INTRODUCTION TO OPEN ROTATOR CUFF REPAIR
While arthroscopic techniques have become the standard for many rotator cuff pathologies, the open repair of rotator cuff tears remains an indispensable procedure in the armamentarium of the orthopaedic surgeon. It is particularly indicated for massive, chronically retracted tears, revision surgeries, and cases requiring complex tendon transfers or biologic augmentation.
Understanding the pathoanatomy of the torn rotator cuff is paramount. Most tears are not simply transverse; they possess a significant longitudinal component, rendering them oval, U-shaped, or triangular. Consequently, all but the smallest of tears require multidirectional mobilization. The tendon must be advanced both anteriorly and laterally—not merely laterally—to restore the anatomical footprint and correct the resting length of the muscle-tendon unit. In tears exceeding 2 to 3 cm in diameter, the infraspinatus tendon is almost invariably involved, necessitating a comprehensive approach to both the superior and posterior aspects of the cuff.
PREOPERATIVE PLANNING AND EXPOSURE
Surgical Approach
The standard approach for an open rotator cuff repair is the anterolateral deltoid-splitting approach. Meticulous care must be taken to preserve the deltoid origin and protect the axillary nerve, which courses approximately 5 cm distal to the lateral border of the acromion.
Surgical Warning: Compromise of the deltoid origin or iatrogenic injury to the axillary nerve transforms a potentially successful rotator cuff repair into a catastrophic functional failure. Always maintain a strict "safe zone" during deltoid splitting.
Once the subacromial space is accessed, a thorough bursectomy is performed to identify the tear geometry. When the defect has been identified and its size approximated, attention is turned to the repair itself. In chronic tears, the tendon edges are often scarred, retracted, and adherent to surrounding structures, making mobilization the most critical phase of the operation.
TENDON MOBILIZATION TECHNIQUES
Adequate mobilization is the cornerstone of a tension-free repair. Excessive tension at the repair site is the most common mechanism of failure, typically resulting in the suture cutting through the tendon (cheese-wiring).
Posterior Mobilization
Begin mobilization posteriorly with the infraspinatus. Using a blunt probe, a Cobb elevator, or a gloved finger, systematically release adhesions both inside and outside the joint capsule.
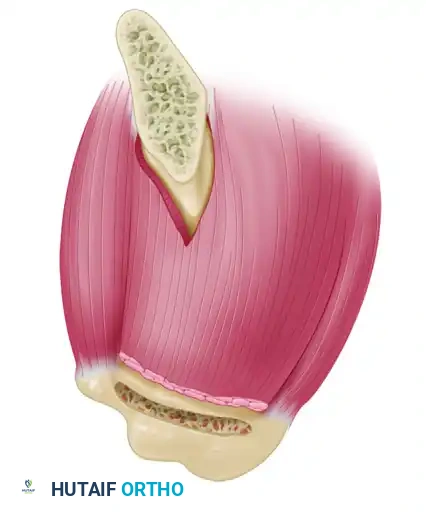
Clinical Pearl: Do not dissect below the level of the teres minor. Aggressive inferior dissection risks injury to the axillary nerve within the quadrangular space, or the suprascapular nerve as it courses through the spinoglenoid notch near the inferior border of the supraspinatus fossa.
Anterior Mobilization
Continue mobilization anteriorly toward the supraspinatus. The supraspinatus and subscapularis muscles have strong fascial attachments to the coracoid base via the coracohumeral ligament. Sharp release of the coracohumeral ligament in this area is essential and allows significant lateral excursion of the supraspinatus.
If necessary, additional superior exposure can be gained by resecting the distal 1.0 to 1.5 cm of the clavicle at the acromioclavicular (AC) joint. However, this should be strictly reserved for patients with concomitant, symptomatic AC joint arthrosis to avoid unnecessary destabilization.
Capsular Release
If the supraspinatus and infraspinatus tendons remain severely retracted and adequate length cannot be obtained despite the aforementioned releases, a capsular release is indicated.
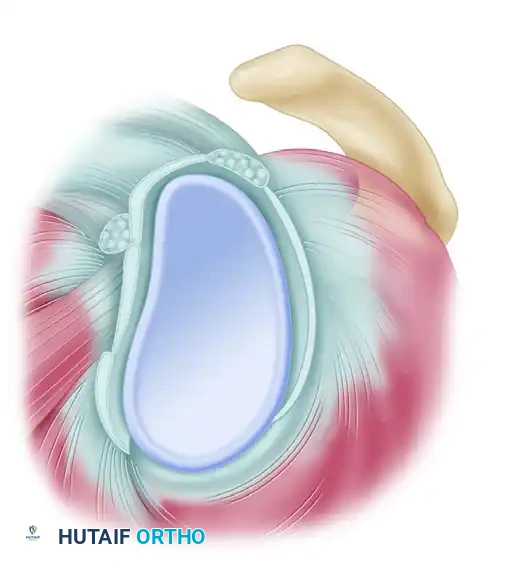
Incise the capsule sharply at its insertion into the glenoid labrum. If necessary, carry this incision from the 8-o’clock position posterior to the 4-o’clock position posterior. This allows the muscle belly to slide laterally independent of the glenohumeral capsule.
Note: The use of a second posterior incision over the scapular spine to increase mobilization has been described historically (e.g., the Debeyre technique), but it results in a loss of muscle strength of at least one grade and is generally not recommended in modern practice.
FOOTPRINT PREPARATION AND REPAIR BIOMECHANICS
The goals of mobilization and preparation are fourfold:
1. Obtain tissue of adequate biomechanical strength.
2. Position the tendon anatomically without tension, nerve damage, or deltoid compromise.
3. Decompress the subacromial space to prevent mechanical impingement.
4. Maximize the surface area for tendon-to-bone healing.
Creating the Bony Trough
Débride the end of the mobilized tendon to obtain a raw, bleeding edge, taking care not to confuse the true tendon with the overlying hypertrophic bursa.
Use a rongeur, osteotome, or motorized high-speed burr to create a shallow trough running the length of the exposed footprint on the greater tuberosity. This trough accommodates the thickness of the supraspinatus and infraspinatus tendons.
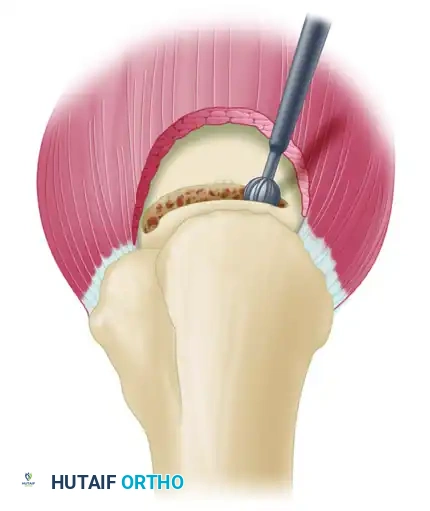
Surgical Pearl: Always bevel the proximal edge of the trough with a burr or rasp. A sharp cortical edge will act as a fulcrum, leading to premature suture abrasion and failure at the tendon-bone interface.
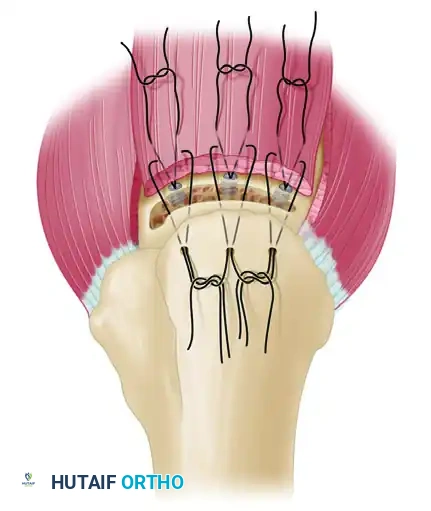
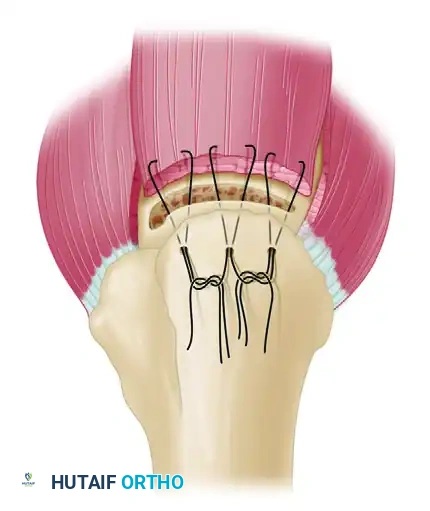
The Double-Row Repair Technique
We advocate for a double-row technique, suturing the tendon to bone in a cancellous trough in combination with suture anchor fixation. This construct significantly reduces tension on the primary trough repair, increases the footprint contact area, and provides superior biomechanical yield strength compared to single-row repairs.
- Medial Row (Anchors): Place two or three rotator cuff suture anchors immediately medial to the articular margin of the trough at a 45-degree angle (the "deadman's angle").
- Suture Passing: Pass the sutures through the rotator cuff tendon 3 to 5 mm medial to the free edge.
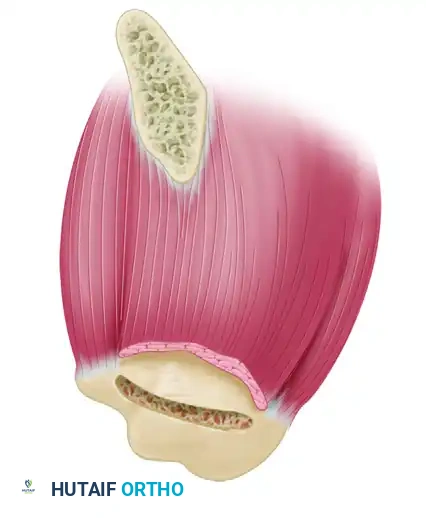
- Lateral Row (Transosseous Tunnels): Drill holes for sutures 2 to 3 cm distal to the trough on the lateral humeral cortex. Connect these holes to the trough using a No. 5 Mayo needle, a towel clip, or a specialized suture-passing instrument.
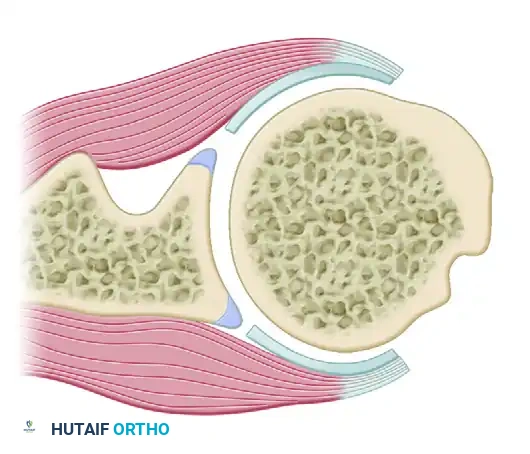
Pitfall: Take extreme care not to fracture the thin cortical bone of the lateral humerus, which is frequently osteoporotic in this patient demographic. Space the drill holes at least 5 mm (preferably 1 cm) apart to provide an adequate cortical bridge over which to tie the knots.
- Knot Tying: With No. 2 nonabsorbable suture, use a double loop technique (superior to inferior, and inferior to superior) in a horizontal mattress manner. This configuration helps push the tendon down directly into the bleeding cancellous trough. Tie the anchor sutures down on top of the tendon with four or five alternating half-hitches to prevent impingement of the knot stack.

Salvage Note: If the lateral humeral cortex fractures during the tying of the transosseous sutures, lateral row suture anchors can be used as a salvage procedure. Anchors have adequate holding power in cancellous bone and are excellent alternatives in problematic, osteoporotic situations.
MANAGEMENT OF MASSIVE AND IRREPARABLE TEARS
Occasionally, despite the surgeon’s most diligent efforts to mobilize the tendons, tension remains unacceptably high. Cordasco and Bigliani identified five critical factors that improve the results of operative treatment for large and massive rotator cuff tears:
1. Adequate subacromial decompression.
2. Maintaining the integrity of the deltoid origin.
3. Mobilizing torn tendons and performing an interval slide when indicated.
4. Repairing tendons directly to bone.
5. Carefully supervising and staging postoperative rehabilitation.
In the ideal repair, the arm can be brought down to the patient’s side without tension. If tension remains, several salvage options exist, though none are biomechanically ideal.
Marginal Convergence (Partial Repair)
Partial repair of massive rotator cuff tears is an excellent alternative to simple débridement. The initial step is a side-to-side tendon repair that results in "marginal convergence" toward the greater tuberosity.
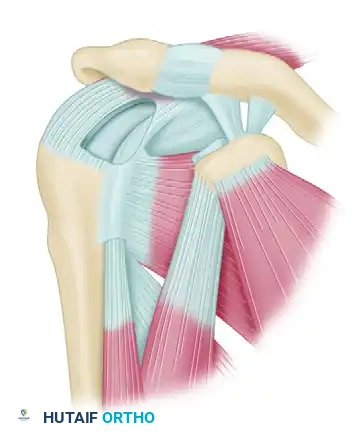
By suturing the anterior and posterior leaves of the tear together, the surgeon decreases the strain at the free margin of the rotator cuff, enhancing the mechanics of the construct and converting a massive U-shaped tear into a smaller crescentic tear that can then be repaired to bone.
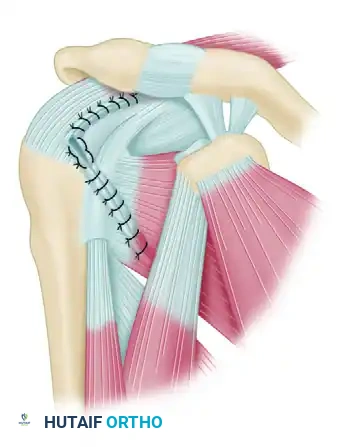
The McLaughlin Technique (Nonanatomical Repair)
If the tendon cannot reach the anatomical footprint, McLaughlin described suturing the tendon to a trough in bone at whatever point it can reach on the humeral head.
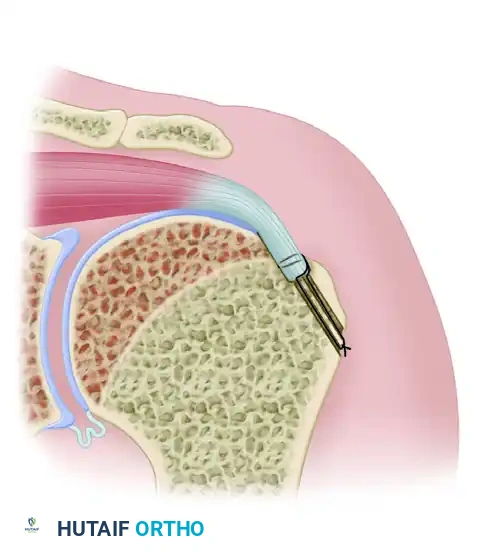
This may require creating a trough more proximally (approximately 2 cm medial to the greater tuberosity) through the anterior neck area. While this achieves a watertight closure and restores the force couple, the mechanical advantage of the muscle-tendon unit is significantly diminished due to the proximal advancement.
BIOLOGIC AUGMENTATION AND SCAFFOLDS
In an effort to decrease tension, increase construct strength, and improve the biologic healing environment, numerous augmentation methods have been developed. These include autografts (biceps, subscapularis, fascia lata), allografts, xenografts, and synthetic grafts.
Extracellular Matrix (ECM) Scaffolds
The most frequently used augmentation methods involve scaffold devices derived from extracellular matrix (ECM) of human, porcine, bovine, or equine sources. Examples of FDA-approved devices include:
* Restore SIS (Porcine)
* CuffPatch (Cross-linked Porcine)
* GraftJacket (Human Dermis)
* TissueMend (Bovine Fetal Dermis)
Biomechanical models suggest that in an augmented repair, 70% to 80% of the load is distributed to the native tendon, with 20% to 30% carried by the augmentation device. However, clinical follow-up studies in humans have reported mixed results regarding surgical outcomes and complication rates (such as sterile inflammatory reactions).
Platelet-Rich Plasma (PRP) and Orthobiologics
Molecular and cellular studies have heavily targeted the tendon-bone interface. Platelet-rich plasma (PRP) has become a popular adjunct, defined as a volume of plasma with a platelet count above whole blood baseline.
Despite its popularity, the literature remains equivocal. While some small prospective studies show improved UCLA and Constant scores at 2 years, larger randomized controlled trials comparing rotator cuff repairs with and without PRP augmentation have found no statistically significant differences in clinical scores or MRI-confirmed healing rates.
POSTOPERATIVE CARE AND REHABILITATION
The choice of suture type and technique can improve initial repair strength, but decreasing the postoperative activity level is equally critical to prevent failure.
Phase I: Protection (0 to 6 Weeks)
* The patient is placed in an abduction pillow, low-profile pillow sling, or shoulder immobilizer for 6 weeks.
* The repair is biomechanically weakest at 3 weeks, and tendon strength remains less than it was at the time of surgery for the first 3 months.
* The sling is removed only for strictly supervised, passive, assisted exercises in flexion and external rotation to prevent adhesive capsulitis and disuse atrophy.
Phase II: Early Motion (6 to 12 Weeks)
* Empirically, patients are advanced to isometric exercises of external rotation and flexion at 6 weeks.
* Active-assisted range of motion is initiated, gradually weaning the patient off the abduction sling.
Phase III: Strengthening (12+ Weeks)
* At 12 weeks, full active motion and progressive resistance exercises are permitted.
* Patients must be explicitly cautioned that overaggressive use of the extremity can lead to disruption of the repair for up to 6 to 12 months, depending on the initial tear size and tissue quality.
Clinical Pearl: Patient compliance is the most unpredictable variable in rotator cuff surgery. It is difficult to enforce the use of an abduction splint for 6 weeks due to hygiene, comfort, and driving restrictions. However, without early passive movement, adhesive capsulitis is highly likely, potentially producing a poorer functional result than the original tear.
CONCLUSION
Open repair of the rotator cuff remains a highly effective, durable procedure when executed with precision. Success hinges on meticulous tendon mobilization, anatomical footprint restoration using robust double-row or transosseous techniques, and strict adherence to a staged rehabilitation protocol. By respecting the biomechanical and biological limits of the tissue, orthopaedic surgeons can consistently achieve excellent pain relief and functional restoration, even in the setting of massive and complex tear patterns.
