Introduction & Epidemiology
Rheumatoid arthritis (RA) is a chronic, systemic inflammatory autoimmune disease primarily affecting synovial joints, leading to progressive cartilage destruction, bone erosion, and joint deformity. The glenohumeral joint is frequently involved, with estimates suggesting clinically significant shoulder pain in 60-90% of RA patients over the disease course. Initial manifestations often include pain, stiffness, and restricted range of motion, evolving into rotator cuff pathology, glenoid erosion, humeral head collapse, and eventual arthropathy.
A critical consideration in the management of RA patients, particularly those with glenohumeral involvement, is their heightened susceptibility to septic arthritis. The incidence of septic arthritis in RA patients is significantly higher than in the general population, ranging from 0.5% to 5.0%, with some reports indicating up to 10% in specific cohorts. This increased risk is multifactorial, attributed to chronic synovitis, corticosteroid use, immunosuppressive disease-modifying anti-rheumatic drugs (DMARDs) and biologics, compromised skin integrity, and repeated joint injections. The diagnosis of septic arthritis in RA is notoriously challenging due to overlapping symptoms with RA flares, crystal-induced arthropathy, and chronic pain, often leading to delayed diagnosis and potentially devastating outcomes including rapid joint destruction and systemic sepsis. Early recognition and aggressive management are paramount to preserve joint function and patient life. This review will delineate critical diagnostic and management considerations for glenohumeral RA, with a particular focus on when and how to suspect and manage concomitant septic arthritis.
Surgical Anatomy & Biomechanics
Understanding the surgical anatomy of the glenohumeral joint, particularly within the context of RA pathology, is crucial for both diagnostic aspiration and surgical intervention. The shoulder joint is a diarthrodial ball-and-socket joint comprising the humeral head and the glenoid fossa of the scapula.
Bony Anatomy
- Humeral Head: Typically hemispherical, but in RA, it can undergo significant erosion, subchondral cyst formation, or osteonecrosis. Superior migration due to rotator cuff insufficiency is common.
- Glenoid: The glenoid fossa is typically pear-shaped, shallow, and oriented anterolaterally. RA causes progressive glenoid erosion, often eccentric (posterior or superior), and can lead to biconcave glenoid morphology or severe bone loss, making prosthetic implantation challenging.
- Acromion: Impingement and erosion can occur, contributing to rotator cuff tears.
Soft Tissue Anatomy
- Joint Capsule: Thickened and inflamed in RA, often with hypertrophic synovium (pannus) that invades and destroys cartilage and bone. The capsule is capacious, allowing significant joint fluid accumulation, which can mask effusion size in RA.
- Rotator Cuff: Highly susceptible to degeneration, attritional tears, and rupture in RA, even without significant trauma. The supraspinatus, infraspinatus, teres minor, and subscapularis contribute to dynamic stability. Tears compromise abduction and external rotation, leading to superior migration of the humeral head and "cuff tear arthropathy" pattern, which necessitates reverse total shoulder arthroplasty (rTSA) considerations.
- Biceps Tendon: The long head of the biceps tendon, intra-articular and often inflamed (tenosynovitis) in RA, can rupture, leading to a "Popeye" deformity.
- Deltoid Muscle: Critical for shoulder function, especially in rTSA where it becomes the primary abductor. Preserving deltoid integrity and innervation (axillary nerve) is paramount.
- Neurovascular Structures: The axillary nerve wraps posteriorly around the surgical neck of the humerus, typically 5-7 cm distal to the acromion, and is at risk during inferior capsular release or humeral osteotomy. The brachial plexus and axillary artery/vein are situated anteriorly and medially, requiring careful retraction.
Biomechanical Considerations in RA
RA fundamentally alters glenohumeral biomechanics:
*
Instability:
Progressive capsular laxity, rotator cuff tears, and bone loss contribute to superior or posterior subluxation.
*
Pain & Stiffness:
Synovial inflammation, effusion, and capsular contracture limit range of motion.
*
Muscle Imbalance:
Rotator cuff dysfunction leads to altered force couples, further contributing to humeral head migration and glenoid erosion patterns.
*
Infection Response:
In the context of septic arthritis, the inflamed RA synovium becomes a fertile ground for bacterial proliferation. The effusion, often present due to RA activity, can mask the inflammatory response specific to infection. The compromised blood supply to the pannus and cartilage can hinder antibiotic penetration.
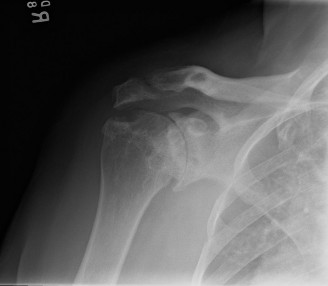
(This image, ideally, would depict the glenohumeral joint with typical RA erosions, or perhaps a diagram illustrating key neurovascular structures and surgical approaches to the shoulder.)
Indications & Contraindications
The decision-making process for intervention in glenohumeral RA is complex, weighing patient symptoms, disease activity, functional limitations, and crucially, the presence or suspicion of infection.
Operative vs. Non-Operative Indications in Glenohumeral RA
| Category | Non-Operative Indications | Operative Indications |
|---|---|---|
| Rheumatoid Arthritis (Uncomplicated) | Initial management of pain and stiffness, early disease, mild to moderate glenohumeral involvement, stable rotator cuff, adequate response to medical therapy (DMARDs, biologics, NSAIDs, intra-articular corticosteroids), patient preference against surgery. | Persistent disabling pain refractory to conservative management, significant functional impairment (e.g., inability to perform ADLs), progressive joint destruction (significant cartilage loss, erosion, osteonecrosis), severe rotator cuff deficiency with pseudoparalysis, intractable instability. |
| Suspected/Confirmed Septic Arthritis | Absolute contraindication for elective arthroplasty. Initial broad-spectrum IV antibiotics (after aspiration). Rarely, if patient is too unstable for surgery, non-operative management with antibiotics and close monitoring may be considered, but carries high risk of morbidity/mortality. | URGENT SURGICAL INTERVENTION IS MANDATORY. Diagnostic aspiration and joint fluid analysis. Therapeutic surgical irrigation and debridement (I&D) with synovectomy. In specific cases, arthroscopic lavage may be considered. Open I&D for failed arthroscopic treatment or extensive infection. |
| Post-Septic Arthritis (Chronic Sequelae) | Stable, quiescent infection; residual pain/stiffness after successful infection eradication, managed medically. | Persistent pain, functional loss, or instability secondary to joint destruction after complete eradication of infection (typically 6-12 months post-infection treatment), consideration for delayed arthroplasty (TSA or rTSA) if appropriate. |
Specific Operative Indications for Glenohumeral RA:
- Total Shoulder Arthroplasty (TSA): Indicated for severe glenohumeral arthropathy with an intact or reparable rotator cuff, intractable pain, and significant functional limitation.
- Hemiarthroplasty: Less commonly performed for RA alone, but may be considered for severe humeral head collapse/osteonecrosis with a relatively preserved glenoid, or as a salvage procedure.
- Reverse Total Shoulder Arthroplasty (rTSA): The preferred option for RA patients with severe glenohumeral arthropathy and irreparable rotator cuff tears (cuff tear arthropathy) leading to pseudoparalysis and superior humeral head migration. Also useful in cases of severe glenoid bone loss where primary TSA glenoid component fixation is compromised.
- Synovectomy: Rarely performed in isolation for RA shoulders due to high recurrence rates, but may be an adjunct during other procedures or for refractory synovitis in early disease without significant cartilage loss.
- Arthrodesis: A salvage procedure for failed arthroplasty or severe infection with extensive bone loss where reconstruction is not feasible, offering pain relief at the expense of motion.
Absolute Contraindications for Elective Glenohumeral Arthroplasty:
- Active Septic Arthritis: This is the most critical contraindication addressed by this topic. Arthroplasty in the presence of infection carries an unacceptably high risk of prosthetic joint infection, requiring removal of components.
- Uncontrolled Systemic Infection: Any active infection distant from the shoulder.
- Rapidly Progressive Neuropathic Arthropathy: (e.g., Charcot joint).
- Inadequate Deltoid Muscle Function: For rTSA.
- Severe Psychiatric Illness or Unrealistic Patient Expectations.
- Insufficient Bone Stock: For secure prosthetic fixation (though autograft/allograft may be considered).
Relative Contraindications:
- Severe debilitating comorbidities precluding safe anesthesia.
- Poor skin condition or chronic skin infections in the surgical field.
- Uncontrolled inflammatory disease activity.
- History of previous prosthetic joint infection (requires careful evaluation and often a staged approach).
- Non-compliance with post-operative rehabilitation.
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential for successful outcomes in glenohumeral RA surgery, particularly when septic arthritis is a differential diagnosis.
Diagnostic Workup for Suspected Septic Arthritis:
When a RA patient presents with acute worsening of shoulder pain, swelling, erythema, warmth, or systemic symptoms (fever, chills, malaise), septic arthritis must be aggressively ruled out.
1.
Clinical Assessment:
Detailed history focusing on duration, onset, severity, constitutional symptoms, recent injections, or skin lesions. Physical examination for signs of inflammation, effusion, and pain with passive motion.
2.
Laboratory Studies:
*
CBC with differential:
Leukocytosis is common but not always present, especially in immunosuppressed RA patients.
*
Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP):
Elevated in RA flares, but significantly higher levels or a disproportionate rise should raise suspicion for infection. Trend monitoring is critical.
*
Blood Cultures:
Obtain at least two sets if systemic symptoms are present.
*
Procalcitonin:
May be useful to differentiate bacterial infection from inflammatory flare, though its utility in RA is still being defined.
3.
Joint Aspiration:
This is the
gold standard
for diagnosing septic arthritis.
*
Technique:
Performed under sterile conditions, often with ultrasound guidance, using a large-bore needle (e.g., 18-gauge) to obtain adequate synovial fluid. Common approaches include the posterior interval between the deltoid and infraspinatus, or the anterior approach lateral to the coracoid process.
*
Fluid Analysis:
*
Cell Count with Differential:
WBC count >50,000 cells/µL (or >100,000 cells/µL in RA) with >75% neutrophils is highly suggestive. However, in RA, pre-existing inflammation can elevate WBCs, so a significant
change
from baseline or a disproportionately high neutrophil count is more indicative.
*
Gram Stain:
Rapid preliminary identification of bacteria. Sensitivity is variable (20-75%).
*
Culture and Sensitivity:
Definitive diagnosis and guides antibiotic therapy. Extended culture for atypical organisms may be needed.
*
Glucose and Protein:
Low glucose (<50% serum glucose) and high protein (>4 g/dL) are supportive but non-specific.
*
Crystal Analysis:
To rule out concomitant crystal arthropathy (e.g., gout, pseudogout), which can mimic septic arthritis.
4.
Imaging:
*
Plain Radiographs:
May show soft tissue swelling, periarticular osteopenia, or erosions, but are not diagnostic for early infection. Baseline RA changes are expected.
*
Ultrasound:
Can visualize joint effusion, synovitis, and guide aspiration.
*
MRI:
Useful for evaluating soft tissue involvement, bone edema, osteomyelitis, and abscess formation. Gadolinium enhancement can delineate synovial inflammation and fluid collections.
*
CT Scan:
Provides excellent bony detail, useful for assessing bone loss, erosions, and guiding aspirations in complex cases.
Pre-Operative Planning for Arthroplasty (Elective Cases, Infection Ruled Out):
- Imaging: Standard AP, true AP (Grashey), axillary lateral, and scapular Y views. CT scan is often indispensable for assessing glenoid bone stock, version, and wear patterns, especially for RA patients with complex anatomy. MRI may be used for rotator cuff evaluation.
- Medical Optimization: Close collaboration with a rheumatologist to optimize RA disease control and manage immunosuppression. Corticosteroids may need adjustment. Pre-operative assessment by an internist or anesthesiologist to manage comorbidities.
- Pre-operative Antibiotics: Administered within 60 minutes prior to incision, typically a first or second-generation cephalosporin.
- Blood Management: Type and cross-match for potential transfusion, especially in complex revision cases.
Patient Positioning:
- Beach Chair Position: The most common position for glenohumeral arthroplasty, offering excellent exposure for both anterior and superior approaches, allows for easier conversion to other procedures (e.g., rotator cuff repair), and simplifies visualization of landmarks. The patient is semi-recumbent, with the torso elevated, head secured, and arm draped free. Care must be taken to prevent brachial plexus traction injuries and compartment syndrome in the contralateral limb.
- Lateral Decubitus Position: An alternative, less frequently used for primary arthroplasty, but may be preferred for some arthroscopic procedures or specific revision cases. Requires careful padding and stabilization.
Detailed Surgical Approach / Technique
The surgical approach for glenohumeral RA depends heavily on the primary goal: managing septic arthritis or performing reconstructive arthroplasty after infection is ruled out.
A. Management of Septic Arthritis (Urgent Surgical Intervention)
The primary goal is to drain pus, debride infected tissue, and obtain tissue for culture.
-
Joint Aspiration (Diagnostic/Therapeutic):
- As described above, often performed in the clinic or ER. If the fluid is purulent, aspiration can be therapeutic in the short term. However, aspiration alone is generally insufficient for definitive management of septic arthritis in the shoulder due to its complex anatomy and potential for loculated infection.
- Technique (Ultrasound-Guided): Identify the posterior glenohumeral joint line. Introduce an 18-gauge needle through the posterior soft tissues, aiming towards the glenoid. Aspirate fluid. If aspirate is clear but suspicion remains high, inject saline, then re-aspirate ("washout").
-
Surgical Irrigation & Debridement (I&D) and Synovectomy:
- Indications: Confirmed or highly suspected septic arthritis, especially with purulent effusion, failure of aspiration/antibiotics, or evidence of significant synovitis/fibrin.
-
Approach:
Typically an anterior deltopectoral approach is preferred for wide exposure, though arthroscopic lavage can be considered for less severe cases.
-
Anterior Deltopectoral Approach:
- Incision: A longitudinal incision from the coracoid process extending distally for 8-10 cm along the deltopectoral groove.
- Internervous Plane: Between the deltoid (axillary nerve) and pectoralis major (medial and lateral pectoral nerves). Identify and preserve the cephalic vein, retracting it laterally with the deltoid.
- Subdeltoid Space: Incise the clavipectoral fascia. Retract the conjoined tendon (coracobrachialis and short head of biceps) medially.
- Subscapularis Management: The subscapularis tendon provides anterior access to the joint. It can be released by detaching it from the lesser tuberosity, usually with a small cuff of bone (lesser tuberosity osteotomy) for easier repair, or by splitting the tendon. In acute infection, precise repair may be secondary to thorough debridement.
- Joint Exposure: Incise the anterior capsule. Thoroughly irrigate the joint with copious amounts of saline (e.g., 6-9 liters). Debride all infected, necrotic, and inflammatory synovial tissue (synovectomy). Remove any fibrin clots or loose bodies. Obtain multiple tissue samples for Gram stain, culture, and histopathology.
- Closure: Close the capsule loosely or leave open depending on the severity of infection and the need for continued drainage. Repair the subscapularis tendon. Close deep fascia, subcutaneous tissue, and skin, often leaving a drain in place.
-
Anterior Deltopectoral Approach:
- Arthroscopic Lavage: Can be performed for early, uncomplicated septic arthritis without extensive synovitis or loculations. Provides excellent visualization and allows for thorough irrigation and debridement of accessible areas. Multiple portals (posterior, anterosuperior, anteroinferior) are used. However, open debridement offers better access for complete synovectomy and removal of deeper foci, which is often preferred in complex RA shoulders with advanced disease.
B. Glenohumeral Arthroplasty (Elective, Infection Ruled Out)
The choice between TSA and rTSA is dictated by rotator cuff integrity and glenoid bone stock.
1. Total Shoulder Arthroplasty (TSA)
- Approach: Deltopectoral approach (as described for I&D).
- Internervous Plane: Deltoid (axillary nerve) and pectoralis major (pectoral nerves).
-
Exposure:
- Release the subscapularis tendon (either tenotomy or lesser tuberosity osteotomy).
- Perform capsular release (inferior, superior, posterior) to gain full mobilization of the humeral head and ensure adequate external rotation post-op.
-
Humeral Preparation:
- Osteotomy of the humeral head: Guided by pre-operative templating, typically 30-45 degrees of retroversion and 135 degrees of inclination.
- Reaming and broaching of the humeral canal to prepare for the humeral stem (cemented or uncemented).
- Trial components inserted to assess fit and range of motion.
-
Glenoid Preparation:
- Exposure of the glenoid requires careful retraction of the humeral head.
- Resection of any osteophytes.
- Reaming of the glenoid surface to remove articular cartilage and create a bleeding subchondral bone bed.
- Preparation of anchor holes for the glenoid component, ensuring appropriate version and inclination. Glenoid bone loss in RA can be significant, requiring bone grafting or specialized components.
- Cementation of the polyethylene glenoid component.
-
Reduction & Closure:
- Insert definitive humeral head prosthesis onto the stem.
- Reduce the joint.
- Repair the subscapularis tendon securely to the lesser tuberosity or its anatomical footprint.
- Close layers, typically with a drain.
2. Reverse Total Shoulder Arthroplasty (rTSA)
- Approach: Deltopectoral approach is standard. The deltoid muscle becomes crucial for function, so its integrity and innervation must be protected.
- Exposure: Similar initial steps to TSA, including subscapularis release and careful capsular releases.
-
Glenoid Preparation:
- The glenoid is prepared for the glenosphere , which is the convex articulating surface.
- Peripheral osteophytes are removed.
- A central reamer prepares the glenoid vault.
- A baseplate is secured to the glenoid, typically with multiple cancellous screws, aiming for stable fixation and optimal inferior positioning to maximize deltoid tension and minimize impingement. This often requires careful management of significant glenoid bone loss in RA.
- The glenosphere is then secured to the baseplate.
-
Humeral Preparation:
- Humeral head osteotomy is performed, often at a less retroverted angle (e.g., 0-20 degrees) compared to TSA, or even slightly anteverted.
- Reaming and broaching of the humeral canal for the stem.
- A polyethylene liner (humeral cup) is then placed onto the humeral stem.
-
Reduction & Closure:
- Reduce the humeral component onto the glenosphere.
- Assess stability and range of motion.
- The subscapularis is typically not repaired in rTSA as it is often deficient and its repair can limit external rotation and increase dislocation risk. However, some surgeons advocate for partial repair if tissue quality allows and if stability is a concern.
- Close layers, usually with a drain.
Complications & Management
Complications in glenohumeral RA surgery are diverse, encompassing general surgical risks, procedure-specific issues, and unique challenges related to RA and potential infection.
Table of Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (%) | Salvage Strategy | Multi-disciplinary team (rheumatologist, orthopedist, infectious disease specialist). Medical management of symptoms; use of appropriate antibiotics (oral or IV) based on culture and sensitivity. Repeat aspiration to monitor response. Serial physical exams. | Arthrodesis (salvage for painful, unstable, unconstructable joint). Resection arthroplasty (for intractable infection/bone loss where reconstruction is impossible). Staged arthroplasty (excision arthroplasty, prolonged antibiotic course, then delayed prosthetic implantation). |
Detailed Complications Specific to RA and Septic Arthritis:
1. Septic Arthritis in RA Patients (A Critical Diagnostic & Management Challenge)
- Challenges: Delayed diagnosis due to masked symptoms (pre-existing inflammation, immunosuppression), polyarticular involvement.
- Pathogens: Staphylococcus aureus is most common. Gram-negative organisms, anaerobes, and fungi can occur, especially in immunocompromised patients.
-
Management:
- Urgent Joint Aspiration: As discussed, for diagnosis.
- Empiric Antibiotics: Broad-spectrum intravenous antibiotics immediately after obtaining cultures (e.g., vancomycin plus a third-generation cephalosporin or piperacillin-tazobactam).
- Surgical I&D: Prompt surgical irrigation and debridement is crucial. Delayed intervention significantly increases morbidity and mortality. This involves arthroscopic lavage or open debridement and synovectomy.
- Duration of Antibiotics: Typically 4-6 weeks (IV initially, then oral based on sensitivities and clinical response).
- Rheumatoid Medications: Often temporarily held, especially biologics and strong immunosuppressants, after discussion with rheumatology. Corticosteroids may be continued or tapered cautiously if patient is steroid-dependent.
2. Periprosthetic Joint Infection (PJI) (Post-Arthroplasty)
- Incidence: Higher in RA patients (reported 2-5%) compared to OA patients due to immunosuppression and compromised skin.
- Diagnosis: Joint aspiration is key (cell count, culture). Elevated ESR/CRP post-op are non-specific but concerning.
-
Management:
- Acute PJI (<3-4 weeks post-op): Debridement, antibiotics, and implant retention (DAIR) may be attempted if the components are well-fixed and there are no signs of extensive infection or necrosis.
-
Chronic PJI (>4 weeks post-op) or Failed DAIR:
Two-stage revision arthroplasty is generally preferred.
- Stage 1: Removal of all prosthetic components, thorough debridement and synovectomy, placement of an antibiotic-loaded cement spacer, and 6-12 weeks of targeted IV antibiotics.
- Stage 2: Re-implantation of new prosthetic components after infection markers (ESR/CRP) have normalized and interval aspiration is negative.
- Salvage: Resection arthroplasty or shoulder arthrodesis if re-implantation is not feasible or fails.
3. Rotator Cuff Failure (Post-TSA)
- Incidence: Higher in RA due to underlying disease, poor tissue quality.
- Management: Non-operative (physical therapy, pain management). Revision surgery with rotator cuff repair (if possible) or conversion to rTSA if irreparable cuff tear and persistent pain/dysfunction.
4. Glenoid Loosening (Post-TSA)
- Incidence: Common in RA due to poor bone quality and eccentric wear patterns.
- Management: Non-operative for asymptomatic loosening. Revision arthroplasty (glenoid component revision, often to rTSA if significant bone loss or cuff deficiency).
5. Humeral Loosening/Fracture
- Management: Revision of humeral component, often with grafting. Humeral periprosthetic fractures require open reduction and internal fixation (ORIF) or revision arthroplasty depending on fracture pattern and stability.
6. Instability/Dislocation (Post-Arthroplasty, especially rTSA)
- Incidence: Higher in rTSA (2-10%).
- Management: Closed reduction for acute dislocations. Non-operative management with bracing and therapy for recurrent instability if mild. Revision surgery for persistent instability (e.g., component malposition, soft tissue imbalance).
7. Neurological Injury
- Axillary Nerve: Most commonly injured, leading to deltoid paresis/paralysis.
- Brachial Plexus: Less common but more severe.
- Management: Observation for neuropraxia. Nerve exploration and repair for complete lesions.
8. Scapular Notching (Post-rTSA)
- Incidence: Common (up to 70% radiographic incidence), but not always symptomatic. Inferior impingement of the humeral cup on the inferior scapular neck.
- Management: Symptomatic notching may require revision of the glenosphere position (inferior offset) or humeral cup design.
9. Heterotopic Ossification (HO)
- Incidence: Variable, often asymptomatic.
- Management: Prophylaxis (NSAIDs, radiation therapy) in high-risk patients. Excision if symptomatic and mature.
Post-Operative Rehabilitation Protocols
Rehabilitation following glenohumeral RA surgery or treatment for septic arthritis is tailored to the specific procedure, tissue quality, and patient comorbidities. A multi-disciplinary approach involving the surgeon, physical therapist, and rheumatologist is essential.
A. Post-Septic Arthritis Treatment (I&D)
- Goals: Restore range of motion, prevent adhesions, preserve muscle strength, and ensure complete infection eradication.
-
Initial Phase (0-2 weeks post-I&D):
- Immobilization in a sling for comfort and initial tissue healing.
- Passive Range of Motion (PROM): Gentle, pain-free PROM (pendulum exercises, supine passive flexion/external rotation) to prevent stiffness and maintain joint lubrication, initiated early (often within 24-48 hours) as tolerated after drain removal.
- Pain Management: Aggressive pain control.
- Infection Monitoring: Close monitoring of inflammatory markers, wound healing, and systemic signs of infection.
-
Intermediate Phase (2-6 weeks):
- Progress to Active-Assisted Range of Motion (AAROM) and then Active Range of Motion (AROM) as pain allows.
- Isometrics for rotator cuff and deltoid, progressing to light resistance.
- Gradual discontinuation of sling use.
-
Advanced Phase (6+ weeks):
- Strengthening exercises using resistance bands, light weights.
- Focus on functional activities and return to daily living.
- Proprioceptive training.
- Caution: Avoid excessive stress on healing joint surfaces if there was significant cartilage damage.
B. Post-Total Shoulder Arthroplasty (TSA)
- Goals: Protect subscapularis repair, achieve functional range of motion, restore strength, reduce pain.
-
Phase I: Protection & Early Motion (0-6 weeks):
- Immobilization: Sling continuously for 4-6 weeks, removed only for exercises and hygiene.
-
PROM:
Initiated early (day 1-3) with strict limitations.
- Forward flexion: 90-120 degrees.
- External rotation: Neutral to 30 degrees (to protect subscapularis).
- Internal rotation: To chest.
- No Active Internal Rotation/Subscapularis Loading: For 6 weeks to allow subscapularis repair to heal.
- Hand, wrist, elbow AROM, pendulum exercises.
-
Phase II: Progressive Motion & Light Strengthening (6-12 weeks):
- Sling discontinued.
- Progress to AAROM and then AROM.
- Gentle isometric exercises for deltoid and rotator cuff (except internal rotators initially).
- Gradual increase in external rotation.
-
Phase III: Strengthening & Functional Return (12+ weeks):
- Progressive strengthening of rotator cuff and deltoid.
- Advanced stretching.
- Proprioceptive exercises.
- Return to light activities of daily living. Avoid heavy lifting, impact sports, or extreme ranges of motion indefinitely.
C. Post-Reverse Total Shoulder Arthroplasty (rTSA)
- Goals: Optimize deltoid function, prevent instability/dislocation, achieve functional elevation and abduction. Subscapularis repair is often not a limiting factor.
-
Phase I: Protection & Early Motion (0-6 weeks):
- Immobilization: Sling for 4-6 weeks, similar to TSA but less strict regarding subscapularis protection.
-
PROM:
Initiated early (day 1-3).
- Forward flexion: 90-120 degrees.
- Abduction: 90-120 degrees.
- External rotation: As tolerated, typically 0-30 degrees (less concern for subscapularis).
- Internal rotation: To chest.
- Avoid: Combined adduction, internal rotation, and extension (risk of dislocation). Extreme external rotation (risk of impingement/dislocation).
- Hand, wrist, elbow AROM.
-
Phase II: Progressive Motion & Strengthening (6-12 weeks):
- Sling discontinued.
- Progress to AAROM and then AROM.
- Gentle isometric exercises for the deltoid and periscapular muscles.
- Focus on deltoid strengthening.
-
Phase III: Advanced Strengthening & Functional Return (12+ weeks):
- Progressive strengthening of the deltoid and periscapular muscles.
- Proprioceptive training.
- Return to light activities. Avoid heavy lifting (>10-15 lbs), repetitive overhead activities, or positions of extreme internal rotation/adduction.
General Considerations for RA Patients in Rehabilitation:
- Pain Tolerance & Flare-ups: RA patients may have lower pain tolerance and systemic flare-ups can impede rehab progress.
- Bone & Soft Tissue Quality: Fragile bone, poor soft tissue quality, and chronic inflammation may necessitate a slower, more cautious progression.
- Polyarticular Involvement: Other joint pain or limitations can affect adherence and ability to perform exercises.
- Immunosuppression: Increased risk of infection post-op, requiring meticulous wound care.
- Education: Extensive patient education regarding activity modifications and lifelong precautions is crucial.
Summary of Key Literature / Guidelines
The literature on glenohumeral RA and its intersection with septic arthritis underscores the complexity and critical nature of this clinical scenario.
-
Diagnosis of Septic Arthritis in RA:
- Multiple studies highlight the diagnostic challenge, often emphasizing that classic signs (fever, high leukocytosis) may be blunted in immunosuppressed RA patients.
- Mandatory Joint Aspiration: Consensus is that joint aspiration with synovial fluid analysis (cell count, differential, Gram stain, culture) is the sine qua non for diagnosis. ESR and CRP, while non-specific, are helpful adjuncts, particularly for monitoring trends.
- Delayed Diagnosis: Studies consistently demonstrate that delayed diagnosis leads to worse outcomes, including increased morbidity, mortality, and joint destruction.
-
Management of Septic Arthritis:
- Urgent I&D: The cornerstone of treatment is urgent surgical irrigation and debridement in conjunction with targeted intravenous antibiotics. The debate between arthroscopic and open debridement persists, with open debridement often preferred for extensive infection or in compromised joints due to RA.
- Antibiotic Duration: Guidelines generally recommend 4-6 weeks of antibiotic therapy, with initial parenteral administration.
- Immunosuppression: Management of DMARDs and biologics during active infection requires careful multidisciplinary consideration. Most guidelines suggest temporarily discontinuing biologics and potentially other immunosuppressants, weighing the risk of uncontrolled RA flare against immune suppression.
-
Glenohumeral Arthroplasty in RA:
- Outcomes: Both TSA and rTSA demonstrate reliable pain relief and functional improvement in selected RA patients. However, outcomes are generally less predictable than in osteoarthritis, with higher rates of complications, particularly glenoid loosening in TSA and PJI.
- Reverse Shoulder Arthroplasty (rTSA): Has emerged as the preferred option for RA patients with cuff-deficient arthropathy due to its superior pain relief and functional outcomes in this population. It also provides a more stable construct in the face of compromised soft tissues.
- Glenoid Bone Loss: A significant challenge in RA. Pre-operative CT planning is critical. Techniques such as augmented glenoid components, bone grafting, or superior placement of the rTSA glenosphere are employed to manage severe glenoid erosion.
- Periprosthetic Joint Infection (PJI): RA is a recognized risk factor for PJI. Prevention strategies (e.g., pre-operative patient optimization, careful skin preparation, perioperative antibiotics) are paramount. Management typically follows a staged revision protocol.
-
Timing of Arthroplasty Post-Septic Arthritis:
- Current recommendations advocate for a minimum of 6-12 months between the successful eradication of septic arthritis and elective arthroplasty. This allows for complete resolution of inflammation and infection, confirmed by normalized inflammatory markers and negative joint aspirates. Performing arthroplasty too early significantly increases the risk of recurrent PJI.
In conclusion, managing glenohumeral rheumatoid arthritis requires a comprehensive understanding of disease progression, surgical techniques, and rehabilitative principles. The constant vigilance for and aggressive management of septic arthritis remain a critical differentiator and a core competency for any orthopedic surgeon treating this complex patient population.

