KLIPPEL-FEIL SYNDROME: PATHOPHYSIOLOGY AND EMBRYOLOGY
Klippel-Feil syndrome (KFS) is a complex congenital anomaly characterized by the abnormal fusion of two or more cervical vertebrae. This condition arises from a failure of normal segmentation and cleavage of the cervical somites during the critical third to eighth weeks of embryogenesis. The resulting synostosis may manifest as a simple congenital block vertebra involving only two segments, or it may present as a massive, contiguous fusion encompassing the entire cervical and upper thoracic spine.
The precise etiology of Klippel-Feil syndrome remains multifactorial and, in most sporadic cases, elusive. Contemporary embryological theories propose a primary vascular disruption sequence during early fetal development, leading to localized hypoxia and subsequent failure of somite segmentation. Beals and Rolfe hypothesized that a global embryonic insult—or a series of multiple, temporally distinct insults—could account for both the cervical fusions and the myriad of associated systemic anomalies.
Genetic inheritance plays a definitive role in a subset of patients. Gunderson et al. demonstrated that congenital fusion of the C2-C3 segments often follows an autosomal dominant inheritance pattern, though the genetic transmission of other fusion patterns remains less predictable. Furthermore, Clarke et al. identified a familial KFS variant characterized by consistent C2-C3 fusion, mapping the gene locus to the long arm of chromosome 8 (8q). Environmental factors, particularly maternal alcoholism, have also been strongly implicated; Tredwell et al. reported a 50% incidence of cervical vertebral fusions in patients diagnosed with fetal alcohol syndrome.
Clinical Pearl: While occipitalization of the atlas, hemivertebrae, and basilar impression are frequently observed in patients with Klippel-Feil syndrome, their isolated occurrence without subaxial cervical fusion does not meet the diagnostic criteria for KFS.
EPIDEMIOLOGY AND CLASSIFICATION
Because many patients with minor fusions remain entirely asymptomatic throughout their lives, the true incidence of Klippel-Feil syndrome is difficult to ascertain. Current literature estimates range broadly from 1 in 42,400 births to as high as 3 in 700, with a slight male predominance (1.5:1).
The Feil Classification System
Maurice Feil originally classified the syndrome into three distinct morphological types based on the extent of spinal involvement:
* Type I: Massive block fusion encompassing all cervical and upper thoracic vertebrae.
* Type II: Focal fusion involving only one or two pairs of cervical vertebrae (most common).
* Type III: Cervical fusion occurring in combination with lower thoracic or lumbar vertebral fusions.
CLINICAL PRESENTATION
The classic clinical presentation of Klippel-Feil syndrome is defined by a triad of physical signs:
1. A short neck
2. A low posterior hairline
3. Limited range of neck motion
Despite its historical significance, fewer than 50% of patients present with the complete triad. The triad typically indicates extensive, multi-segmental cervical involvement (Type I). Many patients possess a normal gross appearance, and the syndrome is often an incidental radiographic finding. In severe cases, webbing of the neck (pterygium colli) may be prominent.
The most consistent clinical finding is a restriction in cervical range of motion, with rotation and lateral bending typically more impaired than flexion and extension. If the fusion involves fewer than three vertebrae or is localized to the lower cervical spine, motion limitation may be clinically imperceptible.
Neurological and Mechanical Symptoms
Symptoms in KFS rarely originate from the fused segments themselves; rather, they arise from the adjacent open (unfused) interspaces. These open segments become hypermobile to compensate for the loss of motion at the fused levels, leading to altered biomechanical stress.
* Mechanical Symptoms: Ligamentous and capsular stretching at the hypermobile segment leads to accelerated degenerative joint disease (DJD), manifesting as localized axial neck pain.
* Neurological Symptoms: Hypertrophy of the ligamentum flavum, osteophyte formation, or frank instability at the hypermobile segment can result in nerve root impingement (radiculopathy) or spinal cord compression (myelopathy). Severe cord compression can precipitate spasticity, hyperreflexia, profound muscle weakness, quadriplegia, or even sudden death following minor trauma.
HIGH-RISK BIOMECHANICAL PATTERNS
Fielding et al. and Hensinger et al. identified three specific patterns of cervical spine fusion that carry a disproportionately high risk for late-onset instability, degenerative disease, and catastrophic neurological compromise.
- Pattern 1: C1-C2 Fusion with Occipitalization of the Atlas.
This pattern forces all flexion and extension motion to occur at the atlantoaxial joint. The odontoid process becomes hypermobile and is prone to posterior dislocation, which can critically narrow the spinal canal and compress the cervicomedullary junction. - Pattern 2: Long Fusion with an Abnormal Occipitocervical Junction.
Forces of flexion, extension, and rotation are concentrated through a dysplastic odontoid or a hypoplastic C1 ring. Over time, this abnormal articulation inevitably becomes unstable. (Note: A long fusion with a normal C1-C2 articulation carries a much lower risk and a normal life expectancy). - Pattern 3: A Single Open Interspace Between Two Fused Segments.
Cervical motion is entirely concentrated at this single open segment. The segment acts as a mechanical hinge, rapidly becoming hypermobile, unstable, and severely arthritic.
Surgical Warning: Patients with Pattern 3 fusions are at extreme risk for adjacent segment disease. On dynamic lateral radiographs, the cervical spine will appear to "hinge" or subluxate at this single open segment. Prophylactic stabilization may be indicated if progressive neurological deficits or dynamic instability is documented.
ASSOCIATED SYSTEMIC CONDITIONS
Because the embryonic insult in KFS occurs during the critical period of organogenesis (weeks 3-8), multiple organ systems are frequently involved. A multidisciplinary approach is mandatory.
Orthopedic Anomalies
- Scoliosis and Kyphosis: The most frequent orthopedic complication, occurring in 60% to 70% of patients. Two types exist: congenital scoliosis (due to hemivertebrae or unilateral unsegmented bars) and compensatory idiopathic-like curves below the cervical anomaly. Full-spine radiographs are mandatory.
- Sprengel Deformity: Present in approximately 20% of cases. The failure of the scapula to descend from its embryonic cervical position results in an elevated, dysplastic shoulder, often tethered by an omovertebral bone.
- Cervical Ribs: Occur in 12% to 15% of patients, increasing the risk of thoracic outlet syndrome.
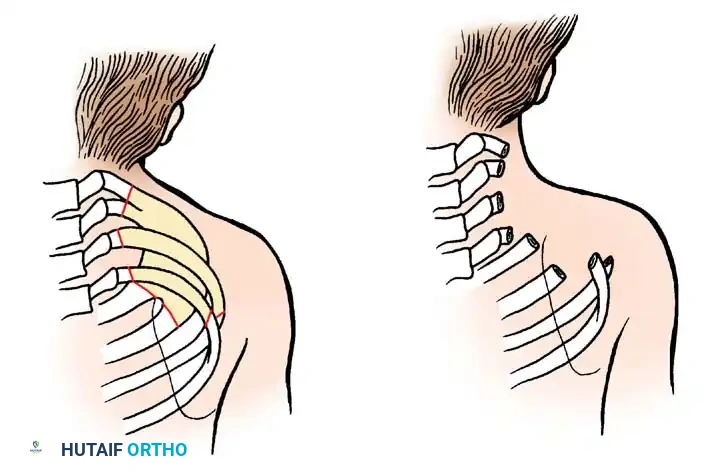
Non-Orthopedic Anomalies
- Renal Abnormalities (30%): Unilateral renal agenesis is the most common. Other anomalies include horseshoe kidney, ectopic kidneys, and hydronephrosis. Routine screening via renal ultrasound is imperative, as these conditions are often asymptomatic but potentially life-threatening.
- Cardiovascular Anomalies (4-14%): Ventricular septal defects (VSD) are the most common. Others include patent ductus arteriosus, coarctation of the aorta, and bicuspid aortic valves.
- Deafness (30%): Hearing loss may be conductive (ossicular ankylosis), sensorineural, or mixed. Early audiometric testing is critical to prevent speech and language delays.
- Synkinesis (Mirror Movements) (20%): Involuntary paired movements of the hands. Autopsy studies suggest incomplete decussation of the pyramidal tracts in the upper cervical cord. This usually improves with age and occupational therapy.
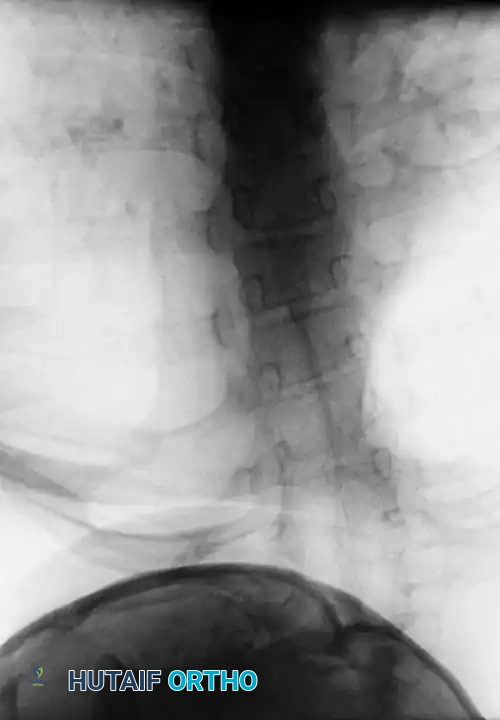
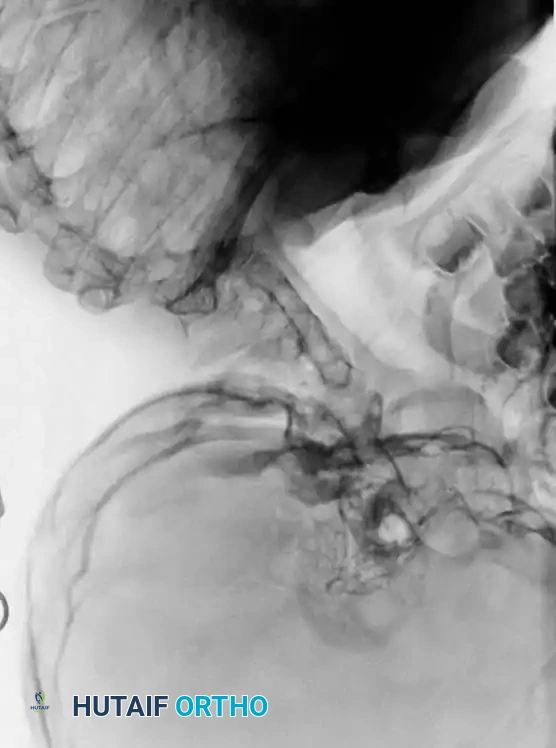
RADIOGRAPHIC EVALUATION
A comprehensive imaging protocol is the cornerstone of KFS management.
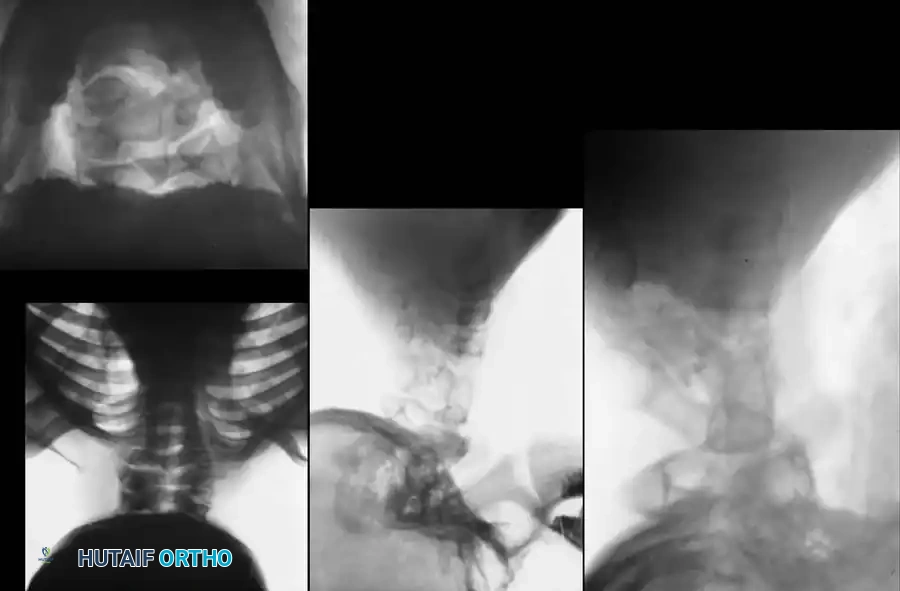
Initial evaluation must include high-quality anteroposterior (AP), open-mouth odontoid, and lateral cervical radiographs.
Dynamic Imaging
Lateral flexion-extension views are the most critical plain films obtained. They are essential for identifying occult atlantoaxial instability or hypermobility at an open segment adjacent to a fusion block.
Advanced Imaging (CT and MRI)
- Computed Tomography (CT): 3D CT reconstructions are invaluable for delineating complex bony anatomy, identifying hemivertebrae, and planning surgical screw trajectories in dysplastic pedicles or lateral masses.
- Magnetic Resonance Imaging (MRI): Mandatory for any patient exhibiting neurological symptoms or those with high-risk fusion patterns. MRI evaluates the spinal cord for myelomalacia, syringomyelia, hydromyelia, or associated Arnold-Chiari malformations.
SURGICAL MANAGEMENT: INDICATIONS AND TECHNIQUES
Surgical intervention in Klippel-Feil syndrome is reserved for patients with documented progressive instability, intractable mechanical pain failing conservative management, or progressive neurological deficits (myelopathy/radiculopathy).
Preoperative Considerations and Positioning
- Airway Management: Intubation can be exceptionally hazardous due to the rigid, shortened neck. Awake fiberoptic intubation is the gold standard to prevent iatrogenic hyperextension injuries to the spinal cord.
- Neuromonitoring: Somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) are mandatory during positioning and throughout the procedure.
- Positioning: For posterior approaches, the patient is placed prone using Mayfield tongs or a halo ring. Extreme care must be taken to maintain the neck in a neutral, unforced position.
Technique 1: Posterior Occipitocervical Fusion (For Patterns 1 & 2)
Patients with atlantoaxial instability or abnormal occipitocervical junctions require rigid stabilization to prevent cervicomedullary compression.
- Exposure: A midline posterior incision is made from the external occipital protuberance down to the subaxial cervical spine. The paraspinal musculature is elevated subperiosteally.
- Instrumentation: Depending on the patient's age and bone quality, occipital plates are secured to the thickest portion of the occiput (near the inion). Cervical fixation is achieved via C2 pedicle/pars screws and subaxial lateral mass screws.
- Decompression: If basilar invagination or severe stenosis is present, a suboccipital craniectomy and C1 laminectomy are performed prior to rod placement.
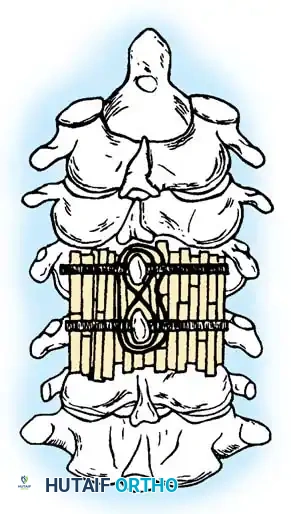
- Arthrodesis: The posterior elements are decorticated using a high-speed burr. Autologous iliac crest bone graft (ICBG) or allograft is meticulously packed over the decorticated surfaces.

(Historical representation of posterior cervical wiring and strut grafting, often utilized before the advent of modern rigid screw-rod constructs).
Technique 2: Anterior Cervical Decompression and Fusion (For Pattern 3)
Patients with a hypermobile, stenotic single open interspace (Pattern 3) often present with anterior cord compression from disc herniation or osteophyte complexes.
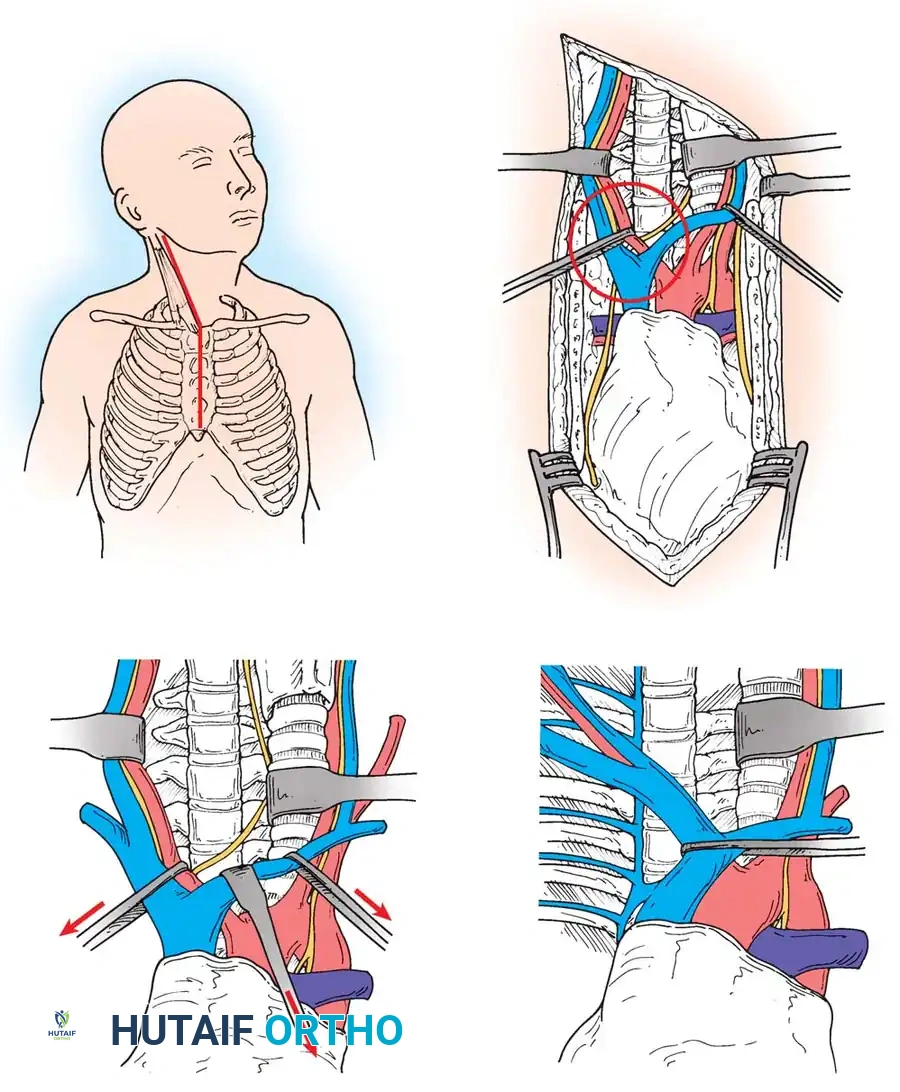
- Approach: A standard Smith-Robinson anterolateral approach is utilized. The dissection proceeds between the carotid sheath laterally and the visceral axis (trachea/esophagus) medially.
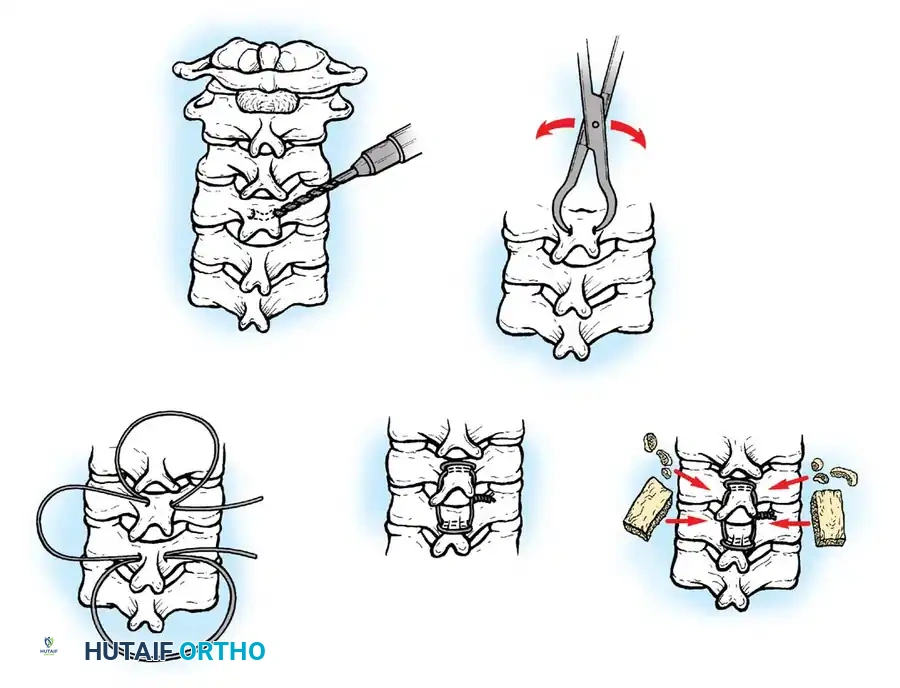
- Discectomy/Corpectomy: The hypermobile disc space is identified fluoroscopically. A complete discectomy is performed. If retrovertebral osteophytes are causing severe cord compression, a partial or complete corpectomy of the adjacent segment may be required.
- Endplate Preparation: The cartilaginous endplates are removed using curettes and a high-speed burr, exposing bleeding subchondral bone to optimize fusion.
- Graft Placement and Plating: A structural allograft, autograft, or titanium cage is impacted into the defect. An anterior cervical plate is then secured with unicortical or bicortical screws to provide immediate biomechanical stability.
Technique 3: Surgical Correction of Sprengel Deformity
For patients with severe cosmetic or functional impairment from an associated Sprengel deformity, surgical relocation of the scapula is indicated, typically performed between 3 and 8 years of age.
- Woodward Procedure: A midline posterior incision is made. The trapezius and rhomboid muscles are detached from the spinous processes.
- Omovertebral Resection: Any omovertebral bone or fibrous band tethering the superomedial angle of the scapula to the cervical spine is extra-periosteally resected.
- Scapular Relocation: The scapula is mobilized and displaced inferiorly to match the contralateral side.
- Muscle Reattachment: The muscular origins are reattached to the spinous processes at a lower, more anatomical level. Care must be taken to avoid stretch injuries to the brachial plexus during inferior displacement.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
Postoperative management is dictated by the surgical approach and the rigidity of the internal fixation.
* Immobilization: Modern rigid screw-rod constructs often require only a hard cervical collar (e.g., Aspen or Miami J) for 6 to 12 weeks. In cases of poor bone quality or non-rigid fixation (e.g., wiring), a Halo vest may be required for up to 3 months.
* Radiographic Follow-up: Upright AP and lateral radiographs are obtained at 2 weeks, 6 weeks, 3 months, and 6 months postoperatively to assess hardware integrity and the progression of arthrodesis.
* Long-Term Surveillance: Because KFS patients have inherently abnormal spinal biomechanics, they require lifelong clinical and radiographic surveillance. Even after successful fusion of a hypermobile segment, the next adjacent open segment is at risk for accelerated degeneration.
PROGNOSIS
The prognosis for Klippel-Feil syndrome is highly variable. Minimally involved patients (Type II with lower cervical fusions) generally lead normal, active lives with no significant restrictions. Conversely, patients with high-risk patterns (Patterns 1, 2, and 3) or severe systemic anomalies require aggressive, multidisciplinary management. Early identification of renal, cardiac, and auditory anomalies, combined with prophylactic stabilization of unstable spinal segments, dramatically improves long-term survivorship and quality of life.
📚 Medical References
- Klippel-Feil syndrome, J Bone Joint Surg 72A:460, 1990.
- Henry AK: Extensile exposure, 2nd ed, Baltimore, 1957, Williams & Wilkins. Itoh T, Tsuji H, Katoh Y, et al: Occipito-cervical fusion reinforced by Luque’s segmental spinal instrumentation for rheumatoid disease, Spine 13:1234, 1988.
- Letts M, Kaylor D, Gouw G: A biomechanical analysis of halo fi xation in children, J Bone Joint Surg 70B:277, 1988.
- Mulpuri K, LeBlanc JG, Reilly CW, et al: Sternal split approach to the cervicothoracic junction in children, Spine 30:E305, 2005.
- Marks DS, Roberts P, Wilton PJ, et al: A halo jacket for stabilisation of the paediatric cervical spine, Arch Orthop Trauma Surg 112:134, 1993.
- McAfee PC, Bohlman HH, Riley LH, et al: The
