INTRODUCTION TO MASSIVE BONE DEFECT RECONSTRUCTION
The management of critical-size bone defects and recalcitrant nonunions remains one of the most formidable challenges in orthopedic surgery. Whether secondary to high-energy trauma, radical tumor resection, severe osteomyelitis, or previously failed osteosyntheses, these defects require a meticulous reconstruction strategy that addresses both mechanical stability and biological vitality.
Historically, the massive sliding graft and the whole fibular transplant have served as the cornerstone techniques for bridging substantial diaphyseal defects. While modern orthopedics has seen the advent of distraction osteogenesis and recombinant bone morphogenetic proteins (rhBMPs), autogenous structural grafting remains an indispensable tool in the reconstructive surgeon's armamentarium. These grafts provide an unparalleled combination of an osteoconductive scaffold, osteoinductive growth factors, and, in the case of vascularized transfers, immediate osteogenic potential.
This masterclass delineates the precise surgical indications, biomechanical principles, and step-by-step operative techniques for massive sliding grafts and fibular transplants, ensuring the postgraduate surgeon is fully equipped to execute these complex procedures.
MASSIVE SLIDING GRAFT
The massive sliding graft, originally devised by Gill and subsequently refined by Flanagan and Burem, is a powerful technique for the treatment of diaphyseal nonunions, particularly in the tibia and femur. The procedure involves harvesting a massive cortical graft—comprising approximately one-half to two-thirds of the bone's circumference and measuring 10 to 15 cm in length—directly from the host bone adjacent to the nonunion site.
Biomechanical and Biological Rationale
The primary advantage of the massive sliding graft is the utilization of local, biologically active autogenous bone that perfectly matches the contour and cortical density of the recipient site. By sliding a massive segment of diaphyseal bone across the pseudarthrosis, the surgeon creates a rigid, intrinsic neutralization plate.
⚠️ Surgical Warning: The High Stakes of Failure
While highly effective for bridging defects, the massive sliding graft is a high-stakes procedure. If a massive sliding graft fails to unite, subsequent grafting procedures are exponentially more difficult. The local bone stock is profoundly depleted, the medullary canal is compromised, and the regional vascularity is heavily scarred. Patient selection and flawless surgical execution are paramount.
Indications and Contraindications
Indications:
* Aseptic nonunions of the tibial or femoral diaphysis.
* Short-segment intercalary bone defects where local bone stock is otherwise healthy.
* Cases where autologous iliac crest harvesting is contraindicated or insufficient.
Contraindications:
* Active or latent osteomyelitis at the nonunion site.
* Severe osteopenia or irradiated bone (which lacks the structural integrity to serve as a graft).
* Critical-size defects exceeding the length that can be safely harvested from the adjacent fragment.
🔪 Surgical Technique 59-3: The Gill Massive Sliding Graft
The following technique details the classic Gill procedure, optimized for modern rigid internal fixation principles.
1. Preoperative Planning and Positioning:
* Obtain orthogonal radiographs and a CT scan to assess the exact geometry of the nonunion and the cortical thickness of the proximal and distal fragments.
* Position the patient supine on a radiolucent table. Apply a sterile pneumatic tourniquet if operating on the tibia.
* Ensure fluoroscopy is available and positioned appropriately.
2. Surgical Approach and Exposure:
* Make an extensile longitudinal incision over the affected bone (e.g., an anterolateral approach to the tibia).
* Carry the dissection down to the periosteum.
* Crucial Step: Split the periosteum longitudinally over both fragments. Elevate the periosteum minimally—only enough to accommodate the osteotomies. Excessive periosteal stripping will devascularize the host bone and doom the graft to failure.
3. Graft Design and Osteotomy:
* Using a sterile surgical marker, map out the graft dimensions. The total length of the combined grafts should be approximately 10 to 15 cm.
* Plan to take the longer graft from the longer bone fragment.
* Using an oscillating saw under continuous cold saline irrigation (to prevent thermal necrosis of the osteocytes), cut a graft from each fragment. The depth of the cut should encompass one-half to two-thirds of the thickness of the bone.
4. Graft Transposition and Fixation:
* Extract both the long and short cortical grafts.
* Place the longer graft directly across the nonunion site, bridging the defect.
* Fasten the massive graft to both host fragments using multiple cortical lag screws (typically 3.5 mm or 4.5 mm, depending on the bone). Ensure absolute rigid compression.
* Clinical Pearl: If the two host fragments are not in direct contact, do not make an effort to acutely shorten the limb to appose them. Rely entirely on the heavy structural graft to bridge the gap and facilitate union.
5. Defect Management:
* Place the shorter graft into the remaining defect created by the harvest of the longer graft.
* Pack any remaining voids with autogenous cancellous bone chips (harvested from the iliac crest or local metaphysis) to maximize the osteogenic potential at the docking sites.
WHOLE FIBULAR TRANSPLANTS (NON-VASCULARIZED)
When local bone stock is insufficient for a sliding graft, the whole fibular transplant serves as an exceptional structural autograft. Because the fibula is tubular, its moment of inertia provides significantly greater bending and torsional strength compared to a flat tibial cortical graft of the same cross-sectional area.
Site-Specific Indications
- Radius and Ulna: The non-vascularized fibula is the gold standard for bridging massive defects in the forearm. It does not unduly crowd the tight fascial compartments of the forearm, making wound closure straightforward while restoring the critical length necessary for pronation and supination.
- Distal Humerus: Useful when combined with a large volume of autogenous iliac cancellous bone to reconstruct the metaphyseal expansion.
- Pediatric Tibia: While too small for adult tibial defects, the pediatric fibula possesses immense remodeling potential. In children, a fibular strut will hypertrophy over years of growth to approach the size and strength of a normal tibia.
- Adult Humerus and Tibia (Contraindicated): In adults, non-vascularized fibular grafts do not hypertrophy sufficiently in high-stress environments (like the humeral shaft or adult tibia) and are highly prone to fatigue fracture.
🔪 Surgical Technique 59-4: Whole Fibular Transplant
1. Preparation of the Recipient Bed:
* Through an appropriate extensile incision, meticulously excise all pseudarthrosis and scar tissue to expose a healthy, bleeding vascular bed.
* Resect all sclerotic, avascular, or nonviable bone from the fragment ends.
* Square the ends of the host bone with a rongeur or oscillating saw.
* Using a drill or a curet, aggressively ream out the medullary canals of both host fragments until punctate bleeding is observed, ensuring access to endosteal blood supply.
2. Restoration of Length:
* Apply longitudinal traction to the extremity. Determine the maximal anatomical length that can be safely restored without placing undue tension on the neurovascular bundles.
3. Fibular Harvest:
* Harvest a fibular transplant from the middle third of the donor leg. Ensure the graft is long enough to bridge the full defect and overlap the host bone fragments sufficiently to permit firm screw fixation.
* Warning: Always preserve the distal 6 to 8 cm of the fibula to maintain the integrity of the distal tibiofibular syndesmosis and ankle mortise. Protect the common peroneal nerve proximally.
4. The Step-Cut Osteotomy:
* Step-cut the fibular transplant at both ends. The intact middle portion of the graft must be the exact size of the defect being bridged.
* Preserve the step-cut pieces removed from each end; these are highly valuable cortical slivers.
* With an osteotome or burr, flatten the corresponding surfaces of the host bone fragments to perfectly receive the step-cut ends of the fibular transplant. This step-cut geometry exponentially increases the surface area for osteoconduction and provides intrinsic rotational stability.
5. Fixation and Biological Augmentation:
* Fit the transplant into the defect. Fix it rigidly to both host fragments using interfragmentary lag screws.
* Cut the preserved step-cut bone pieces into fine slivers. Place these slivers circumferentially around the junctions of the transplant and the host bone.
* Augment the junctions generously with autologous iliac crest cancellous bone chips to jump-start osteogenesis.
* Alternative Technique: If one host fragment is too short to accommodate a step-cut onlay, the transplant can be inserted directly into the medullary canal at that end (intramedullary peg) and applied as an onlay at the other end.
FREE VASCULARIZED FIBULAR GRAFTS (FVFG)
The advent of microsurgery revolutionized the treatment of massive bone defects through the Free Vascularized Fibular Graft (FVFG). By anastomosing the peroneal artery and venae comitantes to recipient vessels, the graft bypasses the prolonged process of "creeping substitution." The bone remains alive, allowing for primary bone healing at the host-graft junctions and immediate resistance to infection.
Indications for FVFG
- Osteonecrosis of the Femoral Head: Used as a vascularized strut to decompress the necrotic lesion and provide structural support to the subchondral plate.
- Irradiated Bone Beds: Non-vascularized grafts universally fail in irradiated tissue due to the lack of a vascular bed. FVFG brings its own blood supply, making it the only viable autogenous option for post-tumor resection reconstruction.
- Upper Extremity Defects > 5 cm: The FVFG performs exceptionally well in the humerus and forearm for massive defects.
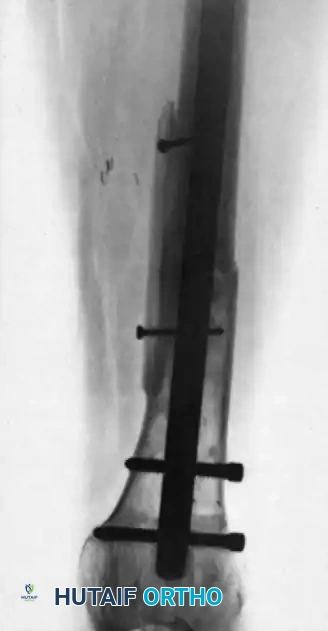
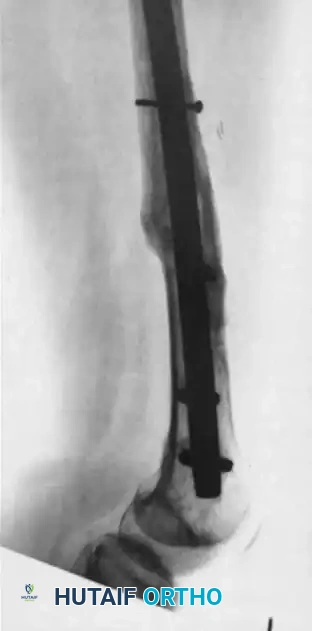
FIGURE 59-8: Posteroanterior (A) and lateral (B) radiographs made 3 years after free vascularized fibular transfer combined with autografting for the management of a fracture nonunion associated with radiation therapy. Note the excellent remodeling, massive hypertrophy, and complete fracture healing.
FVFG vs. Distraction Osteogenesis in the Lower Extremity
While FVFG is highly successful in the upper extremity, its use in the lower extremity is highly debated.
💡 Clinical Pearl: The Lower Extremity Dilemma
Distraction osteogenesis (Ilizarov bone transport) is generally considered far superior to free vascularized fibular grafting for massive defects of the tibia and femur.
The primary limitation of FVFG in the lower extremity is the profound time required for the fibula to hypertrophy sufficiently to withstand full weight-bearing forces. Patients often require years of protective weight-bearing in specialized braces to avoid catastrophic fatigue fractures of the graft. Furthermore, the donor site morbidity (muscle weakness, nerve neuropraxia, ankle instability) is not trivial. Distraction osteogenesis, conversely, generates native-diameter bone that permits earlier weight-bearing and avoids donor site morbidity entirely.
POSTOPERATIVE CARE AND REHABILITATION
The postoperative management of massive structural grafts requires a delicate balance between protecting the mechanical construct and stimulating biological healing.
1. The Biology of Creeping Substitution:
For non-vascularized massive sliding grafts and whole fibular transplants, the surgeon must understand that the graft is initially avascular. Healing occurs via creeping substitution—a prolonged process where osteoclasts resorb the dead graft bone while osteoblasts simultaneously lay down new woven bone.
* The Danger Zone: During the intermediate phase of creeping substitution (typically 6 to 18 months postoperatively), the graft becomes temporarily weaker and more porous before it becomes stronger. This is the period of highest risk for fatigue fracture.
2. Immobilization and Support:
* Postoperative care is similar to routine grafting, but significantly more time is necessary for complete revascularization.
* Even if radiographs show that the ends of the fragments have united with the transplant, full structural strength is not restored until the entire diaphyseal graft has been revascularized and remodeled.
* Consequently, mechanical support must be continued for an extended time.
* Preferably, a custom-molded removable orthosis or a functional brace with joints that allow active and passive range of motion is utilized. This permits joint preservation while shielding the graft from bending and torsional moments.
3. Weight-Bearing Protocol:
* Upper Extremity: Lifting restrictions are strictly enforced until radiographic evidence of complete incorporation at both the proximal and distal docking sites is confirmed.
* Lower Extremity: Patients are kept strictly non-weight-bearing initially. Progression to partial weight-bearing is delayed until bridging callus is visible at the junctions. Full weight-bearing without a brace may take 12 to 24 months, depending on the size of the defect and the age of the patient.
4. Radiographic Monitoring:
* Serial radiographs should be obtained at 6-week intervals initially, then every 3 months.
* The surgeon must look for signs of incorporation (blurring of the osteotomy lines), hypertrophy (cortical thickening in response to stress), or impending failure (radiolucent lines around screws, graft resorption, or stress fractures). If a stress fracture occurs in a massive graft, immediate intervention with prolonged immobilization or supplemental fixation is required to prevent catastrophic failure of the reconstruction.
