Introduction & Epidemiology
Periprosthetic joint infection (PJI) following total shoulder arthroplasty (TSA) represents a devastating complication, significantly impacting patient outcomes, increasing healthcare costs, and often necessitating complex, multi-stage revision surgery. While the incidence of primary TSA is rising, so too is the recognition and management of its complications, with PJI being among the most challenging.
The incidence of PJI following primary TSA is estimated to range from 0.5% to 3.5%, though figures can vary based on definition, surveillance methods, and patient population characteristics. For reverse total shoulder arthroplasty (rTSA), some studies suggest a slightly higher incidence, potentially due to the increased complexity, different patient demographics, or greater soft tissue disruption. Risk factors are multifactorial and can be patient-specific (e.g., diabetes mellitus, obesity, rheumatoid arthritis, immunosuppression, active malignancy, prior infection, poor nutritional status), procedure-specific (e.g., prolonged operative time, allogeneic blood transfusion, extensive soft tissue dissection), or related to microbial factors (e.g., specific virulent organisms). Propionibacterium acnes (now Cutibacterium acnes ) has historically been a notable pathogen in shoulder PJI due to its indolent nature and predilection for shoulder skin flora, often presenting with subtle symptoms. However, with improved sterile techniques and pre-operative skin preparation protocols, its relative prevalence may be shifting, though it remains an important consideration. Other common culprits include Staphylococcus aureus (MSSA/MRSA), coagulase-negative staphylococci, and Gram-negative bacilli.
Early diagnosis is paramount for successful management. PJI presents a diagnostic challenge in the shoulder due to its often-subtle presentation compared to lower extremity arthroplasties. Pain is the most common symptom, but it is non-specific and can overlap with mechanical issues, aseptic loosening, or rotator cuff pathology. Other signs may include stiffness, warmth, erythema, swelling, sinus tract formation (pathognomonic but late), or systemic signs such as fever and malaise (more common in acute infections). The distinction between acute PJI (typically within 3 months post-op) and chronic PJI (beyond 3 months) is crucial as it dictates the optimal treatment strategy.
Surgical Anatomy & Biomechanics
Understanding the surgical anatomy and biomechanics relevant to the shoulder is critical, not only for primary TSA but especially for revision procedures related to PJI. While the initial procedure sets the stage, revision for infection often involves compromised tissue planes, bone loss, and scar tissue, necessitating a precise knowledge of critical structures.
The
deltopectoral approach
is the most common for both primary and revision TSA. This internervous plane lies between the deltoid (axillary nerve innervation) laterally and the pectoralis major (medial and lateral pectoral nerve innervation) medially. Key structures at risk during this approach and subsequent revision include:
*
Cephalic vein:
Located in the deltopectoral groove, it is typically ligated or retracted.
*
Axillary nerve:
Courses inferior to the glenohumeral joint, approximately 5-7 cm distal to the acromion, wrapping around the surgical neck of the humerus. It is especially vulnerable during humeral component removal, extensive capsular release, or calcar bone preparation.
*
Musculocutaneous nerve:
Penetrates the coracobrachialis muscle. While less commonly injured directly during standard approaches, extensive dissection around the coracoid or neurolysis can put it at risk.
*
Brachial plexus:
Located deep and medial to the coracoid process, protected by the conjoined tendon. Excessive retraction or medial dissection can compromise it.
*
Subscapularis tendon:
The primary anterior soft tissue restraint, which must be taken down and repaired. In revision for infection, this tendon may be scarred, attenuated, or deficient, impacting post-operative stability and function.
Bone stock assessment is critical in PJI. Chronic infection can lead to osteolysis, necessitating bone grafting or specialized implants. The glenoid is particularly susceptible to bone loss, requiring careful assessment of the glenoid vault. Humeral bone loss can also complicate stem removal and re-implantation.
Biomechanically, the goal of revision is to achieve infection eradication while restoring functional stability and range of motion. In cases of significant rotator cuff deficiency or bone loss often encountered after multiple surgeries for PJI, an rTSA may be indicated, even if the primary was an anatomical TSA. The rTSA alters the center of rotation, leveraging the deltoid muscle to compensate for rotator cuff dysfunction. However, the constrained nature of rTSA can place higher stress on the glenoid, making glenoid loosening a concern, especially in an infected or previously compromised bone bed. The choice between constrained (rTSA) and unconstrained (aTSA) implants must weigh infection control, soft tissue integrity, bone quality, and patient-specific functional demands.
Indications & Contraindications
The management of infected TSA is highly complex and depends on multiple factors, including the acuity of infection, pathogen virulence, host factors, extent of tissue damage, and implant stability. Treatment strategies range from debridement and implant retention (DAIR) to single-stage or two-stage exchange arthroplasty, and in severe cases, salvage procedures.
Indications for Operative Management:
- Acute PJI (<3-6 weeks post-op or acute hematogenous seeding of a well-fixed prosthesis): Often amenable to DAIR with prosthesis retention. Key criteria for DAIR include symptoms <3 weeks, stable implant, no sinus tract, and susceptible organisms.
-
Chronic PJI (>3-6 weeks post-op):
Typically requires implant exchange.
- Single-stage exchange arthroplasty: Indicated for select cases with susceptible organisms, good soft tissue envelope, minimal bone loss, and an experienced surgical team. Requires definitive pathogen identification pre-operatively.
- Two-stage exchange arthroplasty: The gold standard for chronic PJI, especially with unknown organisms, virulent pathogens (e.g., MRSA), extensive soft tissue compromise, significant bone loss, or failed DAIR. Stage 1 involves implant removal, debridement, and antibiotic spacer placement. Stage 2 involves reimplantation after infection eradication is confirmed.
- Failed DAIR or exchange arthroplasty: Often necessitates a two-stage exchange or salvage procedure.
- Sinus tract communication to the joint: Generally a contraindication to DAIR and indicates the need for implant removal.
Contraindications for Operative Management (or specific procedures):
- Host factors: Uncontrolled medical comorbidities, severe malnutrition, immunocompromise, or active infection elsewhere that preclude major surgery.
- Extensive bone loss or irreparable soft tissue damage: May contraindicate reimplantation, leading to consideration of salvage procedures.
- Virulent, multi-drug resistant organisms: May necessitate two-stage exchange or long-term suppressive antibiotics if surgical options are limited.
- Non-compliant patient: May lead to higher failure rates, especially with complex multi-stage procedures and prolonged antibiotic regimens.
- Acute infection with systemic sepsis and hemodynamic instability: Requires initial stabilization before definitive surgical intervention.
Operative vs. Non-Operative Indications in TSA PJI
| Indication Type | Operative Management | Non-Operative Management |
|---|---|---|
| Acute PJI | DAIR (Debridement and Antibiotic Impregnated Retention): < 3-6 weeks symptoms, stable implant, no sinus tract, susceptible organism. | Not typically indicated as primary treatment for acute PJI due to high failure rates without surgical debridement. May be considered for non-surgical candidates if conservative measures can manage the acute inflammatory response while systemic antibiotics are initiated. |
| Chronic PJI |
Two-Stage Exchange Arthroplasty:
Gold standard for chronic PJI, failed DAIR, virulent organisms, significant bone/soft tissue loss, unknown pathogen.
Single-Stage Exchange Arthroplasty: Select cases with known susceptible pathogen, good soft tissues, minimal bone loss, experienced surgeon. Resection Arthroplasty (Girdlestone Shoulder): Failed multiple revisions, severe bone/soft tissue loss, patient unfit for reimplantation. Arthrodesis: Functional option for young, active patients with failed reconstruction but adequate deltoid function. |
Chronic Antimicrobial Suppression:
For non-surgical candidates, frail patients, those refusing surgery, or if surgical risks outweigh benefits. Goals are pain control and prevention of systemic spread, not eradication. Requires long-term oral antibiotics.
Observation: Rarely indicated, only for very indolent, asymptomatic, culture-negative cases in non-surgical candidates, often with a high risk of surgical complications. |
| Sinus Tract | Requires implant removal; typically two-stage exchange. | Contraindication for non-operative management if active infection. |
| Implant Status |
Stable implant:
DAIR possible for acute PJI.
Unstable/Loose implant: Requires implant exchange (one or two-stage). |
Only considered if implant is stable and other surgical options are exhausted or contraindicated. |
| Patient Factors | Fit for surgery: Most definitive treatments require adequate health. | Multiple comorbidities, severe frailty, high surgical risk: Often limits options to long-term suppression or resection arthroplasty. |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is the cornerstone of successful management of infected TSA. This involves a meticulous diagnostic workup, strategic surgical planning, and optimizing patient health.
Diagnostic Workup
- Clinical Assessment: Detailed history (onset of symptoms, prior surgeries, comorbidities, antibiotic use), physical examination (pain, range of motion, warmth, swelling, erythema, sinus tracts).
-
Laboratory Studies:
- Serum markers: C-reactive protein (CRP), erythrocyte sedimentation rate (ESR). While non-specific, persistently elevated or rising values are highly suggestive of infection. Procalcitonin may be useful in acute settings.
- White blood cell count (WBC) with differential.
-
Imaging:
- Plain Radiographs: AP, scapular Y, axillary views. Assess for loosening, osteolysis, component migration, stress shielding, or heterotopic ossification. Baseline films are crucial for comparison.
- CT Scan: Valuable for assessing bone loss, especially glenoid vault integrity, prior hardware, and planning for complex revisions.
- MRI: Limited utility due to artifact from metal prostheses but can be useful for soft tissue assessment if specific questions arise (e.g., rotator cuff integrity in suspected non-infected cases).
- Nuclear Medicine Scans: Technetium-99m (Tc-99m) bone scan and Indium-111 (In-111) labeled leukocyte scan can aid in differentiating infection from aseptic loosening, particularly in equivocal cases. Combined scans are more specific.
- PET Scan: Fluorine-18 fluorodeoxyglucose (FDG-PET) scan is gaining traction as a highly sensitive and specific imaging modality for PJI, especially when other tests are inconclusive.
-
Joint Aspiration:
The most critical diagnostic step. Performed under strict sterile conditions, ideally off antibiotics for at least 2 weeks. Samples are sent for:
- Cell count and differential: WBC count > 3,000 cells/µL with > 80% neutrophils is highly suspicious.
- Gram stain.
- Aerobic and anaerobic cultures: Crucial for pathogen identification and antimicrobial susceptibility testing. Extended culture incubation (up to 14 days) is essential for indolent organisms like Cutibacterium acnes .
- Molecular diagnostics: Alpha-defensin synovial fluid test has high sensitivity and specificity for PJI, offering rapid results. PCR-based assays can detect bacterial DNA but may suffer from false positives due to prior contamination or non-viable organisms.
Patient Optimization
- Medical Comorbidity Management: Aggressive control of diabetes, smoking cessation, optimization of nutritional status.
- Antibiotic Holiday: Aspiration for culture should ideally be performed after a 2-week antibiotic holiday to maximize yield.
Surgical Planning
- Approach Selection: Typically deltopectoral. Extended deltopectoral or posterior approaches may be needed for specific bone loss patterns or prior surgery.
- Implant Selection: Consider revision stem design (e.g., modular, tapered, porous-coated), bone grafting options, and whether anatomical or reverse TSA is indicated for reimplantation.
- Contingency Planning: Always prepare for significant bone loss, difficulty with component removal, and unexpected intraoperative findings. Have various implant sizes, bone graft material, and cement removal tools available.
Patient Positioning
The patient is typically positioned in the
beach chair position
.
*
Head:
Secured in a headrest, ensuring neutral alignment to avoid brachial plexus traction.
*
Torso:
Elevated to 30-60 degrees, allowing for good visualization and access.
*
Arm:
Free-draped, allowing full range of motion intraoperatively for exposure, component removal, and insertion. Adequate padding for pressure points (e.g., sacrum, heels) is essential. The posterior aspect of the arm and axilla should be readily accessible.
*
Instrumentation:
Ensure the image intensifier can be positioned for adequate visualization during stem removal or glenoid preparation.
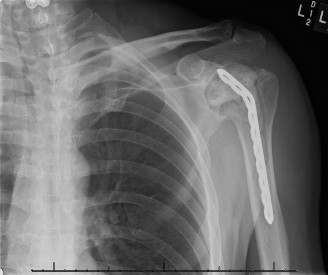
Figure 1: Pre-operative planning image, potentially demonstrating an X-ray or CT reconstruction used to assess bone loss and guide implant selection for revision surgery.
Detailed Surgical Approach / Technique
The surgical technique for managing infected TSA varies significantly based on the chosen strategy (DAIR, single-stage, or two-stage exchange). All approaches emphasize meticulous debridement, extensive tissue sampling, and careful handling of neurovascular structures.
General Principles for PJI Surgery
- Standard Aseptic Technique: Strict adherence to surgical principles.
- Extended Exposure: Often requires longer incisions and more aggressive releases due to scar tissue.
- Comprehensive Debridement: Removal of all non-viable, inflamed, or infected soft tissue. This includes synovium, granulomatous tissue, and any remaining cement mantle (if components are being removed).
- Extensive Irrigation: Pulsatile lavage with copious amounts of saline (e.g., 6-9 liters) is critical to reduce bacterial load.
- Multiple Tissue Cultures: At least 5-6 periprosthetic tissue samples from different sites (e.g., capsule, synovium, bone-implant interface) are sent for aerobic and anaerobic cultures, with extended incubation.
- Biofilm Disruption: For DAIR, aggressive scrubbing of the implant surfaces to disrupt biofilm is essential.
- Neurovascular Protection: Constant awareness of the axillary nerve, brachial plexus, and associated vessels.
Debridement, Antibiotics, and Implant Retention (DAIR)
- Indications: Acute PJI (<3-6 weeks post-op) or acute hematogenous seeding of a well-fixed, non-loose prosthesis, no sinus tract, susceptible organism identified.
-
Technique:
- Standard deltopectoral approach.
- Open the capsule, debride all infected synovial and granulomatous tissue.
- Thoroughly irrigate the joint.
- Aggressively scrub all accessible implant surfaces (humeral head, glenoid component, stem collar) with a sterile brush to disrupt biofilm.
- Exchange the modular components (polyethylene liner if present, humeral head).
- Place a drain.
- Close in layers.
- Administer systemic culture-specific antibiotics for an extended duration (typically 6-12 weeks, possibly longer for C. acnes ).
Two-Stage Exchange Arthroplasty (Gold Standard for Chronic PJI)
Stage 1: Explantation, Debridement, and Spacer Placement
- Approach: Typically deltopectoral, often requiring careful dissection through scarred tissue. Previous surgical scars are usually excised.
- Capsulotomy and Debridement: Extensive debridement of all inflamed and infected soft tissues.
-
Component Removal:
- Humeral Component: Stem removal can be challenging due to bone ingrowth or cement mantle. Use specialized extraction instruments (e.g., osteotomes, trephines, flexible burrs, impactors) to preserve bone stock. Cemented stems require careful cement mantle removal.
- Glenoid Component: Polyethylene liners are removed first. Cemented glenoids require careful debonding and removal to preserve the glenoid vault. Pegged or keeled components may require specific tools.
- Bone Debridement: Burring of sclerotic bone surfaces to create a viable bleeding bed.
- Pulsatile Lavage: Copious irrigation.
-
Antibiotic Spacer Implantation:
- Cement Spacers: Articulating spacers (e.g., molded from antibiotic-loaded bone cement, often containing high doses of vancomycin and tobramycin) are preferred to maintain joint space, deliver local antibiotics, and facilitate later reimplantation. Non-articulating static spacers may be used if joint stability is not a primary concern or in cases of extreme bone loss.
- Vacuum-Assisted Closure (VAC) therapy: May be used in cases with extensive soft tissue defect or concern for persistent wound issues.
- Wound Closure: Primary closure over drains.
- Systemic Antibiotics: Initiated based on pre-operative cultures or empiric broad-spectrum coverage, adjusted once intraoperative culture results are available. Typically administered for 6-12 weeks.
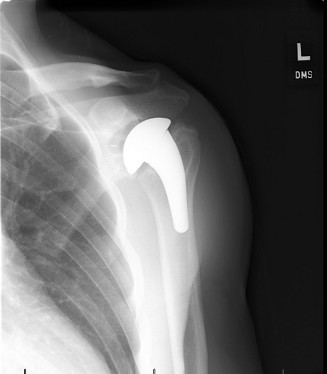
Figure 2: Intraoperative image, likely demonstrating the placement of an antibiotic-impregnated cement spacer in the glenohumeral joint during the first stage of a two-stage exchange for PJI.
Interstage Period:
* Patients remain on systemic antibiotics.
* Serial monitoring of ESR and CRP. These values should trend downwards and ideally normalize.
* A repeat aspiration is often performed once inflammatory markers have normalized and antibiotics have been stopped for 2-4 weeks to confirm infection eradication before proceeding to Stage 2. Negative cultures are mandatory.
Stage 2: Reimplantation
- Approach: Re-exposure through the previous incision. Careful removal of the antibiotic spacer.
- Final Debridement and Irrigation: Removal of any remaining fibrous tissue from the glenoid and humeral beds. Copious lavage.
-
Preparation for Reimplantation:
- Glenoid: Careful preparation, often requiring bone grafting for defects, particularly for rTSA.
- Humerus: Reaming and broaching to fit the definitive humeral component.
-
Component Insertion:
- Glenoid: Cemented or uncemented components based on bone quality and surgeon preference.
- Humeral: Cemented or uncemented stems.
- Humeral Head/Polyethylene Insert: Assemble the final components.
- Capsular Repair: Repair of the subscapularis tendon (if applicable) and capsule to restore stability.
- Wound Closure: Over drains.
- Post-operative Antibiotics: Often continued for a period, or for chronic suppression depending on residual risk.
Single-Stage Exchange Arthroplasty
- Indications: Highly selected cases with known, susceptible, low-virulence organisms, good soft tissue envelope, minimal bone loss, and an experienced surgeon.
-
Technique:
Combines the explantation, debridement, and reimplantation into one procedure.
- Perform extensive debridement, component removal, and copious irrigation as in Stage 1.
- Multiple intraoperative cultures taken.
- Proceed directly to definitive component implantation.
- Intravenous antibiotics administered intraoperatively and post-operatively, based on pre-operative cultures and adjusted as needed.
- Caution: Higher failure rates if patient or infection characteristics are not ideally matched to this strategy.
Complications & Management
Management of PJI is fraught with potential complications beyond persistent infection. These can significantly affect function and patient quality of life.
Common Complications of TSA PJI Treatment
| Complication | Incidence (approximate) | Salvage Strategies |
|---|---|---|
| Persistent/Recurrent Infection | 10-30% after DAIR, 5-15% after 2-stage exchange | Repeat DAIR (if criteria met), conversion to 2-stage exchange, repeat 2-stage exchange, resection arthroplasty, arthrodesis, suppressive antibiotics. |
| Aseptic Loosening/Instability | Variable, higher in revision settings | Revision arthroplasty (if bone stock adequate), conversion to rTSA, glenoid/humeral bone grafting, resection arthroplasty, arthrodesis. |
| Neurovascular Injury | <1% (axillary nerve most common) | Axillary Nerve: Observation, nerve repair/grafting if transected. Brachial Plexus: Observation, neurolysis, rarely nerve transfer. Vascular: Repair. |
| Deltoid Dysfunction/Detachment | Variable, higher with extensive approaches/revisions | Deltoid repair (if possible), observation for functional recovery, latissimus dorsi transfer (rarely), conversion to rTSA (to leverage deltoid). |
| Rotator Cuff Failure | Variable, often pre-existing or exacerbated by surgery | Repair (if reconstructible), superior capsular reconstruction, latissimus dorsi transfer, conversion to rTSA. |
| Periprosthetic Fracture | 1-5% | ORIF with plate/screws, revision stem (longer or locked), allograft-prosthesis composite. |
| Heterotopic Ossification (HO) | Up to 50% radiographically, 5-10% symptomatic | Prophylaxis (NSAIDs, radiation post-op), surgical excision for symptomatic HO (after skeletal maturity). |
| Wound Complications | Hematoma, seroma, dehiscence, necrosis | Drainage, debridement, secondary closure, skin grafting, flap reconstruction. |
| Stiffness/Reduced Range of Motion | Common after infection due to fibrosis | Aggressive physical therapy, manipulation under anesthesia (if not infectious etiology), capsular release (rarely, risks recurrence). |
| Antibiotic-Related Complications | Clostridioides difficile infection, renal/hepatic toxicity, allergic reactions | Discontinuation of offending antibiotic, alternative antibiotics, supportive care (e.g., fluid management, anti-diarrheals). |
| Glenoid Bone Loss | Common after multiple revisions or chronic infection | Glenoid bone grafting (autograft/allograft), custom implants, revision to rTSA with augmented glenoid component, resection arthroplasty. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following infected TSA treatment is complex and must be tailored to the specific surgical procedure, quality of soft tissues, and patient factors. The overarching goal is to balance infection eradication, soft tissue healing, and the restoration of function, often in a compromised environment. Unlike primary TSA, rehabilitation protocols after PJI treatment are often more conservative and protracted.
General Principles
- Protection: Prioritize protection of surgical repairs (e.g., subscapularis, deltoid) and healing tissues, especially after extensive debridement or bone grafting.
- Pain Management: Effective pain control is crucial for patient participation in therapy.
- Patient Education: Patients must understand the prolonged nature of recovery and the importance of compliance with activity restrictions and antibiotic regimens.
- Slow Progression: Activities are typically advanced more slowly than after primary arthroplasty due to compromised soft tissues and the risk of recurrent infection or mechanical failure.
- Infection Control: Continuation of antibiotics for an extended period is paramount.
Rehabilitation After DAIR (Debridement and Antibiotic Retention)
-
Initial Phase (0-6 weeks):
- Immobilization: Shoulder immobilized in a sling for 2-4 weeks to protect soft tissue healing, particularly if capsular or rotator cuff repair was performed.
- Passive Range of Motion (PROM): Gentle pendulum exercises and Codman's exercises may begin early. Therapist-assisted PROM, limiting external rotation and elevation, initiated carefully.
- Elbow/Wrist/Hand ROM: Active exercises to prevent stiffness.
- Weight-bearing: No weight-bearing through the affected arm.
-
Intermediate Phase (6-12 weeks):
- Active-Assistive ROM (AAROM): Progress to AAROM as pain decreases and soft tissue healing allows.
- Active ROM (AROM): Gradually introduce AROM.
- Light Strengthening: Isometric exercises for rotator cuff and periscapular muscles may begin, avoiding high-load activities.
-
Advanced Phase (3-6 months+):
- Progressive Strengthening: Increase resistance and complexity of exercises.
- Functional Activities: Gradual return to activities of daily living.
- Return to Sport: Generally discouraged for high-impact or overhead sports.
Rehabilitation After Two-Stage Exchange Arthroplasty
Stage 1 (After explantation and spacer placement):
*
Immobilization:
Shoulder typically immobilized in a sling for comfort and to protect the wound.
*
Early Motion (if articulating spacer):
Gentle pendulum exercises, elbow/wrist/hand ROM. Limited PROM for the shoulder to prevent stiffness and maintain joint space for Stage 2. The goal is primarily comfort and hygiene, not aggressive motion.
*
Non-weight-bearing:
No weight-bearing through the affected limb.
*
Strict Adherence to Antibiotics:
Critical during the interstage period.
Stage 2 (After reimplantation of definitive prosthesis):
*
Initial Phase (0-6 weeks):
*
Immobilization:
Sling use for 4-6 weeks, sometimes longer, depending on soft tissue repair (e.g., subscapularis repair often dictates sling use for 6 weeks, with external rotation precautions).
*
PROM:
Gentle pendulum exercises. Therapist-assisted PROM initiated cautiously, respecting soft tissue repairs. Limits for external rotation, elevation, and extension are often more conservative than in primary TSA.
*
Elbow/Wrist/Hand ROM:
Active exercises.
*
Intermediate Phase (6-12 weeks):
*
AAROM/AROM:
Progress gradually based on pain and soft tissue healing.
*
Light Isometrics:
Introduce carefully.
*
Advanced Phase (3-6 months+):
*
Progressive Strengthening:
Increase resistance slowly.
*
Functional Training:
Work towards restoring functional independence.
*
Return to Activity:
Very gradual, often with permanent restrictions on heavy lifting or overhead activities.
Rehabilitation After One-Stage Exchange Arthroplasty
- Rehabilitation protocols generally follow those for Stage 2 of a two-stage exchange or primary TSA, but often with a more cautious progression due to the inherent complexity and higher risk profile. Protection of soft tissue repairs and adherence to antibiotic regimens remain critical.
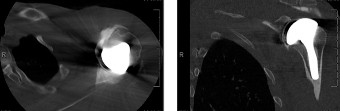
Figure 3: Post-operative image, possibly a plain radiograph showing the definitive implant in place after successful two-stage revision for TSA PJI, with good component alignment.
Summary of Key Literature / Guidelines
The management of periprosthetic shoulder infection relies on a multidisciplinary approach, guided by consensus statements and evolving evidence. Key guidelines and literature inform current best practices.
-
International Consensus Meeting (ICM) on Periprosthetic Joint Infection (2013, 2018, 2023): These consensus meetings provide the most comprehensive and widely accepted diagnostic criteria and treatment algorithms for PJI, including specific recommendations for the shoulder. Key takeaways include:
- Diagnostic Criteria: Emphasizing a combination of clinical signs, serum inflammatory markers (ESR, CRP), synovial fluid analysis (WBC count, %PMN, alpha-defensin), and multiple intraoperative cultures. A positive sinus tract communicating with the joint is diagnostic.
- Management Algorithms: Clear flowcharts guiding treatment decisions based on acuity (acute vs. chronic), pathogen characteristics, host factors, and implant stability.
- Role of Cutibacterium acnes : Specific recommendations for extended culture incubation and prolonged antibiotic courses for C. acnes infections, which are often indolent and challenging to eradicate.
- Antibiotic Regimens: Guidance on empiric and definitive antibiotic choices, duration, and the role of biofilm-active agents (e.g., rifampin for staphylococcal infections).
-
Multisociety Consensus (MSIS) Criteria: Similar to ICM, the Musculoskeletal Infection Society (MSIS) also offers diagnostic criteria that are largely consistent, further strengthening the framework for diagnosis.
-
Literature on DAIR Success Rates: Studies consistently show that DAIR has the highest success rates for acute PJI with early diagnosis, sensitive organisms, and a healthy host. Success rates significantly decrease with chronic infections, virulent pathogens, or the presence of a sinus tract. A critical component is the exchange of modular components (humeral head, polyethylene liner) to reduce biofilm burden.
-
Two-Stage Exchange as Gold Standard: Numerous studies validate the two-stage exchange as the most reliable method for eradicating chronic shoulder PJI, especially with challenging organisms or extensive bone and soft tissue compromise. Success rates typically range from 80-95%. The choice and content of antibiotic-loaded cement spacers (e.g., high-dose vancomycin and tobramycin) are well-established.
-
Single-Stage Exchange Efficacy: While potentially attractive due to fewer surgeries, single-stage exchange for chronic shoulder PJI is generally reserved for highly selected cases. The literature suggests comparable success rates to two-stage exchange in these specific patient populations , but careful patient selection and expert surgical technique are paramount.
-
Salvage Procedures: Arthrodesis and resection arthroplasty remain viable options for failed multiple revisions, severely compromised soft tissues, or patients unfit for further reconstruction. Literature highlights the trade-offs in function and pain relief with these procedures.
-
Future Directions: Ongoing research focuses on improving diagnostic tools (e.g., advanced molecular diagnostics, specific biomarkers), novel antimicrobial delivery systems (e.g., coated implants, biodegradable carriers), and optimizing antibiotic regimens (e.g., personalized pharmacokinetics). The role of probiotics and microbiome modulation is also an area of emerging interest.
In summary, the management of infected TSA is an evolving field, demanding a rigorous diagnostic workup, individualized treatment planning, meticulous surgical execution, prolonged antimicrobial therapy, and dedicated rehabilitation. Adherence to current consensus guidelines and a high index of suspicion are crucial for optimizing outcomes in this challenging patient population.
