Mastering Complex Hand and Upper Extremity Infections: A Comprehensive Surgical Guide
Key Takeaway
Complex hand infections, particularly in intravenous drug users, present severe limb- and life-threatening challenges. Prompt recognition of necrotizing fasciitis, clostridial myonecrosis, and atypical mycobacterial infections is paramount. Successful management mandates aggressive, serial surgical débridement, targeted broad-spectrum antimicrobial therapy, and meticulous physiological support. This guide details the pathophysiology, surgical approaches, and postoperative protocols required to optimize functional outcomes and minimize mortality in these high-risk orthopaedic emergencies.
INFECTIONS IN INTRAVENOUS DRUG ADDICTS
The management of upper extremity infections in intravenous (IV) drug users presents a formidable challenge to the orthopaedic surgeon. The pathophysiology of these infections is intrinsically linked to the mechanical and biological sequelae of repeated, unsterile percutaneous injections. Intravenous drug users are disproportionately colonized with methicillin-resistant Staphylococcus aureus (MRSA), which serves as the primary pathogen in the majority of these cases.
The fundamental lack of asepsis during skin preparation and the injection of particulate-laden, illicit substances account for the high incidence of severe soft tissue compromise. Furthermore, the phenomenon of "skin popping" (subcutaneous extravasation of drugs due to venous sclerosis or missed venipuncture) creates a localized zone of tissue ischemia and necrosis, providing an ideal nidus for bacterial proliferation. Consequently, while systemic septicemia can occur, the infectious burden is typically localized initially, expanding rapidly through compromised fascial planes.
Classification of Hand Infections in Drug Addicts
To systematize surgical decision-making, hand infections in this demographic are categorized into four distinct types based on anatomic depth and structural involvement:
- Type I: Infection confined to the skin and subcutaneous tissues. These typically manifest as localized abscesses or cellulitis, most frequently observed on the dorsum of the fingers or hand where venous access is often attempted.
- Type II: Infection extending to the extensor tendon apparatus, with potential involvement of the underlying periosteum and cortical bone. These may present with localized osteomyelitis or septic extensor tenosynovitis.
- Type III: Infection involving the flexor tendon sheath (suppurative flexor tenosynovitis). This carries the worst functional prognosis due to the risk of rapid tendon necrosis, adhesion formation, and subsequent loss of digital excursion.
- Type IV: Infections complicated by the sequelae of arterial injury. This includes mycotic aneurysms, arterial thrombosis, and subsequent distal gangrene requiring complex vascular reconstruction or amputation.
Necrotizing Fasciitis: Pathophysiology and Clinical Presentation
Necrotizing fasciitis is a rapidly progressive, life-threatening soft tissue infection characterized by widespread necrosis of the superficial fascia with relative sparing of the underlying muscle in its early stages. In IV drug users, the introduction of polymicrobial flora (including anaerobes) into ischemic subcutaneous pockets precipitates this catastrophic infection.
Clinically, the hallmark of necrotizing fasciitis is pain out of proportion to the apparent physical findings. As the infection progresses, the fascia liquefies, producing a characteristic thin, "dishwater," foul-smelling exudate pathognomonic of anaerobic involvement.
Clinical Pearl: The visible cutaneous manifestations—bruising, erythema, and irregular areas of skin necrosis—drastically underestimate the true extent of fascial involvement. The infectious process undermines the skin, spreading rapidly along fascial planes. If left unchecked, myonecrosis follows as bacterial invasion breaches the epimysium.
Proximal spread to the chest wall from an upper extremity source carries a staggering mortality rate of up to 75%. Systemic complications are severe and rapid, including:
* Disseminated intravascular coagulopathy (DIC)
* Septic shock and multiorgan failure
* Metastatic abscess formation (liver, spleen, brain, lungs)
* Thrombosis of digital vessels leading to irreversible gangrene
Surgical Management of Necrotizing Fasciitis
Successful treatment hinges on a high index of suspicion, immediate broad-spectrum antibiotic administration, and emergent, aggressive surgical débridement.
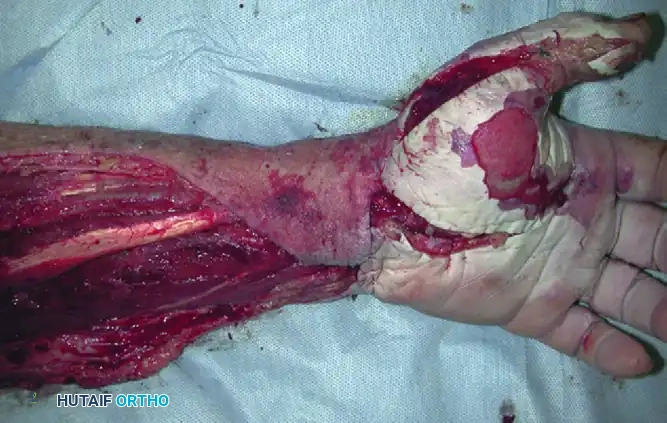
Surgical Approach and Technique:
1. Positioning and Preparation: The patient is positioned supine. A pneumatic tourniquet is applied but exsanguination with an Esmarch bandage is strictly contraindicated to prevent proximal seeding of the infection. The limb is elevated for 3 minutes prior to tourniquet inflation.
2. Incision: Extensile incisions are mandatory. Mid-axial or Brunner zigzag incisions are utilized in the digits, extending proximally into the palm and forearm as dictated by the extent of fascial necrosis. The goal is to preserve viable skin flaps while exposing the entire zone of infection.
3. Débridement: All necrotic tissue must be radically excised. The fascia will often appear dull, gray, and non-bleeding. The "finger sweep" test is highly sensitive; normal fascia resists blunt dissection, whereas necrotic fascia separates easily from the underlying muscle.
4. Specimen Collection: Deep tissue cultures (not superficial swabs) must be sent immediately for aerobic, anaerobic, fungal, and mycobacterial evaluation. Gram stains provide critical early guidance for the infectious disease team.
5. Wound Management: The wound is never closed primarily. It is packed open or managed with negative pressure wound therapy (NPWT) to manage exudate and promote angiogenesis.
6. Serial Débridement: The patient must return to the operating room every 24 to 48 hours for mandatory "second-look" procedures until the infection is completely eradicated and a healthy, granulating wound bed is established.
7. Reconstruction: Once sterility is achieved, delayed closure, split-thickness skin grafting, or regional/free flap coverage is performed. In cases of irreversible necrosis or to control overwhelming systemic sepsis, amputation may be life-saving.
Postoperative Protocol and Medical Management
Empiric antibiotic therapy must be initiated immediately after cultures are obtained. Standard regimens include broad-spectrum coverage for Staphylococcus and Streptococcus (e.g., vancomycin or linezolid for MRSA), high-dose penicillin or clindamycin for anaerobes, and an aminoglycoside (gentamicin) or third-generation cephalosporin for gram-negative organisms.
Surgical Warning: The prevalence of penicillin-resistant organisms necessitates early consultation with an infectious disease specialist to tailor antimicrobial therapy based on regional antibiograms and definitive culture sensitivities.
Nutritional support is paramount. Hyperalimentation (enteral or parenteral) is required to combat the massive catabolic state induced by severe sepsis and to provide the necessary substrates for wound healing. Hyperbaric oxygen (HBO) therapy may serve as a beneficial adjunct, particularly in refractory anaerobic infections, by increasing tissue oxygen tension and enhancing leukocyte bactericidal activity.
GAS GANGRENE (CLOSTRIDIAL MYONECROSIS)
Gas gangrene is a rare but catastrophic infection that poses an immediate threat to both life and limb. While the overall incidence in the United States is estimated at 1,000 to 3,000 cases annually, its rapid onset and profound systemic toxicity demand immediate recognition and radical surgical intervention.
Microbiology and Pathophysiology
Clostridium perfringens (formerly C. welchii) is the most frequently isolated organism, though other clostridial species and nonclostridial organisms (in up to 85% of cases) can produce similar clinical pictures. Clostridia are anaerobic, saprophytic, gram-positive bacilli ubiquitous in soil and human mucous membranes.
These organisms thrive in environments with low oxygen tension—such as necrotic tissue, hematomas, and ischemic muscle. Under these conditions, clostridial spores transition into a vegetative state and release a devastating array of exotoxins:
* Alpha Toxin (Lecithinase): The most lethal toxin, responsible for profound myonecrosis, hemolysis, and platelet destruction.
* Theta Toxin: Causes hemolysis and severe cardiotoxicity.
* Kappa Toxin (Collagenase): Breaks down connective tissue, facilitating rapid fascial spread.
* Nu Toxin (Deoxyribonuclease) & Mu Toxin (Hyaluronidase): Further degrade tissue architecture.
The metabolic byproducts of these organisms include hydrogen sulfide and carbon dioxide, which accumulate in the tissues to produce the characteristic clinical and radiographic sign of gas (crepitus).
Classification of Clostridial Infections
- Type 1 (Clostridial Contamination): Positive bacteriological culture from a wound without clinical signs of systemic infection or local tissue invasion.
- Type 2 (Clostridial Cellulitis): Infection of the subcutaneous tissue producing foul-smelling gas, but sparing the underlying muscle and lacking severe systemic toxicity.
- Type 3 (Clostridial Myonecrosis / Gas Gangrene): Invasive infection of the muscle with profound myonecrosis, massive gas production, and severe systemic septic shock.
Clinical Presentation and Diagnosis
Gas gangrene is classically associated with open fractures, severe crush injuries, soil contamination, and the primary closure of contaminated wounds. However, it can also occur in closed fractures or following elective surgery in immunocompromised patients.
The clinical course is terrifyingly rapid. The earliest symptom is severe, unrelenting pain in the affected limb that is entirely out of proportion to the visible injury. This is rapidly followed by:
* Progressive, tense edema of the limb.
* Tachycardia that is disproportionately high compared to the patient's mild initial fever.
* Development of hemorrhagic skin bullae and blebs.
* A characteristic "sweet and foul" smelling purulent drainage.
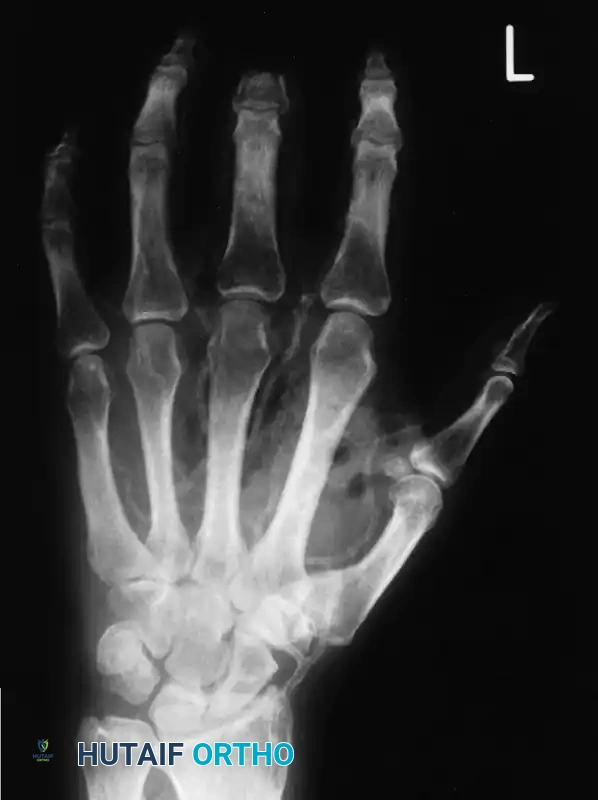
Soft tissue gas may be visible on radiographs before it is clinically palpable. Within 24 hours, palpable crepitus becomes evident. Systemic collapse follows rapidly, characterized by massive intravascular hemolysis, acute renal failure, and refractory septic shock. The mortality rate remains high: 19% overall, and approximately 5% in posttraumatic extremity cases managed optimally.
Surgical and Medical Management
Patients require immediate admission to an intensive care unit (ICU). Endotracheal intubation and aggressive hemodynamic support with vasopressors and massive fluid resuscitation are often required to manage septic shock. Close monitoring for hemolytic anemia and acute kidney injury is mandatory.
Surgical Intervention:
Surgical débridement must be emergent and radical. There is no role for conservative management in established clostridial myonecrosis.
1. Incision and Exposure: Long, extensile incisions are made to decompress all involved fascial compartments.
2. Muscle Assessment: Muscle viability is assessed using the "4 C's": Color, Consistency, Contractility, and Capacity to bleed. Any muscle that is dark, non-contractile, friable, or avascular must be excised entirely.
3. Amputation: In cases of diffuse myonecrosis where limb salvage would leave a non-functional, toxic appendage, emergent guillotine amputation is the treatment of choice to save the patient's life.
4. Wound Care: Wounds are strictly left open. The patient is scheduled for mandatory repeat débridements every 24 hours until the infection is arrested.
High-dose intravenous penicillin remains the antibiotic of choice for C. perfringens, often combined with clindamycin, which halts the ribosomal synthesis of clostridial exotoxins. Hyperbaric oxygen therapy is a valuable adjunct, as high oxygen tensions are directly bactericidal to anaerobic clostridia and halt alpha-toxin production, though it must never delay emergent surgical débridement.
MYCOBACTERIAL INFECTIONS OF THE HAND
While acute bacterial infections dominate the emergency setting, chronic, indolent infections of the hand and wrist are frequently caused by mycobacteria. These require a high index of suspicion, specialized culture techniques, and a combination of radical surgery and prolonged pharmacotherapy.
Tuberculous Infections (Mycobacterium tuberculosis)
Though uncommon in the modern era, M. tuberculosis can manifest in the upper extremity as chronic tenosynovitis, osteomyelitis, septic arthritis, or dactylitis (spina ventosa). It frequently presents as a painless, progressive swelling of the flexor tendon sheaths, historically termed a "compound palmar ganglion."
Compression of the median nerve within the carpal tunnel is a common secondary complication due to the massive proliferation of hypertrophic synovium.
Surgical Management:
When unexplained, chronic tenosynovitis is encountered, tissue must be sent for acid-fast bacilli (AFB) staining, mycobacterial culture, and histological examination (looking for caseating granulomas).
Treatment mandates a radical tenosynovectomy. The flexor retinaculum is released, and the hypertrophic synovium is meticulously excised from the flexor tendons, taking care to preserve the critical A2 and A4 pulleys to prevent bowstringing. If the carpal bones are involved (tuberculous osteomyelitis), aggressive bone débridement and subsequent wrist arthrodesis may be necessary. Surgery is always paired with a prolonged course (6-12 months) of multi-drug antituberculous chemotherapy (Rifampin, Isoniazid, Pyrazinamide, Ethambutol).
Nontuberculous Mycobacterial Infections
The most common nontuberculous mycobacteria (NTM) infecting the hand are Mycobacterium marinum and Mycobacterium kansasii. Infections by M. fortuitum and M. chelonae are less frequent but equally challenging.
Mycobacterium marinum (Swimming Pool Granuloma)
M. marinum is an atypical mycobacterium found in aquatic environments (swimming pools, fish tanks, marine environments). Infection typically occurs when an open wound or minor abrasion is exposed to contaminated water.
Clinically, it presents as a poorly healing ulcer, nodule, or chronic tenosynovitis. Because of its indolent nature and associated joint swelling, it is frequently misdiagnosed as rheumatoid arthritis or gout.
Pitfall: A classic, disastrous clinical scenario involves a patient receiving a corticosteroid injection for presumed inflammatory arthritis or tenosynovitis, which rapidly exacerbates the underlying M. marinum infection, leading to widespread tissue destruction.
Diagnostic Nuances:
Standard bacterial cultures will be negative. M. marinum is a photochromogenic, temperature-sensitive organism. Cultures must be specifically requested and incubated on Lowenstein-Jensen medium at 30°C to 32°C (cooler than standard core body temperature, reflecting the organism's predilection for the cooler extremities).
Mycobacterium kansasii
M. kansasii behaves similarly to M. marinum but is less strictly associated with aquatic exposure. It should be highly suspected in cases of chronic, recurrent synovitis that mimics rheumatoid arthritis but is confined to a single digit or joint.
A hallmark presentation is a patient who undergoes a routine carpal tunnel release for median nerve compression, only to return weeks later with recurrent, massive volar swelling and returning neuropathy due to aggressive mycobacterial synovial proliferation.
Magnetic Resonance Imaging (MRI) is highly sensitive for detecting the extent of tenosynovitis. A classic MRI finding is the presence of "rice bodies" (fibrinous, cartilaginous-like loose bodies within the tendon sheath), which can be radiographically mistaken for synovial chondromatosis.
Surgical and Medical Strategy for NTM
The cornerstone of treatment for NTM infections of the hand is excisional surgery combined with targeted antimicrobial therapy.
- Diagnostic and Therapeutic Synovectomy: Radical excision of the infected synovium reduces the bioburden, decompresses involved nerves, and provides adequate tissue for definitive histological and microbiological diagnosis.
- Culture Protocols: Whenever a chronic, atypical hand infection is aspirated or surgically explored, the surgeon must explicitly order routine bacterial, fungal, and specialized mycobacterial cultures.
- Pharmacotherapy: Because NTM organisms grow slowly, culture results may take up to 6 weeks. Empiric therapy (often involving clarithromycin, ethambutol, and rifampin) may be initiated postoperatively in consultation with an infectious disease specialist. Antimicrobial therapy is typically continued for a minimum of 3 to 6 months following the resolution of clinical symptoms.
You Might Also Like