Resolve Radius or Ulna Nonunions: Top Surgical Treatments
Key Takeaway
Learn more about Resolve Radius or Ulna Nonunions: Top Surgical Treatments and how to manage it. A diaphyseal nonunion of the forearm occurs when a fracture of the radius or ulna fails to heal within 6 months. These nonunions are classified as hypertrophic, characterized by abundant callus, or atrophic, with poor blood supply and little callus. While rare due to modern fixation, they commonly result from inadequate treatment or fixation.
Introduction and Epidemiology
A diaphyseal forearm fracture is strictly defined as a nonunion when clinical and radiographic healing has failed to occur within six months of the index injury, or when serial imaging demonstrates no progression of healing over three consecutive months. While the advent of modern dynamic compression plating and strict adherence to Arbeitsgemeinschaft für Osteosynthesefragen (AO) principles have made diaphyseal forearm nonunions relatively rare, they remain a formidable challenge when they do occur. Current literature reports nonunion rates of approximately two percent for the radius and four percent for the ulna following primary open reduction and internal fixation.
Nonunions are broadly classified into two distinct physiologic categories based on vascularity and osteogenic potential. Hypertrophic nonunions are characterized by abundant callus formation and a rich vascular supply. They represent a failure of mechanical stability rather than a failure of biology. In the forearm diaphysis, hypertrophic nonunions are exceedingly rare, comprising less than ten percent of all nonunion cases. Atrophic nonunions, conversely, are characterized by a lack of callus formation, sclerotic bone ends, and poor local vascularity. These lesions represent a dual failure of both mechanical stability and local biology, necessitating surgical strategies that address both deficiencies.
The pathogenesis of diaphyseal forearm nonunions is multifactorial but predominantly iatrogenic, resulting from technical errors during the index procedure. Inadequate fixation, specifically the failure to obtain a minimum of six cortices of screw purchase proximal and distal to the fracture site, significantly elevates the risk of nonunion. Furthermore, failure to achieve absolute stability via interfragmentary compression or dynamic compression plating allows for micromotion that exceeds the strain tolerance for primary bone healing.
Biologic factors also play a critical role. Comminution drastically increases the risk of nonunion, with studies demonstrating a twelve percent nonunion rate in comminuted diaphyseal fractures treated with dynamic compression plates. Extensive periosteal stripping during the initial exposure, severe soft tissue devascularization, high-energy open fractures with segmental bone loss, and deep surgical site infections are primary local risk factors. Systemic patient comorbidities, including poorly controlled diabetes mellitus, chronic corticosteroid use, profound malnutrition, smoking, and renal osteodystrophy, further compromise the osteogenic cascade and increase the probability of nonunion.
Surgical Anatomy and Biomechanics
Osteology and Articulations
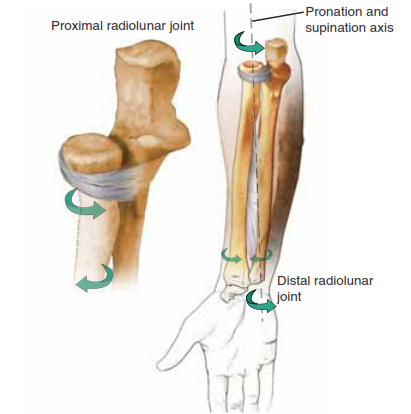
The forearm must be conceptualized not as two isolated long bones, but as a complex, highly constrained bi-articular joint. The radius and ulna function as a unified kinematic chain, articulating proximally at the proximal radioulnar joint (PRUJ) and distally at the distal radioulnar joint (DRUJ). Pronation and supination are achieved through the complex rotation of the morphologically curved radius around the relatively linear axis of the stationary ulna.

The ulna serves as the stable post of the forearm, possessing a relatively straight diaphyseal geometry. In contrast, the radius exhibits a complex three-dimensional morphology characterized by an apex radial bow and an apex dorsal bow. The precise restoration of this radial curvature is paramount during nonunion surgery; failure to restore the radial bow diminishes the interosseous space, leading to mechanical impingement and a profound loss of rotational motion. A loss of the normal radial bow by as little as five degrees can result in a measurable decrease in pronosupination.
Interosseous Membrane and Soft Tissues
The interosseous membrane (IOM) spans the interval between the radius and ulna, compartmentalizing the forearm and providing critical longitudinal stability. The central band of the IOM is a robust ligamentous structure that originates on the proximal radius and inserts on the distal ulna, effectively transferring axial loads from the radiocarpal joint to the radiocapitellar and ulnohumeral joints.
In the setting of a chronic nonunion with associated bone shortening, the IOM undergoes contracture and fibrosis. If significant shortening of either the radius or ulna is left uncorrected, the intricate anatomy of the forearm joint is disrupted. Malalignment of the DRUJ secondary to radial shortening results in ulnocarpal impaction, chronic pain, and severe loss of wrist kinematics. Furthermore, chronic pain limits pronation and supination, exacerbating IOM fibrosis and leading to permanent rotational deficits even if osseous union is eventually achieved.
Surgical Approaches and Internervous Planes
The diaphyseal portions of the radius and ulna are enveloped by complex neurovascular anatomy that dictates surgical exposure.
The volar approach to the radius (Henry approach) utilizes the internervous plane between the brachioradialis (innervated by the radial nerve) and the flexor carpi radialis (innervated by the median nerve). Proximally, the radial artery and its recurrent branches (the leash of Henry) must be carefully mobilized or ligated to access the supinator muscle, which is incised to expose the proximal radius. The posterior interosseous nerve (PIN) lies within the substance of the supinator and is at significant risk during proximal radial exposures.
The dorsal approach to the radius (Thompson approach) exploits the internervous plane between the extensor carpi radialis brevis (radial nerve) and the extensor digitorum communis (posterior interosseous nerve). This approach provides excellent access to the middle and proximal thirds of the radius but places the PIN at higher risk as it exits the supinator muscle.
The approach to the ulnar diaphysis is relatively straightforward, utilizing the subcutaneous border of the ulna. The internervous plane lies between the extensor carpi ulnaris (posterior interosseous nerve) and the flexor carpi ulnaris (ulnar nerve). While neurologically safe, the thin soft tissue envelope over the ulna demands meticulous handling to prevent wound breakdown and hardware prominence.
Indications and Contraindications
Surgical intervention is the definitive standard of care for diaphyseal forearm nonunions. Once a nonunion is established, spontaneous healing will not occur. Nonoperative management is universally associated with progressive deformity, hardware failure, chronic pain, and irreversible loss of upper extremity function.
| Parameter | Operative Management | Non Operative Management |
|---|---|---|
| Primary Indications | Aseptic atrophic nonunion, aseptic hypertrophic nonunion, septic nonunion (staged approach), hardware failure, progressive deformity, symptomatic pseudoarthrosis. | Absolute medical contraindication to anesthesia, non-ambulatory patient with painless pseudoarthrosis and minimal functional demand (exceedingly rare). |
| Contraindications | Active untreated severe systemic infection, unstable medical comorbidities precluding anesthesia. | Almost all diaphyseal forearm nonunions in functional adults. |
| Timing | Elective, following thorough metabolic workup and preoperative planning. Septic cases require immediate hardware removal and debridement. | N/A |
| Expected Outcomes | High union rates (over 90%) with modern plating and grafting techniques. Restoration of functional ROM. | Progressive bone loss, hardware breakage, severe pain, loss of pronosupination. |
Pre Operative Planning and Patient Positioning
Clinical and Radiographic Evaluation
Comprehensive preoperative planning begins with a meticulous clinical examination. The surgeon must assess the existing surgical incisions, the quality of the soft tissue envelope, and the presence of any underlying nerve deficits. Range of motion at the elbow, wrist, and forearm must be documented.
High-quality, orthogonal anteroposterior and lateral radiographs of the entire forearm, including the elbow and wrist joints, are mandatory. These images allow the surgeon to assess the nonunion morphology (atrophic vs. hypertrophic), the extent of bone loss, the presence of hardware failure, and the alignment of the PRUJ and DRUJ.

Computed tomography (CT) scanning with multiplanar reconstruction is highly recommended for preoperative templating. CT provides precise quantification of segmental bone defects, identifies subtle intramedullary sclerosis, and helps determine the exact dimensions of structural bone grafts that may be required. If an occult infection is suspected, magnetic resonance imaging (MRI) or a tagged white blood cell scan may be utilized, though their utility is often limited by metallic artifact from retained hardware.
Metabolic and Laboratory Workup
A rigorous metabolic evaluation is critical for identifying reversible systemic causes of nonunion. Routine laboratory investigations should include a complete blood count (CBC), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) to screen for subclinical infection.
To optimize the biologic milieu, surgeons must evaluate the patient's nutritional and endocrine status. A comprehensive metabolic panel, serum 25-hydroxyvitamin D, intact parathyroid hormone (PTH), calcium, phosphorus, and thyroid-stimulating hormone (TSH) should be obtained. Vitamin D deficiency is highly prevalent in nonunion populations and must be aggressively replenished prior to elective nonunion takedown. In patients with diabetes, a hemoglobin A1c level is required to ensure adequate glycemic control, as hyperglycemia severely impairs osteoblast function and microvascular perfusion.
Operating Room Setup
The patient is positioned supine on the operating table with the affected extremity extended onto a radiolucent hand table. A non-sterile pneumatic tourniquet is applied high on the brachium. The iliac crest must be prepped and draped into the sterile field if autogenous bone grafting is anticipated.
Fluoroscopy is positioned opposite the surgeon, allowing for unhindered intraoperative imaging. The surgical team must ensure that a comprehensive hardware removal set is available, including broken screw extractors, metal-cutting burrs, and trephines, as retained and integrated hardware frequently complicates nonunion takedowns.
Detailed Surgical Approach and Technique
Exposure and Hardware Removal
The surgical approach is dictated by the location of the nonunion and the placement of prior incisions. Whenever possible, previous surgical incisions should be utilized to minimize soft tissue devascularization. For the radius, the volar Henry approach is preferred for middle and distal third nonunions, while the dorsal Thompson approach may be utilized for proximal third lesions. The ulnar approach follows the subcutaneous border.
Meticulous soft tissue dissection is paramount. The periosteum should be elevated only as much as necessary to expose the nonunion site and apply the new fixation construct. Extensive periosteal stripping will further compromise the already tenuous blood supply of an atrophic nonunion.
Hardware removal is often the most technically demanding portion of the procedure. Integrated plates and cold-welded screws require patience and specialized extraction tools. If a screw head is stripped, a conical extraction device or a hollow reamer (trephine) may be necessary. Care must be taken to preserve as much host bone stock as possible during hardware extraction to prevent iatrogenic fracture or the creation of a larger segmental defect.
Nonunion Takedown and Preparation
Once the hardware is removed, the nonunion site is systematically addressed. In the setting of an atrophic nonunion, the fibrous pseudoarthrosis tissue must be radically excised until healthy, bleeding cortical bone is encountered (the "paprika sign").

The sclerotic bone ends are resected using a microsaw or sharp osteotomes. It is critical to open the medullary canal on both the proximal and distal segments. This is achieved using sequential drill bits or sharp curettes. Re-establishing medullary patency is essential for restoring endosteal blood flow and allowing the migration of osteoprogenitor cells into the nonunion gap.
Following canal preparation, the adjacent cortical bone is decorticated using a sharp osteotome. This technique, often referred to as "shingling" or "petal" elevation, increases the surface area for bone graft incorporation and stimulates a regional acceleratory phenomenon to jump-start the healing cascade.
Bone Grafting Techniques
The management of the osseous defect depends entirely on its size and the mechanical stability required.
For hypertrophic nonunions, where biology is robust, bone grafting is rarely necessary. The treatment focuses entirely on achieving rigid mechanical stability, typically through compression plating, which reliably leads to union.
For atrophic nonunions, biologic augmentation is mandatory. If the nonunion takedown results in intimate cortical contact without shortening, autogenous cancellous bone graft harvested from the anterior or posterior iliac crest is packed circumferentially around the decorticated site. Cancellous autograft remains the gold standard, providing osteogenic cells, osteoinductive growth factors, and an osteoconductive scaffold.

If radical debridement of sclerotic bone results in a segmental defect greater than 1 to 2 centimeters, or if radial/ulnar length must be restored to correct DRUJ impingement, a structural graft is required. A tricortical iliac crest bone graft (ICBG) is meticulously shaped to fit the defect precisely. The structural graft restores longitudinal length, re-establishes the radial bow, and provides immediate mechanical support to the fixation construct. In cases of massive bone loss where autograft is insufficient or contraindicated, structural fibular allografts supplemented with autogenous bone marrow aspirate concentrate (BMAC) or recombinant human bone morphogenetic proteins (rhBMP-2) may be utilized.
Plate Application and Fixation Principles
The cornerstone of diaphyseal nonunion surgery is the application of rigid internal fixation. The AO principles of anatomic reduction, stable fixation, preservation of blood supply, and early mobilization must be rigorously applied.
Standard 3.5-millimeter dynamic compression plates (DCP), limited contact dynamic compression plates (LC-DCP), or locking compression plates (LCP) are the implants of choice. The plate should be of sufficient length to allow for a minimum of eight cortices (four screws) of secure purchase in healthy bone both proximal and distal to the nonunion site. In osteoporotic bone or revision settings with multiple empty screw holes, locking plates offer superior pull-out strength and construct stability.

For oblique nonunions, an interfragmentary lag screw should be placed to achieve absolute stability, followed by a neutralization plate. For transverse hypertrophic nonunions, the plate is applied in compression mode using eccentrically drilled screws to compress the nonunion interface.
When bridging a structural graft in an atrophic nonunion, the plate functions as a bridge plate. It is crucial to avoid placing screws through the structural graft if it compromises the graft's integrity, though a single position screw can occasionally be used to secure the graft within the defect.
Following plate application, final fluoroscopic images are obtained to confirm the restoration of the radial bow, the precise alignment of the DRUJ, and the appropriate positioning of all hardware.

Wound closure must be performed with careful attention to compartment pressures. The deep fascia is routinely left open to prevent postoperative compartment syndrome. The subcutaneous tissues and skin are closed in a tension-free manner over a closed suction drain, which is typically removed on postoperative day one.
Complications and Management
The surgical management of diaphyseal forearm nonunions carries a distinct complication profile due to the compromised local biology, altered anatomy, and the necessity for extensive surgical dissection.
| Complication | Incidence | Etiology and Pathophysiology | Prevention and Salvage Management |
|---|---|---|---|
| Persistent Nonunion | 5-10% | Inadequate mechanical stability, failure to address metabolic deficiencies, insufficient bone grafting, occult infection. | Prevention: Rigid fixation (8 cortices per segment), robust autografting, strict metabolic optimization. Salvage: Revision plating, structural grafting, and consideration of biologic adjuvants (rhBMP-2). |
| Deep Surgical Site Infection | 2-5% | Poor soft tissue envelope, prolonged operative time, preexisting subclinical infection. | Prevention: Meticulous soft tissue handling, prophylactic antibiotics, tension-free closure. Salvage: Radical serial debridement, hardware removal if unstable, placement of antibiotic spacers, culture-directed systemic antibiotics, and delayed reconstruction. |
| Hardware Failure | 3-7% | Fatigue failure of the plate or screws due to persistent motion at the nonunion site prior to osseous consolidation. | Prevention: Use of appropriately sized 3.5mm plates, avoiding short plates, ensuring structural support for large defects. Salvage: Hardware extraction, re-debridement of the nonunion, placement of a longer, thicker plate, and supplemental bone grafting. |
| Radioulnar Synostosis | 2-4% | Heterotopic ossification bridging the radius and ulna, often due to simultaneous approaches or severe trauma to the interosseous membrane. | Prevention: Avoid operating on both bones through a single incision. Meticulous hemostasis. Salvage: Delayed surgical excision of the synostosis (after 6-12 months of maturation), followed by prophylactic radiation therapy or indomethacin. |
| Posterior Interosseous Nerve (PIN) Palsy | 1-3% | Iatrogenic traction, compression, or transection during the proximal radial exposure (Henry or Thompson approach). | Prevention: Careful dissection, avoiding excessive retraction on the supinator muscle, avoiding blind placement of retractors on the radial neck. Salvage: Observation for neurapraxia (EMG at 6 weeks if no recovery). Tendon transfers if permanent paralysis occurs. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol must be carefully tailored to the intraoperative findings, specifically the rigidity of the fixation and the quality of the host bone.
In the immediate postoperative period, the upper extremity is immobilized in a bulky Jones dressing with a well-padded sugar-tong splint to control edema and provide soft tissue rest. The extremity is strictly elevated.
At the first postoperative visit (typically 10 to 14 days), the splint and sutures are removed. If rigid internal fixation was achieved and bone quality is adequate, early active and active-assisted range of motion of the elbow, wrist, and forearm is initiated. Early mobilization is critical to prevent capsular contracture, minimize IOM fibrosis, and optimize cartilage nutrition at the radiocapitellar and radioulnar joints.
Patients are instructed to maintain strict non-weight-bearing precautions on the operative extremity. Lifting is restricted to less than one pound (the weight of a coffee cup). Passive stretching and aggressive physical therapy are generally avoided until early clinical and radiographic signs of union are evident, typically around 6 to 8 weeks postoperatively.
Serial radiographs are obtained at 6 weeks, 12 weeks, and 6 months to monitor the progression of bone healing and the incorporation of structural grafts. Progressive resistance exercises and gradual return to weight-bearing are permitted only after radiographic consolidation is confirmed, which may take 4 to 6 months in the setting of complex atrophic nonunions treated with structural grafting.
Summary of Key Literature and Guidelines
The evolution of surgical treatment for diaphyseal forearm nonunions is deeply rooted in the biomechanical principles established by the AO Foundation. Historical literature clearly demonstrated that nonoperative management or intramedullary fixation of forearm nonunions resulted in unacceptably high failure rates due to the inability to control rotational forces.
The seminal work by Anderson et al. established compression plating as the gold standard for diaphyseal forearm fractures, a principle that translates directly to nonunion management. Their studies highlighted that rigid fixation with 3.5-millimeter plates effectively neutralizes bending and torsional forces, providing the absolute stability required for primary bone healing.
Ring, Jupiter, and colleagues have contributed extensively to the modern understanding of forearm nonunions. Their retrospective analyses emphasized the critical distinction between atrophic and hypertrophic nonunions. They demonstrated that while hypertrophic nonunions consistently heal with the application of rigid compression plating alone, atrophic nonunions demand a combined approach of mechanical stabilization and biologic augmentation. Their work validated the use of structural iliac crest bone grafts for restoring radial length and correcting DRUJ malalignment, reporting union rates exceeding 90% when these principles are strictly followed.
Recent biomechanical and clinical studies have evaluated the utility of locking compression plates (LCP) in the revision setting. While standard non-locking plates remain highly effective for the majority of nonunions, guidelines now suggest that locking plates provide superior construct stiffness in osteoporotic bone or in revision scenarios where host bone stock is compromised by multiple empty screw tracts. Furthermore, emerging literature supports the adjunctive use of orthobiologics, such as rhBMP-2 and BMAC, in recalcitrant nonunions or massive segmental defects, though autogenous iliac crest bone graft remains the gold standard against which all other biologic therapies are measured.
In summary, the successful resolution of radius or ulna nonunions requires a profound understanding of forearm kinematics, meticulous preoperative planning, precise execution of AO fixation principles, and the judicious application of biologic grafts to restore both the mechanical integrity and the complex rotational function of the upper extremity.
You Might Also Like

