Decoding Shoulder Pain: Diagnosing Suprascapular Nerve Entrapment
Introduction & Epidemiology
Suprascapular nerve entrapment (SSNE) is a frequently overlooked cause of shoulder pain and dysfunction, often masquerading as more common pathologies such as rotator cuff tears, labral injuries, or glenohumeral arthritis. It represents a complex diagnostic challenge given its varied presentation and the deep, often insidious nature of the pain. The suprascapular nerve (SSN) is a mixed motor and sensory nerve, critical for shoulder function, innervating the supraspinatus and infraspinatus muscles and providing sensory feedback to the posterior and superior aspects of the shoulder capsule, acromioclavicular joint, and subacromial bursa. Entrapment can occur at several anatomical locations, leading to a spectrum of symptoms from vague, deep aching pain to profound weakness and atrophy of the rotator cuff musculature.
The epidemiology of SSNE is difficult to precisely quantify due to underdiagnosis, but its incidence is recognized in specific populations. Overhead athletes, particularly volleyball players, baseball pitchers, and tennis players, exhibit a higher prevalence, often related to repetitive microtrauma, traction injuries, or adaptive changes in scapular kinematics leading to dynamic entrapment. Lesions at the spinoglenoid notch, distal to the supraspinatus branch, are frequently observed in these athletes, manifesting primarily as infraspinatus atrophy and weakness, with or without pain. In the general population, causes include space-occupying lesions such as ganglion cysts (often associated with posterior labral tears), paralabral cysts, lipomas, or osteochondromas. Other etiologies involve direct trauma, fractures of the scapula, hypertrophy of the transverse scapular ligament or spinoglenoid ligament, scarring from previous surgery, and compression by venous engorgement, especially in cases of thoracic outlet syndrome or in specific occupational settings. An understanding of the various entrapment sites and their corresponding clinical presentations is paramount for accurate diagnosis and effective management.
Surgical Anatomy & Biomechanics
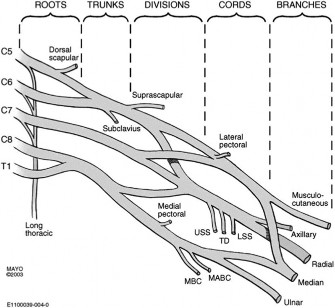
A thorough understanding of the detailed surgical anatomy and biomechanics of the suprascapular nerve is fundamental to diagnosing and treating SSNE. The SSN originates from the upper trunk of the brachial plexus, primarily from the C5 and C6 nerve roots, with occasional contributions from C4. It courses posteriorly and laterally, deep to the trapezius and omohyoid muscles, towards the superior border of the scapula.
Suprascapular Notch Entrapment
The most common site of entrapment is at the suprascapular notch, where the nerve passes beneath the superior transverse scapular ligament (STSL). This ligament can ossify, thicken, or narrow the notch, predisposing to compression. The suprascapular artery, conversely, typically courses
above
the STSL.
The nerve then enters the supraspinous fossa, supplying motor branches to the supraspinatus muscle and sensory branches to the posterior and superior glenohumeral capsule and the subacromial bursa. Entrapment here affects both supraspinatus and infraspinatus, leading to generalized weakness and atrophy of the rotator cuff muscles, often accompanied by diffuse, poorly localized shoulder pain that may radiate to the neck or arm.
Spinoglenoid Notch Entrapment
The SSN then wraps around the lateral border of the scapular spine, entering the infraspinous fossa through the spinoglenoid notch. At this second critical juncture, it passes beneath the inferior transverse scapular ligament (also known as the spinoglenoid ligament). This site is particularly vulnerable to compression by space-occupying lesions, such as ganglion cysts originating from posterior labral tears, or dynamic entrapment due to repetitive overhead motion. Entrapment at the spinoglenoid notch affects only the infraspinatus muscle, resulting in isolated infraspinatus atrophy and weakness, often with less severe or absent pain compared to suprascapular notch entrapment. The distinct clinical presentation helps localize the level of compression.
Nerve Biomechanics and Dynamic Entrapment
The biomechanics of the SSN are complex. During shoulder abduction and external rotation, the nerve is stretched and can be compressed against the osseous structures, particularly at the suprascapular notch. Repetitive microtrauma from overhead activities can lead to chronic inflammation, fibrosis, and nerve irritation. Dynamic compression by the spinoglenoid ligament or traction injuries from forceful adduction and internal rotation can further exacerbate nerve compromise. Scapular dyskinesis, where abnormal scapular motion alters the pathway of the nerve, can also contribute to dynamic entrapment. Understanding these dynamic factors is crucial, especially in athletic populations, as static imaging may not always reveal the underlying pathology.
Indications & Contraindications
The diagnosis of SSNE requires a high index of suspicion, correlating clinical findings with electrodiagnostic studies and advanced imaging. Indications for intervention, whether conservative or operative, are based on symptom severity, duration, response to initial non-operative management, and objective evidence of nerve compromise.
Clinical Evaluation and Diagnostic Process
The clinical presentation of SSNE is diverse. Patients typically report a deep, aching pain in the posterior or posterolateral aspect of the shoulder, often exacerbated by overhead activities or reaching across the body. Weakness in abduction and external rotation may be noted, along with visible atrophy of the supraspinatus and/or infraspinatus muscles. Tenderness may be elicited over the suprascapular notch or spinoglenoid notch. Specific provocative tests, such as the suprascapular nerve stretch test (neck flexion and contralateral rotation with shoulder adduction) or the cross-body adduction test, may reproduce symptoms. However, these are often non-specific.
Electrodiagnostic studies (electromyography and nerve conduction velocity studies) are paramount for objective confirmation of SSNE, providing information on nerve demyelination, axonal degeneration, and the level of entrapment. Findings such as increased distal motor latency, decreased nerve conduction velocity across the notch, and denervation potentials (fibrillations, positive sharp waves) or chronic neurogenic changes (polyphasic motor units) in the supraspinatus and/or infraspinatus muscles are highly suggestive. Magnetic resonance imaging (MRI) and magnetic resonance neurography (MRN) are crucial for visualizing the nerve, identifying space-occupying lesions (e.g., ganglion cysts, labral tears), evaluating muscle edema (acute denervation) or fatty atrophy (chronic denervation), and assessing the integrity of the ligaments. Diagnostic injection of local anesthetic into the suprascapular notch can also provide temporary relief and aid in diagnosis.
Indications for Management
| Category | Non-Operative Indications | Operative Indications |
|---|---|---|
| Symptoms | Mild to moderate pain, intermittent weakness. | Persistent, severe pain refractory to conservative care. Progressive motor weakness or atrophy. |
| Duration | Acute onset (within 3-6 months). | Chronic symptoms (typically >6 months) with objective evidence of entrapment. |
| Pathology | No clear structural compression (e.g., idiopathic entrapment, inflammatory causes). | Documented structural compression (e.g., ganglion cyst, paralabral cyst, ossified transverse scapular ligament, bony spur, tumor). |
| Response to Therapy | Improvement with physical therapy, activity modification, NSAIDs, local injections. | Failure of comprehensive, supervised conservative management (minimum 3-6 months). |
| Electrodiagnostics | Mild changes, no significant axonal loss, or improving nerve function. | Persistent or progressive denervation changes, significant axonal loss on EMG/NCS. |
| Imaging | No significant space-occupying lesion or nerve impingement identified. | Clear identification of a compressive lesion or severe narrowing of the suprascapular or spinoglenoid notch on MRI/MRN. Progressive muscle atrophy. |
| Patient Factors | Compliance with rehabilitation, desire for non-invasive approach. | Active individual (e.g., athlete) with performance limitations due to nerve compromise. |
Contraindications for Operative Intervention
Absolute contraindications for suprascapular nerve decompression are rare and primarily include severe medical comorbidities precluding surgery, active infection, or an alternative definitive diagnosis accounting for the patient's symptoms. Relative contraindications include:
*
Resolved Symptoms:
Spontaneous resolution of pain and weakness.
*
Unwillingness to Comply:
Patient non-compliance with post-operative rehabilitation protocols.
*
Mild or Non-Progressive Symptoms:
If symptoms are mild, non-bothersome, and stable, and do not significantly impact daily activities or athletic performance.
*
Irreversible Neuropathy:
In cases of extremely chronic, severe denervation with complete fatty atrophy of the supraspinatus and infraspinatus, the potential for motor recovery post-decompression may be limited, though pain relief can still be a valid goal.
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is essential for successful suprascapular nerve decompression, minimizing risks, and optimizing outcomes. This phase focuses on confirming the diagnosis, localizing the entrapment site, and preparing the patient for surgery.
Pre-Operative Planning
-
Diagnostic Confirmation:
- Clinical Examination: Re-evaluation of pain patterns, range of motion, and muscle strength. Palpation for atrophy and tenderness.
- Electrodiagnostic Studies (EMG/NCS): Review of most recent studies to confirm the level of entrapment (suprascapular vs. spinoglenoid notch) and assess the severity of nerve injury (demyelination vs. axonal loss), which influences prognostic expectations for recovery.
- Advanced Imaging (MRI/MRN): Detailed review of imaging to identify any space-occupying lesions (ganglion cysts, labral tears, tumors), hypertrophic ligaments, or bony spurs. MRN is particularly useful for visualizing the nerve's course and potential points of compression. The relationship of a cyst to the glenohumeral joint capsule and labrum must be fully understood if a cyst is present.
- Diagnostic Injection: If performed, confirm the efficacy of the suprascapular nerve block in transiently alleviating pain.
-
Surgical Approach Selection: The specific entrapment site dictates the surgical approach.
- Suprascapular Notch Decompression: Typically requires an open posterior approach, though an arthroscopic anterior approach combined with a posterior portal can be considered.
- Spinoglenoid Notch Decompression: Can often be performed arthroscopically, especially for ganglion cysts associated with posterior labral tears, or via a posterior open approach.
- Combined Entrapment: May require a hybrid or staged approach.
-
Anesthesia Consultation: Discussion with the anesthesiologist regarding regional anesthesia options (e.g., interscalene block) in addition to general anesthesia for post-operative pain management.
-
Informed Consent: Detailed discussion with the patient regarding the nature of the procedure, potential benefits (pain relief, strength improvement), risks (nerve injury, infection, incomplete decompression, recurrence), and expected recovery timeline.
Patient Positioning
Proper patient positioning is critical for adequate exposure, surgical safety, and ergonomic efficiency.
-
General Anesthesia: Intubation with general endotracheal anesthesia is standard.
-
Beach Chair Position (most common for open posterior approach):
- The patient is positioned semi-recumbent at approximately 45-60 degrees.
- The head is secured in a headrest, ensuring the neck is slightly flexed and rotated away from the operative shoulder to provide optimal access.
- The torso is tilted slightly away from the operative side, allowing the scapula to fall into a more prominent position.
- The operative arm is draped free to allow full range of motion intraoperatively for dynamic assessment and traction. It is often supported by an arm holder or table-mounted support.
- Padded pressure points are used to prevent nerve compression (e.g., ulnar nerve at the elbow, peroneal nerve at the fibular head) and skin breakdown.
- Ensure the ipsilateral hip is positioned at the break of the bed for optimal surgeon access.
-
Lateral Decubitus Position (alternative, especially for arthroscopic spinoglenoid notch decompression):
- The patient is positioned on their non-operative side.
- The operative arm is suspended in traction (approximately 45-60 degrees abduction and 15 degrees forward flexion) using an arm holder, with 10-15 lbs of traction weight.
- Axillary roll placed to protect the brachial plexus and ensure proper blood flow.
- Pillows placed between the knees and ankles.
- Careful padding of bony prominences.
- This position provides excellent posterior access, especially for arthroscopic management of spinoglenoid cysts and posterior labral tears.
Irrespective of the chosen position, ensuring adequate padding and meticulous attention to extremity positioning is paramount to prevent iatrogenic nerve injuries or pressure sores.
Detailed Surgical Approach / Technique
The surgical approach for suprascapular nerve decompression varies based on the site of entrapment (suprascapular notch vs. spinoglenoid notch) and the surgeon's preference (open vs. arthroscopic). The goal is precise nerve decompression while minimizing damage to surrounding structures.
Arthroscopic Suprascapular Nerve Decompression (Primarily Spinoglenoid Notch)
Arthroscopic techniques have become increasingly popular, particularly for spinoglenoid notch pathology like ganglion cysts associated with posterior labral tears.
-
Portals: Standard posterior portal (2 cm medial and 1 cm inferior to the posterolateral corner of the acromion) for visualization. A posterolateral portal, an accessory posterior portal (often more lateral and inferior to the standard posterior portal), or a Neviaser portal can be used for instrumentation. A superomedial portal (superior to the scapular spine, medial to the acromial angle) can be useful for direct visualization of the suprascapular notch area.
-
Diagnostic Arthroscopy: Initial comprehensive glenohumeral arthroscopy is performed to identify any intra-articular pathology, especially posterior labral tears, which are frequently the source of ganglion cysts causing SSNE at the spinoglenoid notch. If a labral tear is identified, it should be repaired.
-
Spinoglenoid Notch Decompression:
- Cyst Identification: The arthroscope is directed posteriorly. The infraspinatus fossa is explored, identifying the spinoglenoid notch. Ganglion cysts typically appear as encapsulated fluid collections compressing the nerve.
- Nerve Localization: The suprascapular nerve is identified as it passes through the spinoglenoid notch. It lies deep to the infraspinatus muscle belly. Neurolysis is performed by decompressing the cyst, either by aspirating its contents or, more commonly, by incising its capsule and marsupializing it into the infraspinous fossa. Care must be taken to avoid direct nerve injury.
- Ligament Release (if indicated): If the spinoglenoid ligament itself is hypertrophied and compressing the nerve, it can be carefully released using an arthroscopic shaver or radiofrequency ablator.
- Confirm Decompression: Visualize the nerve after decompression to ensure a tension-free course.
Open Suprascapular Nerve Decompression (Suprascapular Notch and Spinoglenoid Notch)
The open approach provides excellent visualization and is often preferred for more complex cases, ossified ligaments, or direct neurolysis at the suprascapular notch.
-
Incision: A transverse incision is made over the superior aspect of the scapula, approximately 2-3 cm inferior to the clavicle, extending from the acromioclavicular joint medially towards the medial border of the scapula, conforming to Langer's lines. Alternatively, a saber-cut incision along the scapular spine may be used for a more posterior approach.
-
Muscle Dissection and Internervous Plane:
- The skin and subcutaneous tissues are incised.
- The trapezius muscle is identified. For suprascapular notch decompression, the trapezius can be carefully split longitudinally along its fibers or incised and reflected. The omohyoid muscle, if present, is identified and retracted or partially released.
- The plane between the trapezius and supraspinatus fascia is developed.
- The supraspinous fossa is exposed by retracting the trapezius superiorly and medially.
-
Suprascapular Notch Identification and Decompression:
- The superior border of the scapula is palpated, leading to the suprascapular notch. The superior transverse scapular ligament (STSL) bridges this notch.
- The suprascapular artery typically lies above the STSL, while the nerve passes below it.
- Using a small curved hemostat or a nerve hook, the STSL is carefully isolated. It is then sharply incised longitudinally from lateral to medial, directly over the nerve, ensuring complete release. Magnification (loupes) is highly recommended.
- Great care must be taken to protect the nerve and the adjacent suprascapular artery, which can be intimately associated.
- After release, the nerve should be freely mobile. Any constricting fibrous bands or abnormal vessels are lysed.
-
Spinoglenoid Notch Identification and Decompression (if indicated via open approach):
- To access the spinoglenoid notch, the surgeon needs to continue dissecting more laterally and inferiorly.
- The supraspinatus muscle is identified and carefully retracted or split.
- The scapular spine serves as a key landmark. The nerve is followed distally along the supraspinous fossa, deep to the supraspinatus muscle.
- It then courses around the lateral border of the scapular spine to reach the infraspinous fossa.
- The spinoglenoid notch is located inferior to the scapular spine and medial to the glenoid.
- The spinoglenoid ligament (inferior transverse scapular ligament) may be identified, crossing over the nerve. Any ganglion cysts or compressive lesions are addressed as described for the arthroscopic technique, and the ligament is released if hypertrophied.
- Visualization requires careful dissection and retraction, ensuring protection of the nerve branches to the infraspinatus.
-
Closure:
- Hemostasis is meticulously achieved.
- The wound is irrigated.
- Muscle layers (if incised) are carefully reapproximated. The trapezius fascia is repaired.
- Subcutaneous tissues and skin are closed in layers.
- A sterile dressing is applied.
Key Surgical Principles
- Magnification: Use of surgical loupes is strongly encouraged for precise identification and protection of the nerve and vessels.
- Delicate Handling: The suprascapular nerve is delicate; direct traction, clamping, or excessive manipulation must be avoided.
- Complete Decompression: Ensure all constricting elements are released, whether ligamentous, osseous, or space-occupying lesions.
- Neurolysis: Perform neurolysis of the nerve if significant scarring or fibrosis is noted around the nerve.
- Labral Repair: If a posterior labral tear is the source of a ganglion cyst, concurrent arthroscopic repair of the labrum is crucial to prevent cyst recurrence.
Complications & Management
Despite meticulous surgical technique, complications can occur following suprascapular nerve decompression. A thorough understanding of these potential issues and their management is essential for optimizing patient outcomes.
Common Complications and Salvage Strategies
| Complication | Incidence | Description | Salvage Strategy |
|---|---|---|---|
| Iatrogenic Nerve Injury | Rare (0.5-2%) | Direct trauma, stretch, or thermal injury to the suprascapular nerve or its branches during dissection or ligament release. Leads to new or worsened motor/sensory deficits. | Immediate recognition and intraoperative repair if transection/severe laceration. Post-operative neurological consultation. Nerve graft for gaps. Serial EMG/NCS for monitoring recovery. |
| Incomplete Decompression / Recurrence | 1-5% | Failure to fully release all compressive elements, or reformation of a ganglion cyst (if source not addressed). Persistent or recurrent symptoms. | Re-evaluation with advanced imaging (MRN) and electrodiagnostics. Revision surgery to address remaining compression or cyst recurrence (e.g., labral repair). |
| Hematoma Formation | <1% | Bleeding in the surgical site leading to swelling, pain, and potentially nerve compression. | Meticulous hemostasis intraoperatively. Post-operative drain placement in select cases. Hematoma aspiration or surgical evacuation if symptomatic or large. |
| Infection | <1% | Superficial or deep surgical site infection. Presents with pain, erythema, warmth, fever, purulent drainage. | Broad-spectrum antibiotics based on culture results. Superficial wound care. Surgical debridement and irrigation for deep infections. |
| Shoulder Stiffness | 5-10% (variable) | Limitation of glenohumeral range of motion due to post-operative pain, scar tissue, or inadequate rehabilitation. | Aggressive but protected physical therapy. NSAIDs. Intra-articular steroid injections. Manipulation under anesthesia or arthroscopic capsular release in refractory cases. |
| Persistent Pain | 5-15% | Continued pain despite technically successful decompression. May be due to chronic nerve changes, co-existing shoulder pathology, or neuropathic pain. | Multimodal pain management (medications, nerve blocks). Physical therapy to address residual dysfunction. Re-evaluation for alternative pain generators. Neuropathic pain medications. |
| Rotator Cuff Muscle Weakness/Atrophy | Variable | May persist post-operatively, especially in cases of chronic, severe denervation with fatty atrophy. Recovery can take many months to years. | Ongoing physical therapy focusing on strengthening and motor retraining. Realistic patient expectations. May consider tendon transfers in severe, non-recovering cases. |
| Superior Transverse Scapular Ligament Ossification | Rare | Re-ossification or new bone formation at the site of ligament release, leading to recurrent compression. | Follow-up imaging. Revision surgery for bony decompression. |
General Management Principles
- Prevention: The best management for complications is prevention. This includes meticulous surgical technique, proper patient selection, and thorough pre-operative planning.
- Early Recognition: Vigilance for signs and symptoms of complications in the post-operative period is crucial for timely intervention.
- Multidisciplinary Approach: Management of complex complications, especially those involving nerve injury or chronic pain, often benefits from a multidisciplinary team including neurologists, pain specialists, and physical therapists.
- Patient Education: Setting realistic expectations pre-operatively regarding recovery time, potential for residual symptoms, and the possibility of complications helps manage patient satisfaction and compliance.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following suprascapular nerve decompression is a critical determinant of functional outcome. The protocol is tailored to protect the healing tissues, facilitate nerve recovery, and restore full shoulder function, while recognizing that nerve regeneration is a slow process.
Phase I: Immediate Post-Operative (Weeks 0-2)
Goals:
* Pain and edema control.
* Protect surgical site and nerve.
* Maintain passive range of motion (PROM) within protected limits.
Management:
*
Immobilization:
Sling for comfort and protection (often discontinued after 1-2 weeks, depending on surgical findings and stability of associated repairs, e.g., labral repair).
*
Pain Management:
Oral analgesics, NSAIDs, cryotherapy.
*
PROM:
Gentle, protected passive range of motion of the glenohumeral joint (flexion to 90-120 degrees, external rotation to 30-45 degrees with arm at side, internal rotation to chest). Avoid resisted or active range of motion (AROM) to prevent tension on the healing nerve.
*
Scapular Mobility:
Gentle scapular retraction and protraction exercises.
*
Elbow, Wrist, Hand AROM:
Maintain distal joint mobility.
*
Nerve Glides:
Gentle upper extremity nerve gliding exercises to promote nerve mobility.
Phase II: Early Motion & Muscle Activation (Weeks 2-6)
Goals:
* Gradual increase in PROM.
* Initiate active-assisted range of motion (AAROM) and gentle AROM.
* Introduce isometric strengthening.
* Normalize scapular kinematics.
Management:
*
PROM:
Progress to full PROM as tolerated.
*
AAROM/AROM:
Initiate pendulum exercises, supine active-assisted flexion and external rotation using a stick. Progress to unassisted AROM within pain-free limits.
*
Isometric Strengthening:
Gentle isometric exercises for rotator cuff (supraspinatus and infraspinatus), deltoid, and scapular stabilizers. Avoid direct stress on the suprascapular nerve at this stage.
*
Scapular Stabilization:
Begin basic scapular strengthening (e.g., seated rows, scapular retraction).
*
Neurodynamic Exercises:
Continue progressive nerve glides.
Phase III: Progressive Strengthening (Weeks 6-12)
Goals:
* Restore full, pain-free AROM.
* Progressive strengthening of rotator cuff and scapular musculature.
* Improve proprioception and neuromuscular control.
Management:
*
AROM:
Achieve full AROM in all planes.
*
Resisted Strengthening:
Commence light resistance exercises for rotator cuff (internal/external rotation, abduction), deltoid, and scapular stabilizers using elastic bands or light weights.
*
Eccentric Loading:
Begin eccentric strengthening exercises.
*
Proprioception:
Introduce proprioceptive exercises (e.g., rhythmic stabilization, balance exercises).
*
Closed Chain Exercises:
Wall push-ups, modified planks.
Phase IV: Advanced Strengthening & Return to Activity (Weeks 12+)
Goals:
* Maximize strength, power, and endurance.
* Sport-specific or activity-specific training.
* Gradual return to full function.
Management:
*
Intensified Strengthening:
Progress to heavier resistance exercises, plyometrics (if appropriate).
*
Functional Training:
Incorporate multi-planar movements, overhead activities, and sport-specific drills.
*
Endurance Training:
Cardiorespiratory and muscular endurance.
*
Throwing/Overhead Program:
For athletes, initiate a carefully supervised interval throwing or overhead program, gradually increasing intensity and volume.
*
Return to Activity:
Gradual and monitored return to full occupational or athletic activities, guided by strength, endurance, and pain levels.
Key Considerations:
*
Individualization:
Protocols must be individualized based on patient factors (age, activity level, chronicity of nerve injury), specific surgical findings (associated labral repair, extent of nerve damage), and rate of progress.
*
Neurological Recovery:
Emphasize that nerve regeneration is slow. Motor recovery may lag behind pain relief and can take 6-18 months or longer.
*
Scapular Dyskinesis:
Address any persistent scapular dyskinesis, which can contribute to dynamic nerve irritation.
*
Communication:
Close communication between the surgeon, physical therapist, and patient is vital for successful rehabilitation.
Summary of Key Literature / Guidelines
The body of literature on suprascapular nerve entrapment has grown significantly, highlighting its increasing recognition as a source of shoulder pathology. While definitive, high-level evidence from large randomized controlled trials remains somewhat limited, consensus guidelines and systematic reviews provide valuable insights into diagnosis and management.
Diagnostic Modalities
- Electrodiagnostic Studies (EMG/NCS): Consistently reported as the gold standard for objective confirmation of SSNE. Studies by investigators such as Ringel et al. and Post and Grinblat have detailed typical EMG findings (fibrillations, positive sharp waves in acute denervation; polyphasic potentials in chronic denervation) and NCS abnormalities (prolonged distal motor latency, decreased amplitude). However, EMG can be normal in early or mild cases, and changes may take weeks to months to develop post-injury.
- Magnetic Resonance Imaging (MRI) / Magnetic Resonance Neurography (MRN): Critical for identifying the anatomical cause of compression. Studies by Donovan et al. and Fritz et al. underscore the utility of MRN in visualizing the nerve path, identifying space-occupying lesions (ganglion cysts, paralabral cysts), and assessing muscle changes (edema in acute denervation, fatty atrophy in chronic denervation). Posterior labral tears are frequently implicated as the origin of spinoglenoid cysts (Snyder et al., Fealy et al.).
- Diagnostic Blocks: While not specific for nerve entrapment location, a positive response to a suprascapular nerve block (local anesthetic injection) can confirm the nerve as a pain generator, guiding further diagnostic workup.
Management Strategies
- Conservative Management: Initial non-operative treatment (rest, NSAIDs, physical therapy focusing on scapular stabilization and rotator cuff strengthening, corticosteroid injections) is recommended for most patients, particularly those without clear structural compression or with mild symptoms. Literature supports a trial of conservative care for 3-6 months.
-
Surgical Decompression:
- Indications: Persistent symptoms refractory to conservative management, progressive neurological deficit (weakness, atrophy), or clear anatomical compression (e.g., large ganglion cyst, ossified transverse scapular ligament, tumor).
- Open vs. Arthroscopic: Both approaches have demonstrated efficacy. Arthroscopic decompression is favored for spinoglenoid notch cysts, especially when associated with posterior labral tears (Provencher et al., Plancher et al.). This allows for simultaneous labral repair, addressing the cyst's origin and reducing recurrence. Open approaches are often preferred for suprascapular notch pathology, particularly ossified ligaments or complex anatomy, offering direct visualization. Systematic reviews comparing outcomes generally show good to excellent results with both techniques, with complication rates typically low (Boykin et al.).
- Outcomes: Pain relief is achieved in a high percentage of patients (70-90%), with variable recovery of strength, particularly in long-standing cases with significant fatty atrophy. Younger patients and those with shorter symptom duration tend to have better motor recovery.
Special Populations
- Overhead Athletes: SSNE is common in athletes, with dynamic compression at the spinoglenoid notch being a significant mechanism. Studies emphasize the importance of identifying and addressing associated factors like posterior labral tears and scapular dyskinesis in these individuals (Cahill and Palmer, Wuh et al.).
- Associated Pathology: It is crucial to evaluate for concomitant shoulder pathologies, such as rotator cuff tears, glenohumeral instability, and acromioclavicular joint pathology, as they can mimic or coexist with SSNE. Addressing these concurrently, if appropriate, can improve overall outcomes.
Current Guidelines and Future Directions
While no single universally accepted clinical practice guideline specifically for SSNE exists, the consensus in orthopedic literature leans towards:
1.
High index of suspicion
for SSNE in patients with posterior shoulder pain, especially in athletes.
2.
Early and accurate diagnosis
using electrodiagnostics and advanced imaging (MRN).
3.
Initial trial of conservative management
unless clear and significant structural compression or progressive neurological deficit is present.
4.
Surgical decompression
for failed conservative treatment, progressive neurological decline, or anatomically clear, significant entrapment.
5.
Addressing associated intra-articular pathology
, particularly posterior labral tears.
Future research should focus on prospective, randomized trials comparing surgical techniques, optimal rehabilitation protocols, and long-term outcomes, especially concerning complete motor recovery in chronic cases. Enhanced diagnostic tools for dynamic entrapment and improved nerve regeneration strategies also represent important areas for investigation.
