Revision ACL Reconstruction: An Intraoperative Masterclass for Failed Primary Repair
Key Takeaway
This masterclass guides fellows through revision ACL reconstruction, focusing on identifying primary failure causes, meticulous preoperative planning, and precise intraoperative technique. We cover comprehensive anatomy, step-by-step execution including notchplasty and tunnel management, graft preparation, and critical pearls for avoiding pitfalls. Postoperative rehabilitation and complication strategies are also detailed for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The management of a failed primary Anterior Cruciate Ligament (ACL) reconstruction represents one of the most formidable challenges in modern orthopedic sports medicine. As the volume of primary ACL reconstructions continues to rise globally, so too does the absolute number of failures. Revision ACL reconstruction is not merely a repetition of the primary procedure; it is a complex, salvage-oriented intervention that demands meticulous attention to detail, a profound understanding of knee biomechanics, and a strategic, often staged, surgical approach. Our goal in the operative theater is to restore functional stability, prevent further intra-articular degeneration, and return the patient to their desired level of activity, all while navigating compromised bone stock, altered anatomy, and the psychological burden of a recurrent injury.
Understanding the etiology of the primary failure is the cornerstone of successful revision surgery. The natural history of an ACL-deficient knee, particularly after a failed reconstruction, is characterized by progressive intra-articular damage. Persistent anterior and rotational instability places the articular cartilage and menisci at significant risk for irreversible degeneration. We systematically categorize primary graft failures into three distinct, yet occasionally overlapping, etiologies: biologic failure of graft incorporation, technical surgical error, and traumatic re-rupture. Biologic failure occurs when the graft fails to undergo the necessary phases of necrosis, revascularization, and ligamentization, often due to poor host biology, unrecognized low-grade infection, or deleterious inflammatory responses.
Suboptimal surgical technique remains the most common preventable cause of primary ACL reconstruction failure, accounting for the majority of early non-traumatic failures. Tunnel malposition—specifically, a femoral tunnel placed too anteriorly or too vertically—is the most frequent culprit. An anteriorly placed femoral tunnel leads to graft tension in flexion, resulting in graft stretch-out, impingement against the intercondylar roof, or catastrophic failure. Conversely, a vertical graft fails to control rotational kinematics, leaving the patient with a persistent pivot shift despite a seemingly intact graft on sagittal imaging. Other technical errors include inadequate graft fixation, failure to address concomitant ligamentous laxity, and improper tensioning.
Traumatic re-rupture involves a distinct, high-energy injury to a previously intact and incorporated graft. While unavoidable in some high-risk athletic populations, the threshold for re-rupture may be lowered by unrecognized subtle technical errors or failure to address secondary stabilizers. The Multicenter ACL Revision Study (MARS) cohort has provided invaluable epidemiological data, demonstrating that revision ACL reconstructions inherently carry inferior clinical outcomes, lower return-to-sport rates, and higher subsequent failure rates compared to primary procedures. Consequently, the revision surgeon must approach these cases with a comprehensive, algorithmic mindset, leaving no variable unexamined.
Detailed Surgical Anatomy and Biomechanics
In the revision setting, the surgeon's anatomical understanding must extend far beyond the isolated native footprint of the ACL. The knee is a complex, synergistic organ, and failure to appreciate the interplay between the central pivot and the peripheral capsuloligamentous structures will invariably lead to recurrent failure.
The Anterior Cruciate Ligament Bundles
The native ACL is a highly sophisticated, multi-fascicular structure primarily composed of two functional bundles: the anteromedial (AM) bundle and the posterolateral (PL) bundle, named for their tibial insertion sites. The AM bundle is primarily responsible for resisting anterior tibial translation at higher degrees of knee flexion (taut in flexion), while the PL bundle is the primary restraint to anterior translation and rotatory loads near extension. In a revision scenario, the native footprints are often obliterated by previous tunnel drilling and subsequent osteolysis. Identifying the lateral intercondylar ridge (resident's ridge) and the lateral bifurcate ridge on the medial wall of the lateral femoral condyle is paramount, as these osseous landmarks dictate the anatomic center of the native ACL footprint. Failure to restore this anatomic footprint is a primary driver of altered kinematics and subsequent graft failure.
Secondary Stabilizers and the Posterolateral Corner
A critical tenet of revision ACL surgery is that the ACL does not function in isolation. The secondary stabilizers of the knee provide crucial resistance to anterior translation and rotational forces, and their incompetence will rapidly overload a newly reconstructed ACL graft. The Medial Collateral Ligament (MCL) is the primary valgus stabilizer, while the posterior horn of the medial meniscus acts as a vital wedge, providing a secondary restraint to anterior tibial translation. Loss of the medial meniscus, particularly the posterior root, exponentially increases the in situ forces on the ACL graft.
Furthermore, unrecognized injury to the posterolateral corner (PLC) is a notorious cause of primary ACL failure. The PLC, comprising the popliteus tendon, the popliteofibular ligament, and the Lateral Collateral Ligament (LCL), resists varus stress, external tibial rotation, and posterior translation. Chronic PLC deficiency leads to a varus thrust gait, placing immense cyclical strain on the central pivot. Any high-grade injury to these structures must be addressed concurrently with the revision ACL reconstruction, either through repair, advancement, or formal reconstruction.
Neurovascular Considerations
Revision surgery inherently involves navigating scar tissue and distorted anatomy, heightening the risk of iatrogenic neurovascular injury. The popliteal artery and tibial nerve reside in the popliteal fossa, immediately posterior to the posterior capsule. During tibial tunnel drilling, posterior capsular release, or aggressive posterior notchplasty, the surgeon must maintain strict depth control and utilize protective retractors. The common peroneal nerve is uniquely vulnerable laterally, particularly during dissection for lateral extra-articular tenodesis (LET), LCL reconstruction, or hardware removal from the lateral femoral condyle. Meticulous surgical technique, precise portal placement, and an intimate knowledge of these neurovascular relationships are non-negotiable requirements for the revision surgeon.
Exhaustive Indications and Contraindications
The decision to proceed with revision ACL reconstruction is highly individualized, weighing the patient's symptoms, functional demands, and the structural integrity of the joint. The primary indication remains symptomatic, recurrent instability that severely limits the patient's activities of daily living or prevents return to desired athletic pursuits.
However, the surgeon must carefully differentiate between instability and pain. Patients presenting primarily with anterior knee pain, stiffness, or advanced compartmental osteoarthritis in the setting of a failed ACL are generally poor candidates for isolated revision reconstruction. In such cases, the pain is typically driven by chondral wear or meniscal deficiency, and restoring stability will not alleviate these symptoms; in fact, it may exacerbate them by altering joint contact pressures.
| Category | Specific Clinical Scenarios | Rationale / Clinical Impact |
|---|---|---|
| Absolute Indications | Symptomatic anterior/rotational instability | Recurrent giving-way episodes during ADLs or sports. |
| Concomitant repairable meniscal tears | Stabilizing the knee is required to protect the meniscal repair. | |
| Multi-ligamentous knee injury | ACL reconstruction is required as part of a global joint stabilization strategy. | |
| Relative Indications | Asymptomatic instability in a young athlete | Prophylactic stabilization to prevent future chondral/meniscal damage, though controversial. |
| Failed ACL with varus/valgus malalignment | Requires concurrent or staged osteotomy (e.g., HTO or DFO) prior to or alongside revision ACLR. | |
| Absolute Contraindications | Active intra-articular infection | Eradication of infection with hardware removal and antibiotics is mandatory prior to any reconstruction. |
| Advanced, symptomatic osteoarthritis | Revision ACLR will not cure arthritic pain; arthroplasty or non-operative management is preferred. | |
| Complex Regional Pain Syndrome (CRPS) | Surgical trauma will severely exacerbate the neuropathic pain cycle. | |
| Voluntary instability / Psychiatric overlay | High risk of non-compliance and deliberate graft disruption. |
When evaluating a patient for revision, the surgeon must also consider the status of the previous hardware and bone tunnels. If significant osteolysis or tunnel widening (>14mm) is present, a two-stage approach is indicated. The first stage involves hardware removal and bone grafting of the tunnels, followed by a maturation period of 4 to 6 months before the definitive second-stage reconstruction. Attempting a single-stage revision in the face of massive bone loss compromises graft fixation and invites catastrophic failure.
Pre-Operative Planning, Templating, and Patient Positioning
Effective preoperative planning is the blueprint for success in revision ACL surgery. The complexity of these cases demands that the surgeon anticipates potential pitfalls and formulates primary, secondary, and tertiary surgical plans before the patient ever enters the operating room.
Clinical Evaluation and History
The clinical evaluation begins with a meticulous history, dissecting the primary injury, the initial reconstruction, and the subsequent failure. The surgeon must ascertain the mechanism of the primary injury and the exact details of the primary reconstruction. Obtaining the previous operative report is absolutely critical. This document reveals the graft type utilized (autograft vs. allograft; bone-patellar tendon-bone, hamstring, or quadriceps), the fixation methods (interference screws, suspensory buttons, staples), and the status of the menisci and articular cartilage at the time of the index procedure.
The physical examination must be comprehensive. Gait analysis is performed to detect an antalgic gait, a varus thrust (indicating PLC incompetence), or quadriceps avoidance. Instability testing includes the Lachman test, the Anterior Drawer test, and the Pivot Shift test, which is the most specific indicator of rotatory instability. The surgeon must also rigorously evaluate the secondary stabilizers: varus/valgus stress testing at 0 and 30 degrees, the Dial test at 30 and 90 degrees for PLC/PCL integrity, and the varus recurvatum test.
Advanced Imaging and Diagnostic Modalities
Imaging is paramount. Routine weight-bearing anteroposterior (AP), lateral, and skyline patellar radiographs are the first step. These films allow the surgeon to assess the mechanical axis, identify existing hardware, evaluate for osteoarthritis, and critically analyze the previous tunnel positions.

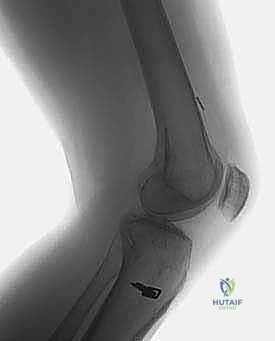
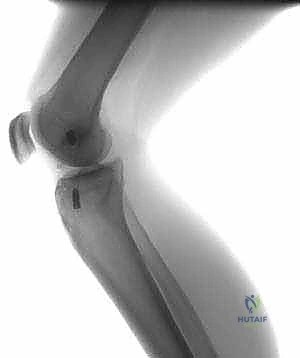
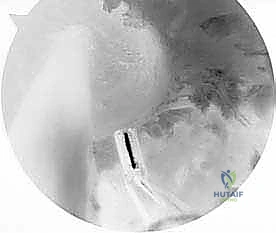
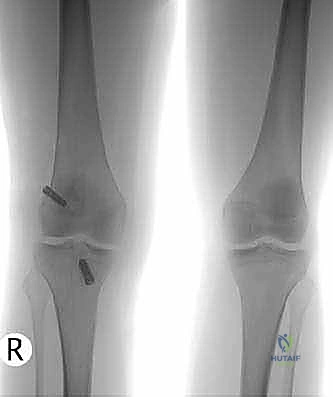
The images above demonstrate a primary ACL reconstruction utilizing a suspensory EndoButton on the femur and staple fixation on the tibia. Understanding the exact location and type of this hardware is essential for planning removal and avoiding interference with new tunnel trajectories.
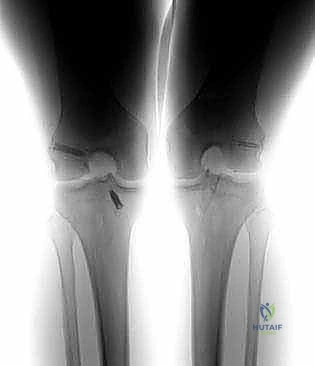
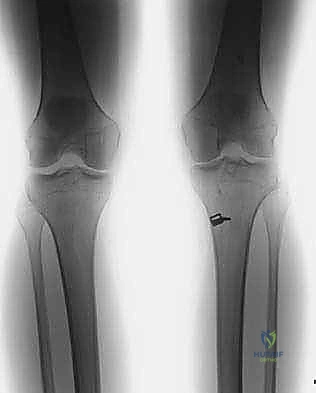
Crucially, the lateral radiograph must be scrutinized for femoral tunnel position. As seen in the images above, an anteriorly placed femoral tunnel (anterior to Blumensaat's line) is a classic technical error leading to graft impingement, stretching, and ultimate failure.
In addition to plain films, Computed Tomography (CT) with 3D reconstruction is the gold standard for evaluating bone loss and tunnel widening. CT provides precise measurements of the existing tunnels, dictating whether a one-stage or two-stage revision is required. Magnetic Resonance Imaging (MRI) is utilized to assess the status of the menisci, articular cartilage, and concomitant ligamentous injuries, though metallic artifact from previous hardware can occasionally obscure the intra-articular details.
Patient Positioning and Examination Under Anesthesia
In the operating theater, the patient is placed in the supine position. A lateral post is utilized, positioned proximal enough on the thigh to allow unencumbered drilling of the tibial tunnel when the knee is flexed. The knee is flexed to 90 degrees over a bump, allowing the neurovascular structures in the popliteal fossa to fall posteriorly. A high-thigh tourniquet is applied but inflated only if visualization becomes compromised.
Before prepping and draping, a rigorous Examination Under Anesthesia (EUA) is mandatory. The EUA provides a true assessment of knee laxity, free from patient guarding. The surgeon must confirm the findings of the awake examination, paying particular attention to the pivot shift grade and any subtle asymmetric opening to varus or valgus stress.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a revision ACL reconstruction is an exercise in precision, adaptability, and meticulous tissue handling. The procedure can be broadly divided into diagnostic arthroscopy, hardware removal, tunnel preparation, and graft fixation.
Diagnostic Arthroscopy and Hardware Removal
Standard anterolateral (AL) and anteromedial (AM) portals are established. A thorough diagnostic arthroscopy is performed, systematically evaluating all compartments. Meniscal pathology and chondral lesions are addressed first. The intercondylar notch is then cleared of scar tissue (cyclops lesions) and the remnants of the failed graft.
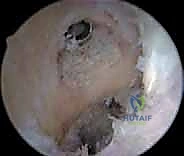
Hardware removal can be one of the most frustrating aspects of revision surgery. Interference screws, particularly bioabsorbable or biocomposite screws that have only partially degraded, must be meticulously cored out or removed with specialized extraction instrumentation.
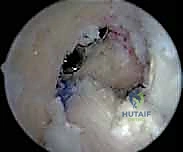
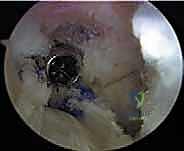
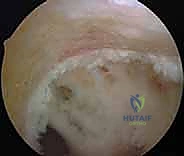
Suspensory cortical buttons (e.g., EndoButtons) on the lateral femoral cortex can often be left in situ if they do not interfere with the new tunnel trajectory. However, if removal is necessary, it typically requires a small lateral incision, splitting the iliotibial band, and dissecting down to the lateral cortex to retrieve the button.
One-Stage Versus Two-Stage Revision Strategies
The decision between a one-stage and two-stage revision is dictated by the degree of tunnel widening and the trajectory of the previous tunnels. If the previous tunnels are anatomic but widened (>14mm), or if they are non-anatomic but intersect the planned anatomic trajectory in a way that compromises fixation, a two-stage approach is mandatory. The first stage involves removing all hardware, debriding the fibrous tissue from the tunnels, and packing the defects with bone graft (autologous iliac crest or allograft dowels). The patient returns 4-6 months later for the definitive reconstruction once CT confirms osseous integration. If the tunnels are <14mm and divergent from the planned anatomic trajectory, a one-stage revision can safely be performed.
Tunnel Preparation and Notchplasty
Achieving anatomic tunnel placement is the most critical step in restoring normal knee kinematics. The femoral tunnel is typically drilled through an accessory anteromedial portal to allow for independent, anatomic placement on the lateral femoral condyle footprint, avoiding the constraints of transtibial drilling.
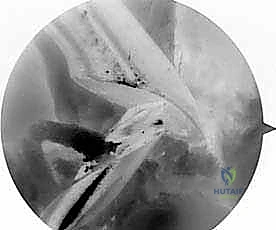
The surgeon must carefully assess the relationship between the old tunnel and the planned new tunnel. If the new tunnel overlaps the old tunnel, the surgeon may need to utilize a larger diameter graft or employ specialized techniques such as stacked interference screws or a suspensory button with a larger cortical footprint to achieve rigid fixation. A targeted notchplasty may be required to prevent graft impingement, particularly if there are significant osteophytes, but aggressive notchplasty should be avoided as it alters the anatomic landmarks and lateralizes the femoral footprint.
Graft Selection, Passage, and Rigid Fixation
Graft selection in the revision setting is complex. If the patient's native tissues allow, autograft (bone-patellar tendon-bone, quadriceps tendon, or contralateral hamstring) remains the gold standard, particularly in young, high-demand athletes. However, allografts are frequently utilized in revision scenarios to minimize donor site morbidity, especially when multiple ligaments require reconstruction or when autograft options have been exhausted.
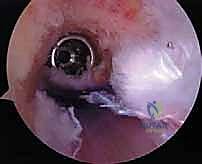
Graft passage is performed under direct arthroscopic visualization. Fixation must be absolutely rigid. In the setting of compromised bone stock, hybrid fixation is highly recommended. For the femur, a suspensory cortical button is often combined with an aperture interference screw. On the tibia, an interference screw is typically backed up with a cortical staple or a suture button. The graft is tensioned and fixed with the knee in 20-30 degrees of flexion, applying a posterior drawer force to reduce the tibia.
In many revision cases, particularly those with a high-grade pivot shift or generalized hyperlaxity, the surgeon should strongly consider augmenting the intra-articular reconstruction with a Lateral Extra-articular Tenodesis (LET) or Anterolateral Ligament (ALL) reconstruction. This provides a critical secondary restraint to internal rotation, protecting the central graft during the vulnerable early healing phases.
Complications, Incidence Rates, and Salvage Management
Revision ACL reconstruction is fraught with potential complications, and the surgeon must be prepared to manage them aggressively. The overall complication rate is significantly higher than in primary procedures, reflecting the compromised biological and mechanical environment of the multiply operated knee.
| Complication | Estimated Incidence | Prevention and Salvage Management Strategy |
|---|---|---|
| Arthrofibrosis / Stiffness | 5 - 10% | Prevention: Aggressive early ROM, particularly extension. Control post-op hemarthrosis. Management: Lysis of adhesions, manipulation under anesthesia (MUA), aggressive physical therapy. |
| Infection (Septic Arthritis) | 0.5 - 2% | Prevention: Strict sterile technique, prophylactic antibiotics, careful handling of allografts. Management: Emergent arthroscopic I&D, copious lavage, hardware retention if graft is viable, targeted IV antibiotics. |
| Recurrent Instability / Re-rupture | 10 - 25% | Prevention: Anatomic tunnel placement, rigid fixation, addressing secondary stabilizers (LET/ALL), strict adherence to rehab. Management: Re-evaluation for technical errors, consideration of osteotomy for malalignment, potential 3rd-time revision (salvage). |
| Hardware Failure / Migration | 1 - 3% | Prevention: Appropriate sizing of screws, bi-cortical purchase for suspensory devices, backup fixation in poor bone stock. Management: Hardware removal, revision of fixation if graft is compromised. |
| Neurovascular Injury | < 1% | Prevention: Meticulous dissection, retractors during posterior work, avoiding over-penetration of guide pins. Management: Immediate vascular surgery consultation for arterial injury; observation and AFO for neuropraxia. |
Arthrofibrosis, particularly loss of terminal extension, is a devastating complication. It is often driven by prolonged immobilization, excessive surgical trauma, or unrecognized cyclops lesions. Prevention relies on achieving full hyperextension intraoperatively and instituting immediate postoperative range of motion protocols.
Infection, though rare, is a surgical emergency. The use of allografts slightly increases this risk. Any patient presenting with disproportionate pain, swelling, erythema, and elevated inflammatory markers (CRP, ESR) must undergo immediate joint aspiration. If septic arthritis is confirmed, emergent arthroscopic irrigation and debridement are mandatory. The graft can often be retained if the infection is caught early and the hardware remains secure, coupled with a prolonged course of culture-directed intravenous antibiotics.
Phased Post-Operative Rehabilitation Protocols
Rehabilitation following revision ACL reconstruction is generally more conservative and protracted than after a primary procedure. The biological incorporation of the graft, particularly in the setting of widened tunnels or allograft usage, is delayed. The rehabilitation protocol must balance the need to protect the healing graft with the imperative to prevent arthrofibrosis.
Phase I: Protection and Early Range of Motion (Weeks 0-4)
The primary goals in the immediate postoperative phase are to control inflammation, restore full passive knee extension, and activate the quadriceps musculature. Weight-bearing status is often modified based on concomitant procedures (e.g., meniscal repair, cartilage restoration). The patient is typically placed in a hinged knee brace locked in extension for ambulation. Patellar mobilization is critical to prevent infrapatellar contracture. Continuous Passive Motion (CPM) machines may be utilized, but active-assisted range of motion is prioritized.
Phase II: Strengthening and Proprioception (Weeks 4-12)
As the graft begins its initial phases of necrosis and early revascularization, it is at its mechanically weakest point. The focus shifts to progressive closed kinetic chain exercises (e.g., leg presses, mini-squats) to protect the graft from excessive shear forces. Open kinetic chain hamstring strengthening is initiated, but open kinetic chain quadriceps extension is typically restricted or limited to a safe arc of motion (90 to 40 degrees) to prevent anterior tibial translation. Neuromuscular control and proprioceptive training (balance boards, single-leg stance) are heavily emphasized.
Phase III: Return to Sport and Advanced Plyometrics (Months 3-12+)
The transition to Phase III is contingent upon the resolution of all effusions, full range of motion, and significant recovery of quadriceps strength (typically >80% of the contralateral limb). Straight-line jogging is gradually introduced, followed by agility drills, plyometrics, and sport-specific cutting maneuvers. Return to competitive play is rarely permitted before 9 to 12 months postoperatively, and in many revision cases, a delay to 12-18 months is advocated to ensure complete ligamentization and biological maturation of the graft. Functional bracing may be utilized upon return to sport, primarily for psychological support and proprioceptive feedback.
Summary of Landmark Literature and Clinical Guidelines
The evidence base guiding revision ACL reconstruction has expanded significantly over the past decade, largely driven by large-scale prospective registries. The Multicenter ACL Revision Study (MARS) group has been instrumental in defining the epidemiology and outcomes of this challenging procedure.
The MARS cohort has definitively shown that clinical outcomes (IKDC, KOOS, Marx activity scales) are significantly lower following revision ACLR compared to primary reconstruction. Furthermore, the MARS data highlights that the use of autograft in the revision setting, when available, yields superior patient-reported outcomes and lower re-rupture rates compared to allograft, reinforcing the preference for autogenous tissue in young, active patients.
Recent literature has also heavily focused on the role of extra-articular augmentation. The STABILITY study group, while initially focused on high-risk primary ACLs, has provided compelling evidence that the addition of a Lateral Extra-articular Tenodesis (LET) significantly reduces the risk of graft rupture and persistent rotatory laxity. This concept has been widely extrapolated to the revision setting. Current clinical guidelines strongly advocate for the routine consideration of LET or ALL reconstruction in revision cases, particularly those with a high-grade pivot shift, chronic instability, or generalized ligamentous laxity, to offload the central intra-articular graft and optimize the biomechanical environment for successful healing.
In conclusion, revision ACL reconstruction is a demanding surgical endeavor that requires a masterclass level of preparation, anatomical knowledge, and technical execution. By rigorously analyzing the etiology of failure, respecting the secondary stabilizers, executing precise anatomic tunnel placement, and adhering to evidence-based rehabilitation, the orthopedic surgeon can successfully navigate this complex landscape and restore function to the multiply operated knee.
