Operative Management of Sacral Fractures and Acute Spinal Cord Injuries
Key Takeaway
Transverse sacral fractures with neurologic compromise require meticulous evaluation and urgent surgical intervention. This guide details the biomechanics, imaging protocols, and step-by-step surgical techniques for sacral laminectomy and decompression. Furthermore, it synthesizes current evidence-based protocols for acute spinal cord injury management, including the controversial role of high-dose methylprednisolone, ensuring optimal neurologic recovery and spinopelvic stability.
Introduction to Spinopelvic Trauma and Neurologic Compromise
High-energy trauma to the axial skeleton frequently results in complex fracture patterns that threaten both mechanical stability and neurologic integrity. Among the most challenging of these injuries are transverse sacral fractures and spinopelvic dissociations. These injuries are often accompanied by severe neurologic deficits, including cauda equina syndrome, sacral nerve root transection, and profound sphincter dysfunction.
The management of acute spinal cord injury (SCI) and sacral root trauma requires a multidisciplinary approach, grounded in a thorough understanding of spinal biomechanics, advanced imaging, and meticulous surgical technique. Historically, the treatment algorithms for these injuries have evolved significantly, transitioning from prolonged conservative bed rest to aggressive surgical decompression and rigid lumbopelvic fixation.
This masterclass synthesizes the foundational principles of acute spinal cord injury management and provides a comprehensive, step-by-step guide to the operative decompression of complex sacral fractures.
Biomechanics and Pathoanatomy
The Denis Three-Column Theory and Sacral Zones
The modern understanding of spinal stability is heavily influenced by the Denis three-column concept, which defines instability based on the disruption of the anterior, middle, and posterior osteoligamentous complexes. When applied to the sacrum, Denis further classified fractures into three distinct anatomic zones based on their relationship to the sacral foramina:
- Zone I (Alar Region): Fractures lateral to the sacral foramina. These rarely cause neurologic deficits (less than 6% incidence) but can be associated with L5 nerve root injuries due to superior displacement of the sacral ala.
- Zone II (Foraminal Region): Fractures extending through the sacral foramina. These carry a higher risk of neurologic injury (up to 28%), typically presenting as unilateral radiculopathies (sciatica-like symptoms).
- Zone III (Central Canal Region): Fractures involving the central sacral canal. These are the most devastating, with a neurologic deficit rate exceeding 50%.
Transverse Sacral Fractures and Spinopelvic Dissociation
Transverse fractures of the sacrum typically occur in Zone III and are often the result of a high-energy axial load combined with hyperflexion (e.g., a fall from a height or a severe motor vehicle collision). When a transverse fracture connects bilateral longitudinal transforaminal fractures, it creates a "U-type" or "H-type" spinopelvic dissociation.
In these patterns, the upper sacrum and lumbar spine are mechanically disconnected from the pelvis. The upper sacral segment typically flexes forward, leading to severe kyphotic deformity and direct compression or transection of the sacral nerve roots within the canal.
Clinical Pearl: A transverse sacral fracture at or above the S3 level is highly pathognomonic for sphincter dysfunction and perineal numbness. Always perform a meticulous digital rectal examination (DRE) to assess resting tone, voluntary squeeze, and perianal sensation in any patient with a suspected pelvic ring or sacral injury.
Acute Spinal Cord Injury (SCI) Protocols
Neurologic Assessment
The initial evaluation of any patient with a suspected spinal injury must follow Advanced Trauma Life Support (ATLS) protocols, followed by a rigorous neurologic assessment using the American Spinal Injury Association (ASIA) Impairment Scale. The ASIA scale standardizes the documentation of motor and sensory function, determining whether an injury is complete (ASIA A) or incomplete (ASIA B-D).
In the context of sacral fractures, the focus is heavily on the lower sacral segments (S2-S5). The presence of "sacral sparing" (preserved perianal sensation or voluntary anal sphincter contraction) is the critical differentiator between a complete and incomplete spinal cord or cauda equina injury.
The Role of Methylprednisolone
The pharmacological management of acute SCI has been a subject of intense academic debate. Historically, the National Acute Spinal Cord Injury Studies (NASCIS II and III) popularized the use of high-dose intravenous methylprednisolone (MPSS) administered within 8 hours of injury. The protocol involved a 30 mg/kg bolus followed by a 5.4 mg/kg/hr infusion for 24 to 48 hours.
However, contemporary evidence-based analyses have critically reappraised these trials. Current guidelines from major orthopedic and neurosurgical societies consider the use of MPSS as an option rather than a standard of care. The modest, often clinically insignificant motor improvements must be weighed against the well-documented risks of severe complications, including:
* Gastrointestinal hemorrhage
* Sepsis and severe wound infections
* Avascular necrosis
* Pulmonary complications
Surgical Warning: In patients with concomitant severe polytrauma, open fractures, or traumatic brain injury, the administration of high-dose steroids is generally contraindicated due to the unacceptably high risk of systemic sepsis and immunosuppression.
Advanced Imaging Protocols
Plain radiographs of the pelvis and lumbar spine are notoriously inadequate for diagnosing complex sacral fractures. The complex osseous anatomy, overlying bowel gas, and the lordotic curve of the lumbosacral junction obscure fracture lines.
Computed Tomography (CT)
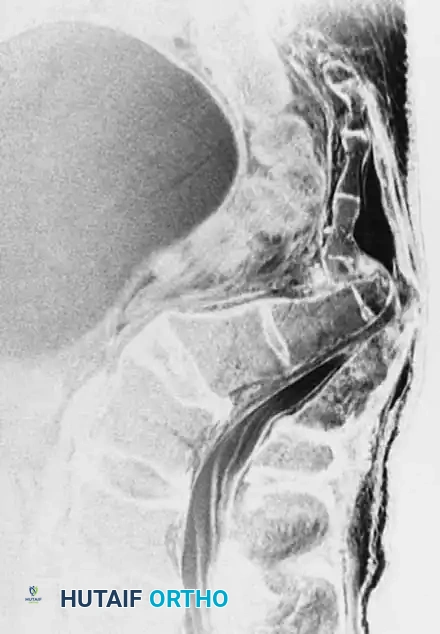
High-resolution, fine-cut CT scanning with sagittal and coronal reconstructions is the gold standard for defining the bony architecture of a sacral fracture. Sagittal reconstructions are absolutely critical for identifying transverse fracture lines, assessing the degree of kyphotic angulation, and evaluating the bony compromise of the sacral spinal canal.

Fig. 35-62 A, Sagittal CT scan of transverse fracture through S3 resulting in transection of sacral nerve roots (arrow).
Magnetic Resonance Imaging (MRI)
While CT defines the bone, MRI is indispensable for evaluating the soft tissues, ligamentous complexes, and neural elements. In the setting of a neurologic deficit, an MRI will clearly delineate hematomas, disc herniations, and the exact nature of neural compression. In severe transverse fractures, MRI can demonstrate complete canal compromise and potential transection of the cauda equina or sacral roots.

Fig. 35-62 B, MRI of sacral fracture resulting in complete canal compromise.
Surgical Indications
The decision to proceed with operative intervention in sacral fractures is based on two primary pillars: neurologic status and mechanical stability.
Indications for Urgent Decompression:
1. Progressive neurologic deficit.
2. Incomplete neurologic deficit with documented persistent neural compression (e.g., cauda equina syndrome).
3. Open sacral fractures requiring debridement.
Indications for Mechanical Stabilization:
1. Spinopelvic dissociation (U-type fractures).
2. Severe kyphotic deformity (>20 degrees) at the fracture site.
3. Intractable pain preventing mobilization in a patient with a mechanically unstable pattern.
Pitfall: Operating on a complete, long-standing neurologic injury (e.g., transected roots presenting days after injury) is unlikely to yield neurologic recovery. In such cases, surgery is indicated primarily for mechanical stabilization and deformity correction to allow for upright sitting and rehabilitation.
Operative Technique: Sacral Laminectomy and Decompression
The following details the surgical approach for a posterior sacral laminectomy to decompress the sacral nerve roots, typically performed in conjunction with lumbopelvic fixation.
1. Patient Positioning and Preparation
- The patient is intubated and carefully log-rolled onto a radiolucent Jackson spinal table.
- The abdomen must hang free to decrease intra-abdominal pressure, thereby reducing epidural venous bleeding.
- All pressure points are meticulously padded.
- Intraoperative neuromonitoring (Somatosensory Evoked Potentials [SSEPs] and Electromyography [EMG] of the lower extremities and anal sphincter) is established.
- The surgical field is prepped widely from the mid-thoracic spine down to the proximal thighs to allow for extensive instrumentation if necessary.
2. Surgical Approach
- A standard midline posterior longitudinal incision is made, centered over the lumbosacral junction and extending distally over the sacrum.
- Subcutaneous tissues are divided in line with the incision. Meticulous hemostasis is achieved using bipolar electrocautery.
- The lumbodorsal fascia is incised, and the paraspinal musculature is elevated subperiosteally off the spinous processes and lamina of the lower lumbar vertebrae and the median sacral crest.
- Dissection is carried laterally to expose the sacral foramina and the posterior superior iliac spines (PSIS) if lumbopelvic fixation is planned.
3. Sacral Laminectomy and Decompression
- The level of the transverse fracture is identified using intraoperative fluoroscopy.
- Using a high-speed burr (e.g., 3mm matchstick or diamond burr), the median sacral crest is thinned over the area of compression.
- Kerrison rongeurs (sizes 2 through 4) are utilized to carefully remove the remaining dorsal cortex of the sacrum, unroofing the sacral canal.
- Caution: The dura in the sacral region is extremely thin and often adherent to the overlying bone, especially in the setting of trauma and hematoma. Dissection must proceed with extreme care to avoid iatrogenic cerebrospinal fluid (CSF) leaks.
- The decompression must extend proximally and distally until normal, uncompressed dura and nerve roots are visualized.
- If a dural tear is encountered (common in severe transverse fractures with root transection), it must be repaired primarily with 4-0 or 5-0 non-absorbable suture (e.g., Prolene or Nurolon). If primary repair is impossible, a dural patch graft (autologous fascia or synthetic matrix) and fibrin glue should be applied.
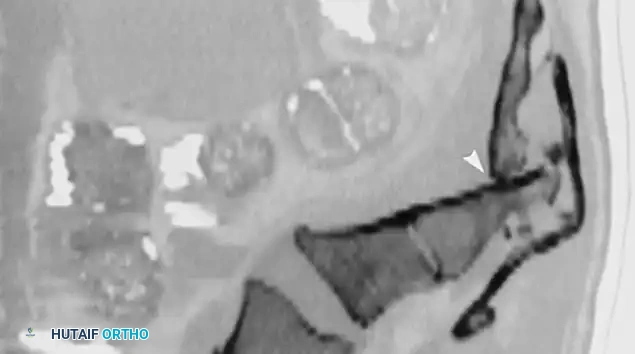
Fig. 35-62 C, Postoperative CT scan shows decompression of sacral spinal canal after laminectomy (arrows).
4. Reduction and Stabilization (Lumbopelvic Fixation)
- Decompression alone in a transverse sacral fracture will exacerbate mechanical instability. Therefore, rigid lumbopelvic fixation is mandatory.
- Pedicle screws are placed in the lower lumbar vertebrae (L4, L5).
- Iliac screws or S2-alar-iliac (S2AI) screws are placed bilaterally to anchor the construct into the pelvis.
- The fracture is reduced using distraction/compression maneuvers across the rods, correcting the sacral kyphosis.
- Posterolateral decortication is performed, and autologous bone graft (often harvested from the PSIS) or allograft is packed into the fusion bed.
5. Closure
- A subfascial drain is placed (unless a dural tear was repaired, in which case drains are generally avoided or placed strictly to gravity to prevent CSF fistula).
- The lumbodorsal fascia is closed tightly in a watertight fashion using heavy, interrupted absorbable sutures (e.g., #1 Vicryl).
- The subcutaneous layer and skin are closed meticulously. Given the poor soft tissue envelope over the sacrum, a robust closure is critical to prevent wound dehiscence.
Postoperative Protocols and Rehabilitation
Acute Postoperative Care
- Hemodynamics: Maintain mean arterial pressure (MAP) > 85 mmHg for the first 5-7 days to ensure adequate perfusion to the injured neural elements.
- Positioning: If rigid lumbopelvic fixation was achieved, patients can typically be mobilized early. If fixation is tenuous, a period of flat bed rest may be required.
- DVT Prophylaxis: Mechanical prophylaxis (SCDs) is initiated immediately. Chemical prophylaxis (e.g., Low Molecular Weight Heparin) is typically started 24-48 hours postoperatively, provided there is no evidence of expanding epidural hematoma.
Long-Term Rehabilitation
Neurologic recovery following sacral root injury is notoriously slow and unpredictable. Patients require intensive inpatient rehabilitation focusing on:
* Bowel and Bladder Management: Intermittent catheterization protocols and bowel regimens are instituted early. Urologic consultation is essential for urodynamic testing.
* Mobility: Wheelchair training, transfer techniques, and gait training (if motor function permits) using appropriate orthoses (e.g., AFOs).
* Skin Care: Education on pressure relief and frequent repositioning to prevent decubitus ulcers, which are highly prevalent in patients with sacral numbness.
Complications and Pitfalls
The surgical management of sacral fractures carries a high complication profile:
1. Wound Infection and Dehiscence: The posterior sacral approach is prone to breakdown due to thin soft tissues, pressure while supine, and proximity to the perineum. Deep infections may require serial debridements and hardware removal once fusion is achieved.
2. Hardware Failure: The spinopelvic junction experiences massive biomechanical shear forces. Pseudarthrosis, rod breakage, or iliac screw loosening can occur, particularly in non-compliant patients or those with severe osteoporosis.
3. Neurologic Deterioration: Iatrogenic injury during decompression or over-distraction during reduction can worsen neurologic deficits.
4. CSF Fistula: Unrecognized or inadequately repaired dural tears can lead to persistent CSF leaks, pseudomeningoceles, and meningitis.
Conclusion
Transverse sacral fractures with neurologic compromise represent one of the most formidable challenges in orthopedic trauma. Successful management hinges on rapid clinical assessment, advanced cross-sectional imaging, and a deep understanding of spinopelvic biomechanics. When indicated, meticulous surgical decompression via sacral laminectomy, coupled with rigid lumbopelvic stabilization, offers the best opportunity for neurologic recovery and restoration of mechanical integrity. Adherence to strict postoperative protocols and multidisciplinary rehabilitation is paramount to optimizing long-term patient outcomes.
📚 Medical References
- Sacral fractures and injuries to the cauda equina, J Bone Joint Surg 27:113, 1945.
- Bordurant FJ, Cotler HB, Kulkarni MV, et al: Acute spinal cord injury: a study using physical examination and magnetic resonance imaging, Spine 15:161, 1990.
- Borrelli J, Koval KJ, Helfet DL: The crescent fracture: a posterior fracture dislocation of the sacroiliac joint, J Orthop Trauma 10:165, 1996.
- Bostman OM, Myllynen PJ, Riska EB: Unstable fracture of the thoracic and lumbar spine: the audit of an 8-year series with early reduction using Harrington instrumentation, Injury 18:190, 1987.
- Bracken MB, Shepard MJ, Collins WF, et al: A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal cord injury, N Engl J Med 322:1405, 1990.
- Bradford DS, Akbarnia BA, Winter RD, et al: Surgical stabilization of fractures and fracture-dislocations of the thoracic spine, Spine 2:185, 1977.
- Bradford DS, McBride GG: Surgical management of thoracolumbar spine fractures with incomplete neurologic defi cits, Clin Orthop Relat Res 218:201, 1987.
- Broom MJ, Jacobs RR: Update 1988: current status of internal fi xation of thoracolumbar fractures, J Orthop Trauma 3:148, 1989.
- Bryant CE, Sullivan JA: Management of thoracic and lumbar spine fractures with Harrington distraction rods supplemented with segmental wiring, Spine 8:532, 1983.
- Calenoff L, Chessare JW, Rogers LF, et al: Multiple level spinal injuries: importance of early recognition, AJR Am J Roentgenol 130:665, 1978.
- Campbell SE, Phillips CD, Dubovsky E, et al: The value of CT in determining potential instability of simple wedgecompression fractures of the lumbar spine, AJNR Am J Neuroradiol 16:1385, 1995.
- Capen DA: Classifi cation of thoracolumbar fractures and posterior instrumentation for treatment of thoracolumbar fractures, Instr Course Lect 48:437, 1999.
- [Carl A: Sacral spine fractures. In Errico TJ, Bauer RD, Waugh T, eds: Spinal trauma, Philadelphia, 1990, Lippincott.
Carl AL, Tranmer BI, Sachs BL: Anterolateral dynamized instrumentation and fusion for unstable thoracolumbar and lumbar burst fractures, Spine 22:686, 1997.](https://pubmed.ncbi.nlm.nih.gov/?term=Carl%20A%3A%20Sacral%20spine%20fractures.%20In%20Errico%20TJ%2C%20Bauer%20RD%2C%20Waugh%20T%2C%20eds%3A%20Spinal%20trauma%2C%20Philadelphia%2C%201990%2C%20Lippincott.%0A%0ACarl%20AL%2C%20Tranmer%20BI%2C%20Sachs%20BL%3A%20Anterolateral%20dynamized%20instrumentation%20and%20fusion%20for%20unstable%20thoracolumbar%20and%20lumbar%20burst%20fractures%2C%20Spine%2022%3A686%2C%201997.)
- Carl AL, Tromanhauser SG, Roger DJ: Pedicle screw instrumentation for thoracolumbar burst fractures and fracturedislocations, Spine 17:S317, 1992.
- Carlson GD, Warden KE, Barbeau JM, et al: Viscoelastic relaxation and regional blood fl ow response to spinal cord compression and decompression, Spine 22:1285, 1997.
- Chan DPK, Seng NK, Kaan KT: Nonoperative treatment in burst fractures of the lumbar spine (L2-L5) without neurologic defi cits, Spine 18:320, 1993.
- Chang KW: A reduction-fi xation system for unstable thoracolumbar burst fractures, Spine 17:879, 1992.
- Chen WJ, Niu CC, Chen LH, et al: Back pain after thoracolumbar fracture treated with long instrumentation and short fusion, J Spinal Disord 8:474, 1995.
- Chipman JG, Deuser WE, Beilman GJ: Early surgery for thoracolumbar spine injuries decreases complications, J Trauma 56:52, 2004.
- Chiras J, Depriester C, Weill A, et al: Percutaneous vertebral surgery: technics and indications, J Neuroradiol 24:45, 1997.
- Choe DH, Marom EM, Ahrar K, et al: Pulmonary embolism of polymethyl methacrylate during percutaneous vertebroplasty and kyphoplasty, AJR Am J Roentgenol 183:1097, 2004.
- Chow GH, Nelson BJ, Gebhard JS, et al: Functional outcome of thoracolumbar burst fractures managed with hyperextension casting or bracing and early mobilization, Spine 21:2170, 1996.
- Clark JE: Apophyseal fracture of the lumbar spine in adolescence, Orthop Rev 20:512, 1991.
- Clohisy JC, Akbarnia BA, Bucholz RD, et al: Neurologic recovery associated with anterior decompression of spine fractures at the thoracolumbar junction (T12-L1), Spine 17(suppl):325, 1992.
- Cotrel Y, Dubousset J: Universal instrumentation (CD) for spinal surgery (technique manual), Greensburg, Penn, 1985, Stuart. Cotrel Y, Dubousset J, Guillaumat M: New universal instrumentation for spinal surgery, Clin Orthop Relat Res 227:10, 1988.
- Court-Brown CM, Gertzbein SD: The management of burst fractures of the fi fth lumbar vertebra, Spine 12:308, 1987.
- Cresswell TR, Marshall PD, Smith RB: Mechanical stability of the AO internal spinal fi xation system compared with that of the Hartshill rectangle and sublaminar wiring in the management of unstable burst fractures of the thoracic and lumbar spine, Spine 23:111, 1998.
- Daniaux H, Seykora P, Genelin A, et al: Application of posterior plating and modifi cations in thoracolumbar spine injuries, Spine 16(suppl):126, 1991.
- Danisa OA, Shaffrey CI, Jane JA, et al: Surgical approaches for the correction of unstable thoracolumbar burst fractures: a retrospective analysis of treatment outcomes, J Neurosurg 83:977, 1995.
- Davies WE, Morris JH, Hill V: An analysis of conservative (nonsurgical) management of thoracolumbar fractures and fracture dislocations with neural damage, J Bone Joint Surg 62A:1324, 1980.
- Davis LA, Warren SA, Reid DC, et al: Incomplete neural defi cits in thoracolumbar and lumbar spine fractures: reliability of Frankel and Sunnybrook scales, Spine 18:257, 1993.
- De Klerk LW, Fontijne WP, Stijnen T, et al: Spontaneous remodeling of the spinal canal after conservative management of the thoracolumbar burst fractures, Spine 23:1057, 1998.
- Delamarter RB, Bohlman HH, Dodge LD, et al: Experimental
You Might Also Like