protocol
rTSA rehabilitation often differs due to the altered biomechanics and the reliance on the deltoid as the primary mover. Subscapularis repair is often not performed or is less critical than in TSA.
Phase 1: Maximum Protection (Weeks 0-4/6)
*
Goals:
Protect surgical site, allow soft tissue healing, initiate early PROM and AAROM to avoid stiffness.
*
Immobilization:
Sling for 3-4 weeks (shorter than TSA due to altered biomechanics), often removed for exercises.
*
Precautions:
*
Avoid combined internal rotation, adduction, and extension past neutral (dislocation risk).
*
Avoid extremes of external rotation and extension (dislocation risk).
*
No lifting, pushing, pulling with the operative arm.
*
Focus on protected movements.
*
Exercises (Daily):
*
Passive & Active-Assisted Range of Motion (PROM/AAROM):
Pendulum, supine passive forward flexion (often can achieve higher ranges earlier than TSA), supine external rotation limited to 0-20 degrees, internal rotation to chest.
*
Scapular Mobility:
Gentle exercises.
*
Elbow, Wrist, Hand ROM.
*
Cryotherapy.
Phase 2: Moderate Protection / Early Deltoid Strengthening (Weeks 4-12)
*
Goals:
Progress AROM and initiate active deltoid strengthening, improve functional use.
*
Immobilization:
Sling typically discontinued.
*
Precautions:
*
Continue to avoid combined movements that cause impingement or dislocation risk.
*
Avoid heavy lifting.
*
Exercises:
*
Active Range of Motion (AROM):
Progress forward flexion, abduction, rotation (often better elevation than rotation).
*
Deltoid Strengthening:
Gentle isometric and then light isotonic exercises for anterior, middle, and posterior deltoid. This is paramount for rTSA function.
*
Scapular Stabilizer Strengthening:
Essential for a stable base of support for the deltoid.
*
Core Strengthening:
To improve overall stability.
Phase 3: Progressive Strengthening & Functional Activities (Weeks 12-24)
*
Goals:
Maximize deltoid strength, achieve maximum functional return.
*
Precautions:
*
Similar restrictions to TSA for high-impact activities.
*
Exercises:
*
Progressive Resistive Exercises:
Increase resistance for deltoid and periscapular muscles.
*
Eccentric Training:
For deltoid.
*
Functional Task Training:
Specific exercises tailored to patient's desired activities.
Phase 4: Return to Activity / Maintenance (Months 6+ & Lifetime)
*
Goals:
Maintain strength, endurance, and ROM.
*
Restrictions:
Similar lifelong restrictions as TSA. Emphasize avoidance of excessive stress on the deltoid and activities that risk dislocation.
*
Maintenance:
Lifelong HEP.
Summary of Key Literature / Guidelines
The field of shoulder arthroplasty for GHOA has seen significant advancements, supported by a growing body of literature and evolving guidelines from professional organizations. Key themes in recent research focus on implant design, surgical technique refinements, complication management, and patient-reported outcomes.
I. Total Shoulder Arthroplasty (TSA):
*
Efficacy:
TSA remains the gold standard for treating primary GHOA with an intact rotator cuff, consistently demonstrating excellent long-term pain relief and functional improvement. Survivorship rates typically exceed 90% at 10 years and 80% at 15 years in well-selected patients.
*
Glenoid Component:
Aseptic loosening of the glenoid component is historically the most common mode of failure in TSA.
*
Cemented Polyethylene:
Remains the most widely used and proven glenoid fixation method. Studies by Wright et al. and Sanchez-Sotelo et al. emphasize meticulous cementing technique for optimal longevity.
*
Bone Grafting/Augments:
For significant posterior glenoid retroversion (Walch B2/C glenoids), studies by Walch, Levy, and Boileau have demonstrated the utility of posterior bone grafting or augmented glenoid components to restore glenoid version and prolong implant survival, reducing glenoid loosening rates and anterior humeral subluxation.
*
Uncemented Glenoids:
While promising in some series, they have generally shown higher revision rates for aseptic loosening compared to cemented components in large registry studies. Ongoing research explores newer designs and materials.
*
Humeral Component:
*
Stemmed:
Both cemented and uncemented stemmed humeral components show comparable results. Uncemented stems are increasingly popular for anatomical restoration and potential for easier revision, though concerns about stress shielding and periprosthetic fracture persist.
*
Stemless:
Emerging as a viable option, particularly in younger patients with good bone stock and primary GHOA. Meta-analyses suggest comparable short-to-midterm outcomes to stemmed prostheses, with potential advantages of bone preservation and easier revision. Long-term data is still maturing.
*
Subscapularis Management:
The integrity and proper repair of the subscapularis are paramount for TSA stability and function. Lesser tuberosity osteotomy is generally favored over tenotomy for its reliable healing and reduced risk of subscapularis insufficiency, as demonstrated by studies from multiple groups.
II. Reverse Total Shoulder Arthroplasty (rTSA):
*
Efficacy:
rTSA has revolutionized the treatment of massive, irreparable rotator cuff tears with pseudoparalysis (cuff tear arthropathy - CTA) and has expanded indications to complex fractures, revision arthroplasty, and severe GHOA with rotator cuff deficiency. It provides predictable pain relief and improved active elevation in this challenging patient population.
*
Complications:
While highly successful, rTSA has a distinct set of complications compared to TSA.
*
Scapular Notching:
Although often asymptomatic, radiographic scapular notching remains a common finding (50-90%). Boileau et al. extensively documented this phenomenon. Strategies to mitigate notching include inferior glenoid baseplate placement, lateralized glenosphere/baseplate designs, and eccentric glenospheres. The clinical significance of asymptomatic notching is debated.
*
Instability/Dislocation:
Rates vary (2-10%), often related to implant positioning, deltoid tension, or patient activity.
*
Acromial/Scapular Spine Fractures:
Occur in 2-5% of cases, often due to stress shielding or excessive deltoid tension. Outcomes are often poor after such fractures.
*
Neuropathy:
Axillary nerve palsy is a known risk, particularly with limb lengthening.
*
Design Evolution:
Early designs by Grammont (medialized center of rotation) paved the way. Newer designs focus on lateralizing the center of rotation (e.g., using lateralized baseplates or glenospheres) to improve deltoid tension, reduce scapular notching, and potentially enhance external rotation, as explored by Frankle, Gerber, and others.
*
Revision rTSA:
Increasingly common due to expanding indications and longer patient lifespans. It presents unique challenges due to bone loss and soft tissue compromise.
III. Patient-Reported Outcome Measures (PROMs):
* Increasingly used in literature and clinical practice to assess the patient's perspective on pain, function, and quality of life. Common PROMs include the American Shoulder and Elbow Surgeons (ASES) score, the Simple Shoulder Test (SST), and the Oxford Shoulder Score (OSS). These objective measures are crucial for comparing outcomes across studies and evaluating new techniques.
IV. Guidelines and Consensus Statements:
* Professional organizations (e.g., American Academy of Orthopaedic Surgeons - AAOS, European Society for Surgery of the Shoulder and Elbow - ESSKA) regularly publish clinical practice guidelines and consensus statements on the diagnosis, non-operative management, and surgical indications for GHOA and shoulder arthroplasty. These provide evidence-based recommendations for optimal patient care. Key areas of focus include appropriate patient selection, infection prophylaxis, and management of complications.
V. Future Directions:
*
Patient-Specific Instrumentation (PSI) and Navigation:
Offer enhanced precision in glenoid and humeral component placement, particularly for complex deformities.
*
Biologic Solutions:
Continued research into cartilage regeneration and biologic augmentation for early GHOA, though still largely experimental for diffuse disease.
*
Smart Implants:
Implants with sensors for monitoring load, temperature, or motion could optimize rehabilitation and detect early complications.
*
Expanded Indications:
Further understanding of rTSA in younger patients and specific fracture patterns.
In conclusion, shoulder arthroplasty represents a highly successful intervention for end-stage GHOA. Continuous research and adherence to evolving best practices are essential for maximizing patient outcomes and navigating the complexities of this challenging pathology.
Clinical & Radiographic Imaging