Sacral Tumor Surgery: Safeguarding Bowel & Bladder Control
Key Takeaway
Your ultimate guide to Sacral Tumor Surgery: Safeguarding Bowel & Bladder Control starts here. Sacral tumors, including primary types like chordoma and more common metastatic ones, often present with insidious, nonspecific symptoms. Surgical management is challenging due to complex anatomy and carries risks such as massive bleeding, wound complications, and neurologic dysfunction, along with potential injury to bowel and bladder. Advanced navigation technology aims to improve resection precision, reduce recurrence, and preserve function.
Introduction and Epidemiology
Tumors involving the sacrum represent a complex intersection of orthopedic oncology and spine surgery. These lesions are broadly categorized into primary and metastatic tumors. Metastatic lesions to the sacrum occur with much greater frequency than primary sacral tumors, frequently originating from breast, prostate, lung, renal, and thyroid carcinomas. The sacrum is also a common site for hematologic malignancies such as multiple myeloma and plasmacytoma.
Among benign primary sacral lesions, the giant cell tumor is the most prevalent. Giant cell tumors often present as large, lytic, and locally aggressive masses capable of significant cortical destruction and soft tissue extension. The underlying pathophysiology involves the overexpression of Receptor Activator of Nuclear Factor Kappa-B Ligand by neoplastic stromal cells, which recruits and activates multinucleated osteoclast-like giant cells, driving aggressive osteolysis.

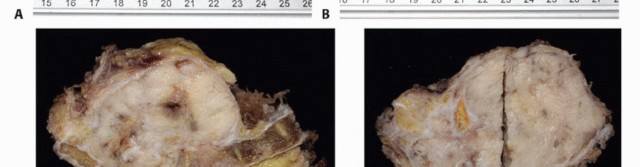
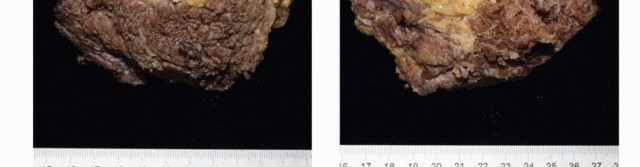
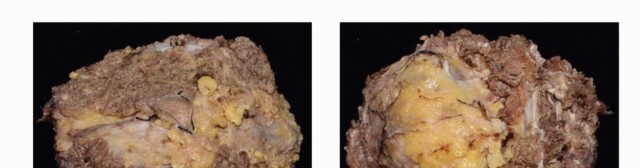
Conversely, the most common primary malignant sacral tumor is the chordoma. Chordomas are low-grade but locally invasive neoplasms arising from notochordal remnants. They are characterized histologically by physaliferous cells and are driven by the overexpression of the brachyury transcription factor. Due to their slow growth rate and relative chemoresistance and radioresistance, en bloc surgical resection with wide margins remains the gold standard for curative intent. Other primary malignancies include chondrosarcoma, osteosarcoma, and Ewing sarcoma, each presenting unique challenges regarding neoadjuvant therapies and surgical timing.

Although schwannomas arise from neural elements rather than the osseous sacrum itself, they are categorized alongside sacral tumors due to their identical clinical presentation, anatomic location within the presacral space, and analogous surgical management principles. Neurofibromas and malignant peripheral nerve sheath tumors must also be considered in the differential diagnosis, particularly in patients with neurofibromatosis type 1.
Clinical Presentation and Diagnostic Challenges
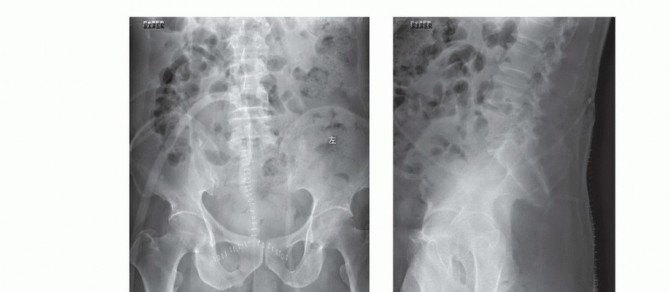
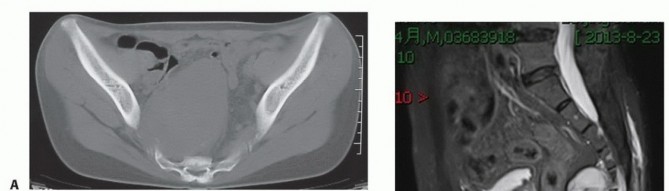
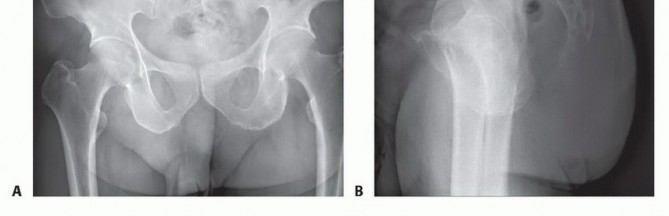
The clinical presentation of sacral tumors is notoriously insidious. Because the presacral space is highly capacious, tumors can achieve massive dimensions before compressing adjacent visceral or neural structures. Symptoms evolve over months to years and are frequently nonspecific, presenting as chronic, dull lower back or coccygeal pain. This insidious onset often leads to an erroneous initial misdiagnosis of lumbar disc herniation, degenerative spine disease, or sacroiliitis.
As the mass effect increases, patients may present with an alteration in bowel or urinary habits. This can manifest initially as constipation or urinary retention due to parasympathetic denervation, eventually progressing to frank fecal or urinary incontinence in severe cases. Advanced high-grade malignancies may precipitate severe intractable pain, ambulatory dysfunction, and fixed posturing due to lumbosacral plexus invasion.

Lower sacral tumors can often be palpated as an anterior mass during a routine digital rectal examination, highlighting the absolute necessity of a thorough physical assessment in any patient presenting with refractory sacrococcygeal pain. Neurological examination must meticulously document perineal sensation, rectal sphincter tone, and lower extremity motor function to establish a baseline prior to any surgical intervention.
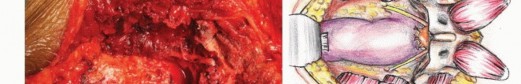
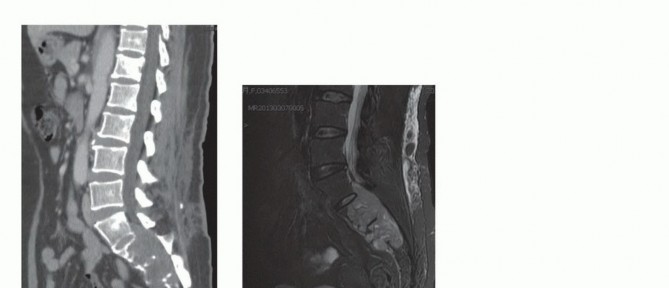
The diagnostic delay is a hallmark of sacral tumors. The average time from symptom onset to definitive diagnosis for sacral chordomas often exceeds twelve months. This delay allows the tumor to expand within the sacral canal, erode through the anterior sacral cortex into the presacral space, and infiltrate the adjacent piriformis, gluteus maximus, and presacral fascia.
Safeguarding bowel and bladder control requires early recognition. The sacral nerve roots, particularly S2 through S4, are intimately involved in the autonomic and somatic innervation of the pelvic floor and sphincters. Tumor encasement of these roots dictates the surgical margins and directly impacts the functional oncologic outcome.
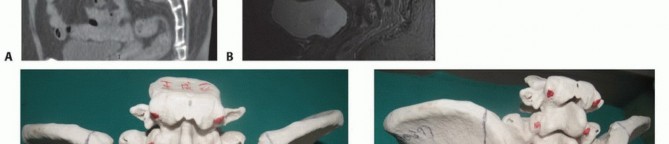
Surgical Anatomy and Biomechanics
A profound understanding of the complex neurovascular and musculoskeletal anatomy of the pelvis is the cornerstone of safe and effective sacral tumor resection. The primary surgical challenge lies in achieving oncologically negative margins while preserving neurologic function and minimizing catastrophic hemorrhage from the presacral venous plexus.
Sacral and Coccygeal Plexuses
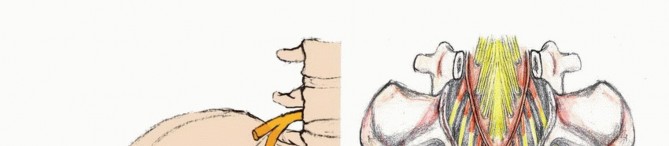
The lumbosacral trunk, comprising the L4 and L5 ventral rami, courses over the sacral ala, combining with the ventral branches of the first three sacral nerves and the upper portion of S4 to form the sacral plexus. The S1 through S3 roots exit through the upper anterior sacral foramina, joining the lumbosacral trunk at the level of the sacroiliac joint. The plexus converges toward the greater sciatic foramen, lying anterior to the sacrum and the piriformis muscle.
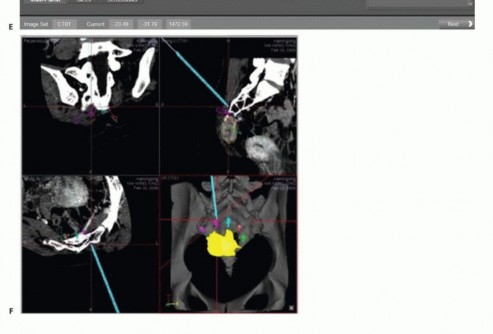
The coccygeal plexus arises from the lower ventral branches of S4, S5, and the coccygeal nerves. This plexus provides anococcygeal filaments that pierce the sacrotuberous ligament to supply the cutaneous region of the coccyx. Understanding this plexus is critical when performing lower sacrectomies or coccygectomies, as resection will result in predictable perianal numbness.
The preservation of bowel and bladder function hinges on the integrity of the S2 through S4 nerve roots. Bilateral resection of S2, S3, and S4 universally results in total loss of voluntary bowel and bladder control, necessitating lifelong catheterization and a bowel management program. Preservation of at least one S3 nerve root unilaterally can salvage partial sphincter control in a significant subset of patients, although normal function is rarely retained.
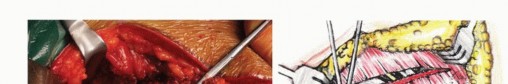
Critical Nerve Branches and Visceral Innervation
The superior gluteal nerve originates from L4 through S1, exiting the pelvis via the suprapiriformis foramen. It provides critical motor innervation to the tensor fasciae latae, gluteus minimus, and gluteus medius. Injury to this nerve during the posterior approach or iliac osteotomies results in a profound Trendelenburg gait.
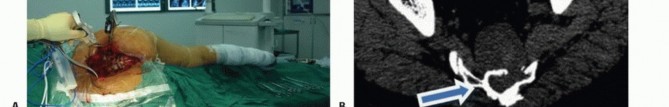
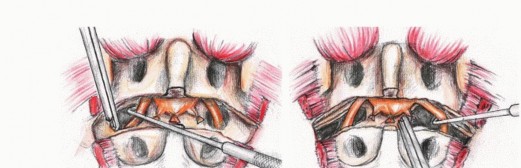
The inferior gluteal nerve originates from L5 through S2, exiting via the infrapiriformis foramen to supply the gluteus maximus. The pudendal plexus, derived from S2 through S4, is the absolute key to sphincter control. It courses posterior to the ischial spine, enters the perineum through the lesser sciatic foramen, and travels along the obturator internus in Alcock's canal. It yields muscular branches to the levator ani and external anal sphincter, alongside visceral branches to the bladder, rectum, and genitalia.
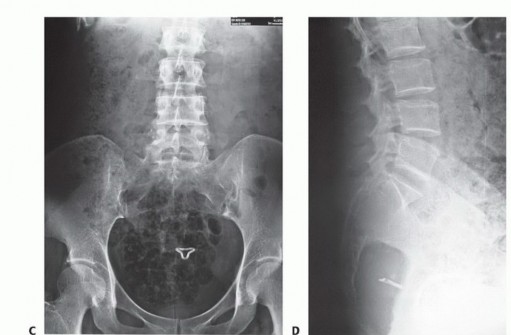
The sciatic nerve, comprising L4 through S3 contributions, exits via the suprapiriformis foramen, lying posterior to the short external rotators. Radical total sacrectomy denervates the lower extremities in the sciatic distribution, resulting in flaccid paralysis below the knee and loss of hamstring function, though quadriceps function remains intact via the femoral nerve. The dural sac typically terminates at the S2-S3 junction, a critical landmark during laminectomy and thecal sac ligation.

Vascular Anatomy and Presacral Hemorrhage Risks
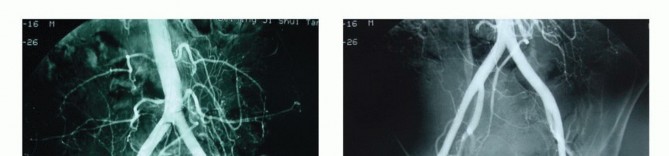
The vascular supply to the sacrum and presacral space is robust and represents a major source of intraoperative morbidity. The median sacral artery arises from the posterior aspect of the distal abdominal aorta, descending vertically anterior to the L5 vertebra and the sacral promontory. It must be identified and ligated early during an anterior approach.
The lateral sacral arteries, branches of the posterior division of the internal iliac arteries, enter the anterior sacral foramina to supply the sacral canal and neural elements. The presacral venous plexus, or Batson's plexus, is a valveless, highly anastomotic network of veins lying between the presacral fascia and the anterior sacral periosteum. Disruption of this plexus during blunt presacral dissection can result in massive, intractable hemorrhage, as these veins easily retract into the sacral osseous foramina where conventional hemostatic techniques fail.
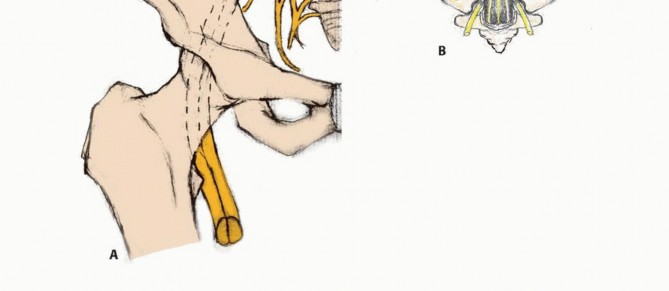
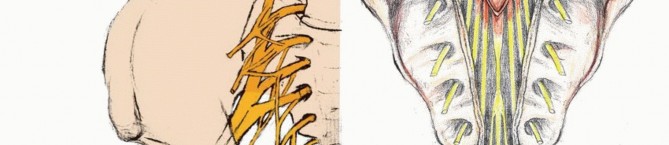
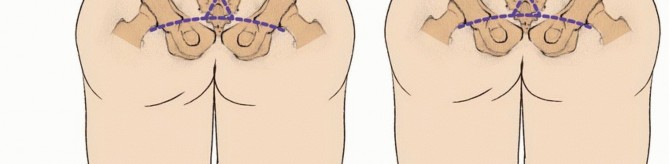
Pelvic Ring Biomechanics
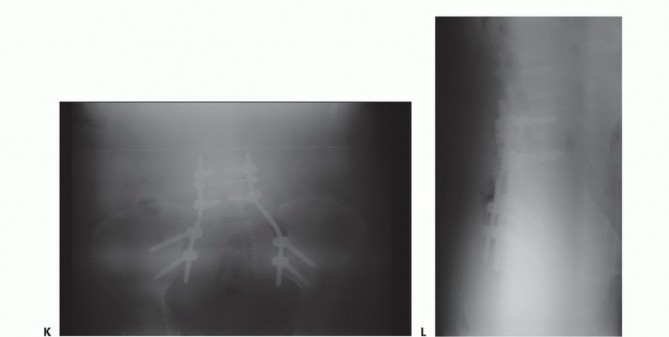
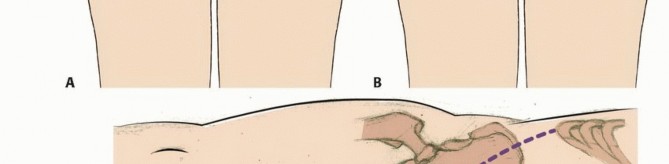
The sacrum serves as the keystone of the pelvic ring, facilitating load transfer from the axial skeleton to the appendicular skeleton via the sacroiliac joints. The strong posterior sacroiliac ligaments are the primary stabilizers of the pelvic ring. Resection of the sacrum above the S1-S2 disc space severely compromises pelvic stability.
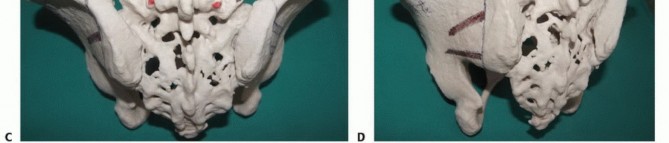
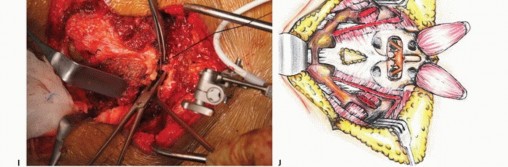
A total sacrectomy results in complete spinopelvic dissociation. In these scenarios, the lumbar spine is entirely disconnected from the iliac wings, necessitating complex, rigid spinopelvic reconstruction to restore weight-bearing capacity and prevent catastrophic superior migration of the pelvis. Even in partial high sacrectomies where less than fifty percent of the sacroiliac joint is preserved bilaterally, prophylactic spinopelvic fixation is heavily indicated to prevent delayed mechanical failure and intractable mechanical back pain.

Indications and Contraindications
The decision to proceed with surgical resection of a sacral tumor requires a meticulous balancing of oncologic principles against the anticipated functional morbidity. The Enneking staging system for benign and malignant musculoskeletal tumors, alongside the Weinstein-Boriani-Biagini classification for spine tumors, guides the surgical strategy.
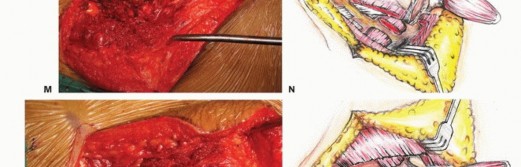
Primary malignant tumors, particularly chordomas and chondrosarcomas, require en bloc wide resection. Intralesional curettage of these pathologies results in near-universal local recurrence with devastating consequences. Conversely, giant cell tumors may be treated with aggressive intralesional extended curettage, high-speed burring, and the application of local adjuvants such as phenol, liquid nitrogen, or argon beam coagulation, combined with systemic denosumab therapy.
Metastatic disease to the sacrum is generally managed non-operatively with palliative radiation therapy, systemic chemotherapy, and bisphosphonates. However, surgical intervention is indicated for solitary renal cell or thyroid metastases where wide resection can be curative, or in cases of intractable pain, impending skin breakdown, or progressive neurological deficit refractory to non-operative measures.
Contraindications to radical sacrectomy include widespread metastatic disease, severe medical comorbidities precluding prolonged anesthesia, and tumor extension that renders negative margins impossible without unacceptable morbidity. Extensive bilateral invasion of the sciatic nerves, massive intrapelvic extension encasing the external iliac vessels, or gross peritoneal seeding generally preclude curative en bloc resection.
Operative Versus Non Operative Decision Matrix
| Clinical Scenario | Recommended Management Strategy | Rationale and Oncologic Goal |
|---|---|---|
| Solitary Sacral Chordoma | En Bloc Wide Resection (Sacrectomy) | Radioresistant tumor; wide margins offer the only chance for long-term disease-free survival. |
| Giant Cell Tumor (Campanacci Grade III) | Neoadjuvant Denosumab followed by Extended Curettage or Resection | Downstaging the tumor reduces intraoperative hemorrhage and joint morbidity; curettage preserves nerve roots. |
| Widespread Metastatic Carcinoma | Palliative Radiation and Systemic Therapy | Surgery does not improve overall survival; focus is on palliation and quality of life. |
| Solitary Renal Cell Carcinoma Metastasis | Pre-operative Embolization and En Bloc Resection | Highly vascular tumor; solitary metastasis resection can significantly prolong survival. |
| Ewing Sarcoma of the Sacrum | Neoadjuvant Chemotherapy, Radiation, and possible Resection | Highly chemosensitive and radiosensitive; surgery reserved for residual disease or local control. |
| Progressive Sphincter Dysfunction in Benign Lesion | Urgent Surgical Decompression | Prevent irreversible ischemia and permanent loss of bowel/bladder function. |
Pre Operative Planning and Patient Positioning
Preoperative planning for sacral tumor surgery is an exhaustive process requiring a multidisciplinary tumor board approach. The surgical team typically includes orthopedic oncologists, spine surgeons, colorectal surgeons, urologists, neurosurgeons, and plastic surgeons.
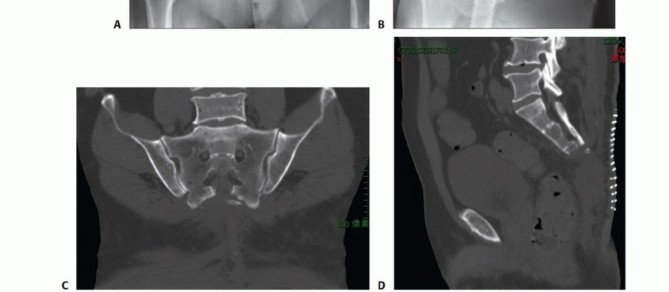
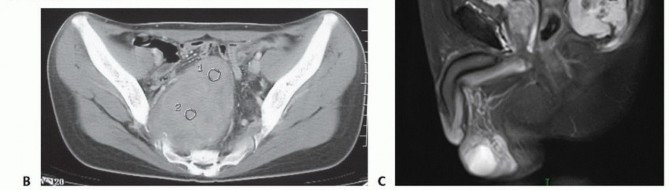
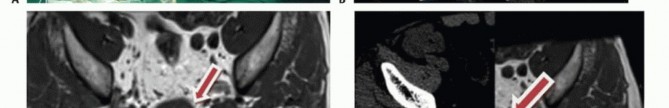
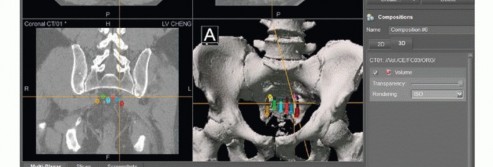
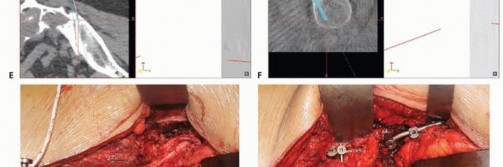
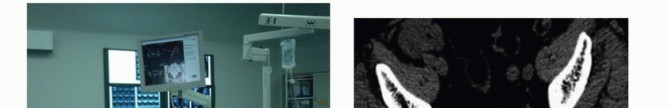
Advanced imaging is mandatory. Magnetic resonance imaging with and without gadolinium contrast is the gold standard for defining the soft tissue extent of the tumor, epidural extension, and involvement of the sacral nerve roots. The relationship of the anterior tumor margin to the mesorectum, presacral fascia, and iliac vessels must be mapped meticulously. Computed tomography is essential for delineating cortical destruction, planning osteotomy trajectories, and designing custom 3D-printed cutting guides or implants.
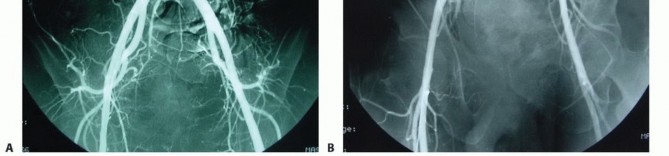
Diagnostic angiography with selective preoperative embolization is highly recommended for hypervascular tumors such as giant cell tumors, renal cell carcinoma metastases, and thyroid metastases. Embolization of the internal iliac artery branches and median sacral artery within twenty-four to forty-eight hours prior to surgery significantly reduces intraoperative blood loss.

Bowel preparation is critical, as the risk of accidental rectal injury during the anterior dissection or osteotomy phase is significant. Prophylactic ureteral stenting performed by urology facilitates intraoperative identification of the ureters, preventing iatrogenic transection during the mobilization of the retroperitoneal structures.
Patient Positioning and Intraoperative Monitoring
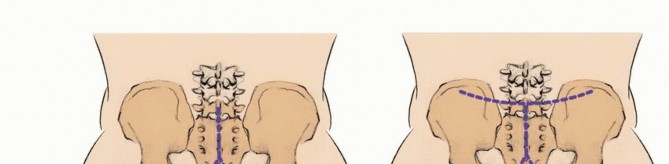
The surgical approach dictates patient positioning. A low sacrectomy (below S3) can often be performed entirely from a posterior approach with the patient in the prone position. High sacrectomies or total sacrectomies require a combined anterior-posterior approach. This can be performed sequentially (supine followed by prone) or simultaneously in the lateral decubitus position, depending on surgeon preference and institutional experience.
When prone, the patient is typically positioned on an open Jackson frame to allow the abdomen to hang freely, reducing intra-abdominal pressure and consequently decreasing epidural venous engorgement. Careful padding of all bony prominences is mandatory due to the prolonged operative time, which frequently exceeds twelve hours.
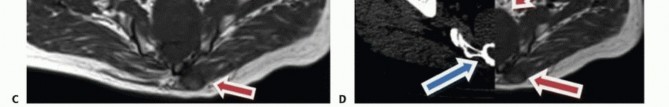
Multimodal intraoperative neuromonitoring is indispensable. Somatosensory evoked potentials and transcranial motor evoked potentials assess the integrity of the spinal cord and lumbosacral plexus. Critically, continuous and triggered electromyography of the external anal sphincter and urethral sphincter is utilized to identify and map the S2-S4 nerve roots during the laminectomy and neural dissection phases. This real-time feedback is vital for maximizing the preservation of continence.
Detailed Surgical Approach and Technique
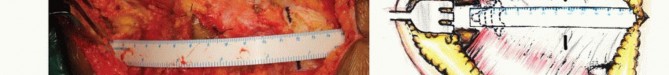
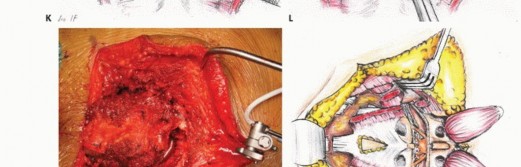
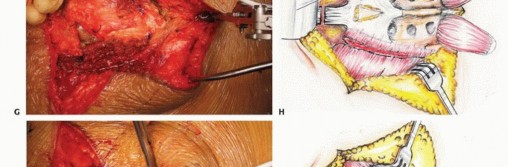
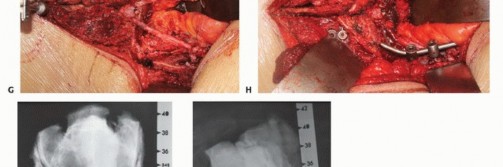
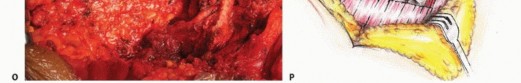
The surgical execution of a radical sacrectomy for a primary malignant tumor is divided into distinct phases. Achieving an en bloc resection requires navigating complex internervous and intermuscular planes while maintaining a continuous cuff of normal tissue around the neoplasm.
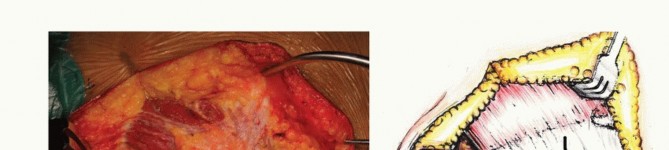
Anterior Dissection and Visceral Mobilization
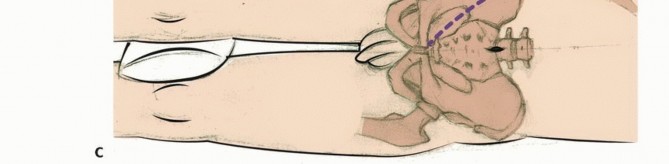
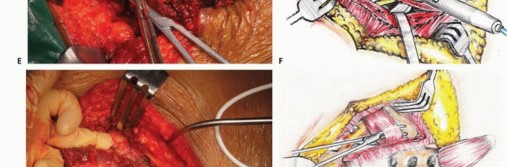
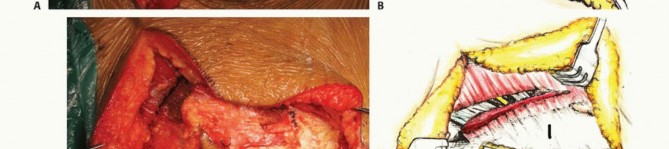
For high sacral tumors, the operation frequently begins with an anterior abdominal approach. A lower midline laparotomy is performed. The parietal peritoneum is incised, and the retroperitoneal space is accessed. The ureters, previously stented, are identified and retracted laterally.
The bifurcation of the aorta and the inferior vena cava are exposed. The median sacral artery and vein are identified, ligated, and divided. The internal iliac arteries and veins are traced distally. Depending on the lateral extent of the tumor, the internal iliac vessels may need to be ligated to permit safe mobilization of the pelvic viscera and to devascularize the tumor bed.
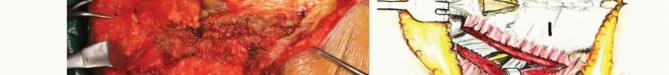
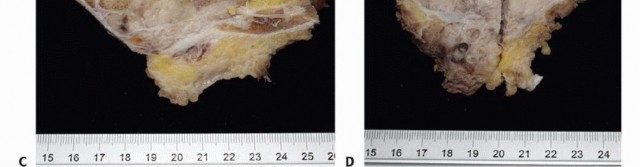
The rectum is mobilized anteriorly. The dissection plane must remain strictly extrafascial, anterior to the presacral fascia, to avoid entering the tumor pseudocapsule. The mesorectum is carefully separated from the anterior sacrum down to the pelvic floor. Once the anterior sacrum is cleared, laparotomy pads or a rigid silastic sheet are placed into the presacral space to protect the viscera during the subsequent posterior osteotomies. The abdomen is then closed, and the patient is repositioned prone.






You Might Also Like