Patient Presentation & History
A 58-year-old male presented to the orthopedic oncology clinic with an 18-month history of progressively worsening sacral pain. The pain was initially insidious, dull, and localized to the gluteal region, but over the past 6 months, it had become constant, more intense, and radiated bilaterally into the posterior thighs, indicative of S1-S2 radiculopathy. He reported exacerbated pain with prolonged sitting and standing, and minimal relief with NSAIDs or acetaminophen. He denied any history of trauma. Of note, the patient had recently developed new onset constipation and experienced occasional urinary hesitancy, suggesting potential S3-S4 neurological involvement. He reported a 5 kg unintentional weight loss over the past year, attributing it to decreased appetite due to chronic pain. Past medical history was significant for well-controlled hypertension and dyslipidemia. He was a non-smoker and consumed alcohol occasionally. No family history of malignancies was reported.
Clinical Examination
On general inspection, the patient appeared in moderate discomfort, adopting an antalgic gait. No obvious skin changes, masses, or asymmetry were noted in the lumbosacral region.
Palpation of the sacrum elicited significant tenderness, particularly over the midline and bilateral sacroiliac joints. A deep, firm, non-mobile mass was vaguely palpable anterior to the sacrum on rectal examination, confirming the suspicion of a presacral component.
Range of motion of the lumbar spine was full but painful at the extremes of flexion and extension. Hip range of motion was unrestricted. Neurological examination revealed:
*
Motor:
Bilateral lower extremity strength was intact (5/5) for hip flexion, knee extension, ankle dorsiflexion, and great toe extension. However, ankle plantarflexion was subtly weakened (4+/5) bilaterally, consistent with S1 radiculopathy.
*
Sensory:
Light touch and pinprick sensation were diminished in the bilateral S1 and S2 dermatomes. Perianal sensation (S3-S5) was also reduced bilaterally, correlating with the patient's reports of altered bowel/bladder function.
*
Reflexes:
Patellar reflexes (L3-L4) were 2+ and symmetrical. Achilles reflexes (S1) were diminished (1+) bilaterally.
*
Sphincter Tone:
Rectal tone was reduced, and the bulbocavernosus reflex was absent, correlating with the patient's urinary hesitancy and constipation. The examination findings collectively pointed towards a space-occupying lesion affecting the distal sacral nerve roots.
Imaging & Diagnostics
Initial diagnostic workup commenced with standard plain radiographs of the lumbosacral spine and pelvis.
-
Plain Radiographs: Anteroposterior and lateral views of the sacrum demonstrated a lytic, expansile lesion centered in the S2-S4 segments of the sacrum. Cortical destruction was evident, particularly on the lateral view, with an associated soft tissue mass projecting anteriorly. Calcification within the lesion was absent, which, while not pathognomonic, is a less common finding in chordomas compared to some other sacral lesions.
-
Computed Tomography (CT) Scan: A CT scan of the pelvis and lumbosacral spine was subsequently performed to better delineate the bony architecture and tumor extent. The CT revealed a destructive, lytic lesion involving S2 to S4, with significant cortical breach and an extensive anterior sacral soft tissue component. Areas of internal septation and mucoid matrix were noted. There was no evidence of overt calcification. The CT scan provided crucial information regarding the extent of bony involvement, potential for pathological fracture, and proximity to critical structures such as the rectum and major pelvic vessels. It was instrumental for surgical planning, particularly in identifying the optimal approach and osteotomy levels.
- Templating: Based on the CT images, a 3D reconstruction was performed to precisely template the planned sacrectomy. This allowed for visualization of potential osteotomy lines to achieve a wide margin and assess the feasibility of preserving critical sacral nerve roots (S2 if possible, S3-S4 typically sacrificed in these resections).
-
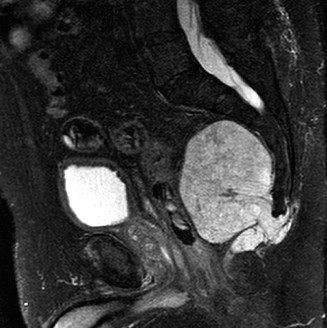
Magnetic Resonance Imaging (MRI): MRI of the lumbosacral spine and pelvis with and without gadolinium contrast was performed to assess the soft tissue extent, intraspinal involvement, and neurovascular relationships.
- T1-weighted images: Showed a heterogeneous, relatively hypointense mass replacing the normal sacral marrow, with areas of intermediate signal intensity. The anterior soft tissue component was clearly visualized.
- T2-weighted images: Demonstrated a characteristically hyperintense, lobulated mass, often described as a "soap bubble" appearance, due to the high mucin content. Intralesional septations were also visible. There was clear evidence of tumor extension into the sacral canal, compressing the thecal sac and nerve roots.
-
Post-contrast T1-weighted images:
Revealed heterogeneous enhancement of the solid components of the tumor. Crucially, the MRI provided definitive evidence of nerve root involvement and the superior extent of the tumor relative to the S1 foramina, which guided the level of sacrectomy.
-
Biopsy: Given the clinical and radiological suspicion of a primary sacral malignancy, a CT-guided core needle biopsy was performed. This was considered the definitive diagnostic step, performed by an experienced interventional radiologist. Multiple cores were obtained from different areas of the lesion.
-
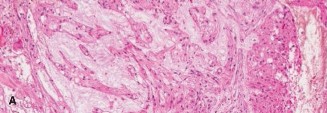
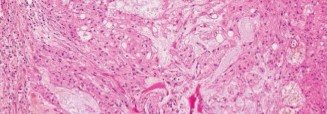
Histopathology:
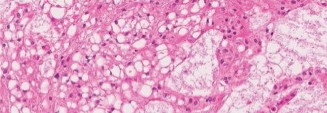
Microscopic examination of the biopsy specimens revealed characteristic physaliferous cells, which are pathognomonic for chordoma. These cells are vacuolated, often multinucleated, and arranged in cords or nests within a myxoid background. Immunohistochemistry was positive for S100 protein and brachyury, confirming the diagnosis. Brachyury positivity is highly specific for chordoma and helps differentiate it from other myxoid lesions.
-
Histopathology:
Microscopic examination of the biopsy specimens revealed characteristic physaliferous cells, which are pathognomonic for chordoma. These cells are vacuolated, often multinucleated, and arranged in cords or nests within a myxoid background. Immunohistochemistry was positive for S100 protein and brachyury, confirming the diagnosis. Brachyury positivity is highly specific for chordoma and helps differentiate it from other myxoid lesions.
-
Staging: Following the biopsy, a full metastatic workup including a PET-CT scan and chest CT was performed, which revealed no evidence of distant metastatic disease. The tumor was staged according to the Enneking surgical staging system for primary bone tumors as Stage IIB (high-grade malignant, extracompartmental). Using the Weinstein-Boriani-Biagini (WBB) surgical staging system, the tumor involved zones 4-8 (anterior-posterior sacrum) and layers A-E (medial to lateral extent, including neural canal involvement).
Differential Diagnosis
The presentation of a destructive sacral lesion necessitates a broad differential diagnosis. Key considerations include:
| Feature | Chordoma | Metastatic Carcinoma (e.g., Prostate, Renal, Lung) | Chondrosarcoma | Giant Cell Tumor of Bone (GCTB) |
|---|---|---|---|---|
| Prevalence | Rare (1-4% of primary bone tumors) | Most common malignant bone lesion in adults | Uncommon (2nd most common primary bone sarcoma) | Relatively common benign but locally aggressive |
| Age Group | Typically >40 years (peak 50-70s) | Typically >50 years | Typically >40 years | Typically 20-40 years |
| Primary Location | Sacrum (50-60%), Skull base (30-35%), Mobile spine | Axial skeleton (spine, pelvis, ribs) | Pelvis, proximal femur, shoulder | Epiphysis/metaphysis of long bones, sacrum (5-10%) |
| Pain Characteristics | Insidious, chronic, progressive, often radicular | Variable, often severe, may be nocturnal | Insidious, chronic, dull | Insidious, chronic, mechanical |
| Neurological Symptoms | Common with sacral involvement (bowel/bladder) | Less common unless advanced spinal cord compression | Less common with pelvic lesions | Rare, unless extensive vertebral collapse |
| Imaging (X-ray/CT) | Lytic, expansile, destructive mass; internal septa; no or minimal calcification | Lytic (renal, lung), blastic (prostate), or mixed; cortical destruction | Lytic, permeative, often with characteristic "rings and arcs" calcification (cartilaginous matrix) | Lytic, expansile, well-defined non-sclerotic margin; no calcification |
| Imaging (MRI) | T1 hypo, T2 hyper (lobulated, "soap bubble"), heterogeneous enhancement | Variable signal depending on primary, often T1 hypo, T2 variable; enhancement | T1 hypo, T2 hyper with lobulated appearance; septal enhancement | T1 hypo, T2 variable (fluid-fluid levels rare), heterogeneous enhancement |
| Histopathology | Physaliferous cells (vacuolated), myxoid matrix; S100 (+), Brachyury (+) | Adenocarcinoma, squamous cell carcinoma, clear cell, etc.; specific markers for primary origin | Chondrocytes in lacunae, myxoid matrix; varying grades; S100 (+) | Mononuclear stromal cells and multinucleated giant cells; H3F3A mutation in 90% |
| Management Principle | Wide en bloc resection + adjuvant proton beam RT | Palliative radiation, chemotherapy, targeted therapy, debulking surgery | Wide en bloc resection | Extended curettage + adjuvant (phenol, cryo), wide resection for recurrence/aggressive |
| Prognosis | Local recurrence common; 5-year survival 50-70% | Variable, depends on primary, stage, burden | Depends on grade; 5-year survival 60-90% | Locally aggressive, high recurrence rate (20-50%) |
Surgical Decision Making & Classification
The definitive diagnosis of chordoma mandates an aggressive oncologic approach. The primary goal of treatment for sacral chordoma is wide en bloc resection to achieve negative surgical margins (R0 resection), which offers the only chance for long-term disease-free survival. Local recurrence rates are high (up to 70%) if intralesional or marginal resections are performed.
Operative vs. Non-operative:
*
Non-operative management:
Palliative in nature, primarily for patients with extensive metastatic disease, severe comorbidities precluding surgery, or those refusing intervention. It typically involves pain management and radiation therapy (conventional external beam radiation, which is often insufficient for tumor control). For a localized sacral chordoma, non-operative management is not indicated as a primary curative strategy.
*
Operative management:
The only curative option. The decision to proceed with surgery is contingent upon the tumor's resectability, the patient's overall health status, and the anticipated functional outcome. In this patient, given his localized disease (Stage IIB, no metastases) and relatively good functional status despite his pain, wide en bloc sacrectomy was the clear choice.
Specific Classifications Guiding Decision-Making:
1.
Enneking Surgical Staging System:
As noted, this patient had a Stage IIB tumor. This classification guides the understanding of tumor aggressiveness and the necessity of wide margins. A Stage IIB lesion indicates an extracompartmental, high-grade malignancy, necessitating wide en bloc resection.
2.
Weinstein-Boriani-Biagini (WBB) Surgical Staging System:
This system is critical for precise preoperative planning of spinal tumor resections, including sacral lesions. It divides the vertebral body (and by extension, the sacrum) into 12 radial zones (1-12) and 6 concentric layers (A-E, plus F for metastasis). For this patient's tumor involving S2-S4 with significant anterior and intraspinal extension, the WBB system would characterize it as involving multiple zones (e.g., 4-8 for anterior and posterior elements) and layers (e.g., C-E for neural canal and paraspinal soft tissue involvement). This detailed mapping allows the surgical team to plan precise osteotomies to achieve wide margins while minimizing injury to uninvolved neurovascular structures. For sacral chordomas, often a hemisacrectomy or total sacrectomy below S2 is required. The level of S2 nerve root preservation is critical for bowel, bladder, and sexual function. Below S2, functional preservation is often not feasible.
The multidisciplinary tumor board, including orthopedic oncology surgeons, radiation oncologists, medical oncologists, neurosurgeons, and pathologists, reviewed the case. The consensus was for a planned high sacrectomy (above S3, possibly including S2 if required for margin) with spinopelvic reconstruction, followed by adjuvant proton beam radiation therapy. The proximity to the rectum mandated careful consideration of a combined anterior-posterior approach or a purely posterior approach with meticulous anterior dissection.
Surgical Technique / Intervention
The surgical goal was an en bloc resection of the sacral chordoma with wide margins, followed by reconstruction to maintain pelvic stability.
Pre-operative Considerations:
*
Bowel preparation:
Mechanical bowel preparation and prophylactic antibiotics were administered due to the proximity to the rectum.
*
Urological consult:
Indwelling urinary catheter placed.
*
Anesthesia:
General anesthesia with invasive arterial and central venous monitoring. Neuromonitoring (SSEP, MEP, free-running EMG) was critical for S2 nerve root identification and protection.
Patient Positioning:
The patient was positioned prone on a radiolucent operating table, typically using a Wilson frame, to allow for optimal exposure of the posterior sacrum and pelvis while facilitating fluoroscopy. All pressure points were meticulously padded.
Surgical Approach:
A
posterior approach
was chosen as the primary route, given the significant posterior and intraspinal tumor burden, with readiness for anterior dissection if required.
-
Incision and Exposure: A midline posterior incision was made from L5 to the distal coccyx. Subperiosteal dissection was performed to expose the posterior elements of L5, the iliac wings, and the entire sacrum. The gluteal muscles were detached from the sacrum laterally.
- This image would represent an intraoperative view after initial exposure, showing the posterior sacrum and surrounding musculature, prior to extensive tumor dissection.
-
Spinopelvic Fixation Planning: Prior to tumor resection, spinopelvic instrumentation was planned and partially performed. Pedicle screws were inserted into L4 and L5 vertebral bodies. Iliac screws were placed into the posterior superior iliac spine (PSIS) or through the sacroiliac joint into the ilium. This provided a stable platform for subsequent reconstruction.
-
Neurovascular Dissection: Meticulous dissection was carried out to identify and protect critical neurovascular structures.
- The S1 and S2 nerve roots were carefully identified and freed from the surrounding tissues superiorly. Given the tumor's extension, the S3, S4, and S5 nerve roots were deemed unreconstructable and were ligated and transected as part of the en bloc resection. The decision to sacrifice S3-S5 roots was made preoperatively based on tumor extent and anticipated functional deficit.
- The gluteal neurovascular bundles were identified and protected.
- Laterally, the sciatic nerve was identified and protected bilaterally.
-
Osteotomies and Tumor Resection:
- Using the pre-operative templating and intraoperative fluoroscopy, the superior osteotomy was performed transversely at a level determined to achieve a wide margin, typically through the S1 vertebral body or just superior to the S2 foramina. This osteotomy was performed with an oscillating saw, ensuring adequate bone margins.
- Lateral osteotomies were then performed through the ala of the sacrum, connecting the superior transverse osteotomy to the inferior limits of resection, typically encompassing the entire tumor within the resected specimen. These osteotomies were often performed with careful use of osteotomes and Gigli saws to minimize collateral damage.
- Anteriorly, careful dissection was performed to separate the tumor from the rectum, often utilizing electrocautery and blunt dissection. The median sacral artery and veins were ligated.
- Once all bony and soft tissue attachments were released, the entire sacral segment containing the chordoma was removed en bloc. The resected specimen was immediately sent for pathological assessment of margins.
-
Reconstruction and Stabilization:
- Following sacrectomy, a significant defect remained. The spinopelvic fixation (L4/L5 pedicle screws connected to iliac screws) was completed with long contoured rods. This reconstructive technique is crucial for maintaining pelvic ring stability and preventing pelvic collapse.
-
Various reconstruction options exist, including custom implants, cage reconstruction, or strut allografts/autographs, but for high sacrectomies, robust spinopelvic instrumentation is paramount.
- This image would represent a post-resection radiograph or 3D reconstruction illustrating the spinopelvic fixation construct, with rods connecting lumbar pedicle screws to iliac screws, spanning the sacral defect.
-
Closure: Hemostasis was meticulously achieved. Drains were placed in the dead space. The gluteal muscles were reapproximated, and the subcutaneous tissue and skin were closed in layers.
Post-Operative Protocol & Rehabilitation
Immediate Post-operative Phase (Days 0-7):
*
Pain Management:
Multimodal analgesia regimen including opioids, NSAIDs, and neuropathic pain medications (gabapentin/pregabalin) to manage both somatic and neuropathic pain from nerve root sacrifice. Epidural analgesia may be utilized initially.
*
Wound Care:
Strict wound care to prevent infection, given the extensive dissection and potential for hematoma. Drains typically remained in place until output was minimal.
*
Neurological Assessment:
Serial neurological checks, particularly bowel and bladder function, perianal sensation, and lower extremity strength. Patient education regarding potential permanent deficits.
*
Bowel/Bladder Management:
Regular monitoring for urinary retention and bowel function. Intermittent self-catheterization education may begin if urinary retention persists. Bowel regimen to prevent constipation and straining.
*
Mobilization:
Out of bed to chair on post-operative day 1, with assistance. Gentle ambulation with a walker or crutches as tolerated, emphasizing trunk control and avoiding excessive lumbar flexion or rotation.
Early Rehabilitation Phase (Weeks 1-6):
*
Physical Therapy (PT):
Initiated focusing on core strengthening, gait training, balance, and transfer techniques. Pelvic floor exercises may be prescribed if innervation allows or to improve compensatory mechanisms.
*
Occupational Therapy (OT):
Assessment for activities of daily living (ADLs) and assistive devices as needed.
*
Neuropathic Pain Management:
Optimization of medication regimen. Referral to pain clinic if necessary.
*
Adjuvant Radiation Therapy:
Once wound healing is satisfactory (typically 4-6 weeks post-surgery), the patient was referred for adjuvant proton beam radiation therapy. Proton therapy is preferred for chordomas due to its ability to deliver a high dose to the tumor bed while sparing surrounding critical structures (e.g., bowel, bladder, residual neural elements) compared to conventional photon therapy.
Long-Term Rehabilitation & Follow-up (Months to Years):
*
Oncology Follow-up:
Regular follow-up with the orthopedic oncology surgeon and radiation oncologist.
*
Imaging Surveillance:
Periodic MRI scans (every 3-6 months for the first 2-3 years, then annually) of the surgical site to monitor for local recurrence. Chest CTs may be performed to monitor for distant metastases, though less common for chordoma.
*
Functional Assessment:
Ongoing PT/OT to maximize functional independence. Address any persistent gait abnormalities, weakness, or sensory deficits.
*
Bowel/Bladder Management:
Continued management of neurogenic bowel/bladder. Referral to a neuro-urologist or gastroenterologist as needed.
*
Psychological Support:
Due to the impact on quality of life, psychological support and counseling may be beneficial.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- Early Biopsy, Correct Location: The definitive diagnosis of chordoma relies on histopathology. Ensure the biopsy is performed by an experienced interventional radiologist or surgeon, targeting the solid component of the tumor, and that the specimen is handled by an expert musculoskeletal pathologist. An inadequate or misdiagnosed biopsy can lead to inappropriate treatment.
- Multidisciplinary Team Approach: Chordoma management is complex and requires input from orthopedic oncology, neurosurgery (for skull base/upper cervical), radiation oncology (proton beam expertise), medical oncology, pathology, and rehabilitation specialists. A tumor board discussion is paramount.
- En Bloc Resection with Wide Margins: This is the cornerstone of curative treatment. Intralesional or marginal resections are associated with high local recurrence rates. The principle of R0 resection is paramount, even if it entails sacrificing significant neurovascular structures.
- Brachyury Immunohistochemistry: This is the most specific immunohistochemical marker for chordoma. Always confirm with Brachyury staining to differentiate from other myxoid tumors (e.g., chondrosarcoma, myxopapillary ependymoma).
- Proton Beam Radiation Therapy: Chordomas are relatively radioresistant, but proton beam therapy delivers high-dose, conformal radiation with minimal scatter, significantly improving local control compared to conventional photon radiation, especially in the post-operative setting for microscopic residual disease or in cases unsuitable for wide resection.
- S2 Sparing Principle: For sacral chordomas, the S2 nerve roots are critical for maintaining some bowel, bladder, and sexual function. If oncologically feasible, preserving at least one S2 root should be a consideration, balancing oncologic efficacy with functional outcome. For tumors involving S3 or lower, functional preservation of bowel/bladder is often not possible.
- Robust Spinopelvic Reconstruction: After high sacrectomy, comprehensive spinopelvic fixation (e.g., L4/L5 to iliac screws with long rods) is essential to restore pelvic stability and prevent subsidence or hardware failure.
Pitfalls:
- Inadequate Biopsy: Obtaining non-diagnostic tissue (e.g., necrotic tissue, blood) or misinterpreting histology can lead to delayed or incorrect treatment. Repeat biopsy should be considered if the initial one is inconclusive.
- Piecemeal Resection: Attempting to debulk the tumor or performing intralesional resection dramatically increases the risk of local recurrence and compromises the chance for cure. It can also lead to contamination of tissue planes.
- Underestimation of Tumor Extent: Insufficient preoperative imaging (e.g., no MRI or lack of contrast) can lead to an incomplete understanding of soft tissue extension or neural involvement, resulting in inadequate margins.
- Neurovascular Injury: Aggressive dissection in the sacral region carries a high risk of injury to the superior gluteal, internal iliac vessels, or sacral nerve roots, leading to significant morbidity (e.g., intractable pain, motor/sensory deficits, hemorrhage, neurogenic bladder/bowel).
- Poor Wound Healing/Infection: The extensive dissection, proximity to the perineum, and large dead space predispose to wound complications, particularly in irradiated fields. Careful surgical technique, meticulous hemostasis, and proper drain management are crucial.
- Delay in Referral: Sacral pain is often initially misdiagnosed as mechanical back pain. Delays in imaging and specialist referral can lead to larger tumors, more extensive resections, and worse outcomes. High index of suspicion for progressive, unremitting sacral pain, especially with neurological symptoms, is critical.
- Hardware Failure: Spinopelvic constructs are subjected to immense biomechanical forces. Incorrect screw placement, insufficient fusion, or early aggressive mobilization can lead to hardware failure, necessitating revision surgery.
- Ignoring Adjuvant Therapy: Even after an R0 resection, the local recurrence rate for chordoma remains significant. Neglecting adjuvant proton beam radiation, especially in cases with close margins or high-grade features, is a pitfall.
