Operative Management and Evaluation of Soft-Tissue Tumors
Key Takeaway
Soft-tissue tumors present a unique diagnostic and surgical challenge in orthopedic oncology. Unlike bone tumors, the absence of pain does not reliably differentiate benign from malignant lesions. This comprehensive guide details the evidence-based evaluation, advanced imaging protocols, strict biopsy principles, and surgical management strategies for soft-tissue neoplasms. Emphasizing multidisciplinary care, it covers staging, wide resection techniques, and adjuvant therapies essential for optimizing local control and patient survival.
Introduction to Soft-Tissue Tumors
The management of soft-tissue tumors represents one of the most complex and high-stakes domains within orthopedic oncology. Ranging from ubiquitous benign lipomas to highly aggressive, life-threatening soft-tissue sarcomas (STS), these lesions demand a rigorous, protocol-driven approach. The evaluation of patients with musculoskeletal neoplasms requires meticulous attention to detail, as the initial diagnostic and surgical steps dictate the ultimate oncologic outcome.
Unlike primary bone tumors, where pain is a hallmark symptom of cortical destruction or periosteal reaction, soft-tissue tumors are notoriously insidious. Most patients with soft-tissue malignancies present with a painless, progressively enlarging mass. Consequently, any deep-seated mass, or any superficial mass larger than 5 cm, must be treated with a high index of suspicion and evaluated comprehensively before any surgical intervention is considered.
CLINICAL PEARL: The presence or absence of pain is a notoriously unreliable indicator of malignancy in soft-tissue tumors. A painless, enlarging mass deep to the fascia must be considered a sarcoma until proven otherwise.
Clinical Evaluation
The clinical evaluation of a suspected soft-tissue tumor begins with a thorough history and physical examination, focusing on the biological behavior of the mass.
History and Physical Examination
- Duration and Growth Rate: Rapid growth over weeks to months is highly suspicious for high-grade sarcomas, whereas slow growth over years suggests a benign etiology (e.g., lipoma, hemangioma).
- Physical Characteristics: Palpation should assess the size, depth (superficial vs. deep to the investing fascia), consistency (firm, fluctuant, rubbery), and mobility. Fixation to underlying bone or overlying skin implies aggressive local invasion.
- Regional Lymph Nodes: While most soft-tissue sarcomas metastasize hematogenously (primarily to the lungs), certain subtypes exhibit a higher propensity for lymphatic spread. Careful palpation of regional lymph node basins (inguinal, axillary, cervical) is mandatory.
ONCOLOGIC APHORISM: The sarcomas most likely to metastasize to lymph nodes can be remembered by the mnemonic SCARE: Synovial sarcoma, Clear cell sarcoma, Angiosarcoma, Rhabdomyosarcoma, and Epithelioid sarcoma.
Advanced Imaging Protocols
Imaging must always precede biopsy. Premature biopsy alters the local tissue architecture, introduces hemorrhage and edema, and severely compromises the diagnostic accuracy of subsequent imaging studies.
Radiography
Standard orthogonal radiographs of the involved extremity are the mandatory first step. While soft-tissue tumors are radiolucent, plain films provide critical diagnostic clues:
* Phleboliths: Pathognomonic for hemangiomas or vascular malformations.
* Calcifications: Stippled or amorphous calcifications may indicate synovial sarcoma or extraskeletal osteosarcoma/chondrosarcoma.
* Fat-Density Radiolucencies: Suggestive of lipomas or well-differentiated liposarcomas.
* Bone Involvement: Extrinsic cortical erosion (saucerization) or frank periosteal reaction indicates secondary bone involvement, altering the surgical plan.
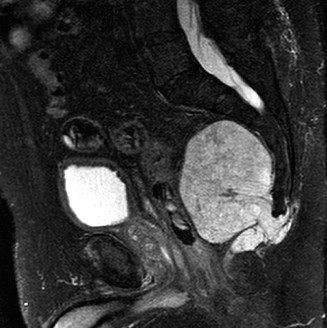
Magnetic Resonance Imaging (MRI)
MRI is the gold standard for evaluating soft-tissue tumors. It provides unparalleled contrast resolution, delineating the tumor's size, compartmental boundaries, and relationship to critical neurovascular structures.
* T1-Weighted Images: Excellent for defining anatomic boundaries and identifying fat (hyperintense) or hemorrhage.
* T2-Weighted Images: Highly sensitive for detecting edema in the reactive zone surrounding the tumor. Most sarcomas are hyperintense on T2.
* Gadolinium Enhancement: Differentiates cystic/necrotic areas from solid, vascularized tumor tissue, guiding the optimal biopsy trajectory.
* Specific Diagnoses: MRI can be virtually diagnostic for certain benign lesions, such as lipomas (isointense to subcutaneous fat on all sequences), hemangiomas ("bag of worms" appearance), and pigmented villonodular synovitis (PVNS) (hemosiderin blooming artifact on gradient-echo sequences).
Computed Tomography (CT) and Systemic Staging
- Local CT: Utilized when MRI is contraindicated (e.g., pacemakers) or to better define subtle osseous involvement.
- Staging CT: A non-contrast CT of the chest is mandatory for all patients with malignant soft-tissue tumors to rule out pulmonary metastases.
- Abdominal/Pelvic CT: Indicated for myxoid liposarcomas (which have a unique propensity for retroperitoneal and extrapulmonary metastases) and for tumors prone to lymphatic spread.
- Technetium-99m Bone Scans / PET-CT: Useful in selected cases to evaluate for distant osseous metastases or to assess the metabolic activity of the primary lesion.
Principles of Biopsy
The biopsy is the most critical step in the initial management of a soft-tissue tumor. A poorly planned biopsy can preclude limb-salvage surgery, necessitating amputation.
SURGICAL WARNING: Never perform an unplanned excision ("whoops" surgery) of a deep-seated soft-tissue mass. Inadvertent violation of a sarcoma pseudocapsule contaminates the entire surgical bed, drastically increasing the risk of local recurrence.
Core Needle Biopsy vs. Open Incisional Biopsy
- Core Needle Biopsy (CNB): The preferred initial modality. It is minimally invasive, carries a lower risk of tumor seeding, and provides sufficient tissue for histologic, immunohistochemical, and molecular analysis in over 90% of cases. It should be performed under ultrasound or CT guidance to target the most viable (enhancing) portion of the tumor, avoiding necrotic centers.
- Open Incisional Biopsy: Reserved for cases where CNB is non-diagnostic.
Strict Biopsy Rules
- Longitudinal Incisions Only: Transverse incisions contaminate multiple anatomic compartments and neurovascular planes, making subsequent wide resection impossible.
- Direct Approach: The biopsy tract must pass through the involved muscle belly, not through intermuscular planes. Contaminating an intermuscular plane requires resection of both adjacent muscles during definitive surgery.
- Meticulous Hemostasis: Post-biopsy hematoma spreads tumor cells far beyond the primary site. A tourniquet may be used but must be deflated prior to closure to ensure absolute hemostasis.
- Tract Excision: The entire biopsy tract (skin, subcutaneous tissue, and muscle) must be excised en bloc with the tumor during definitive resection.
Pathologic Classification and Staging
Staging dictates prognosis and treatment. The two primary systems are the Enneking System (Musculoskeletal Tumor Society - MSTS), which relies on histologic grade (G), anatomic setting (T - intracompartmental vs. extracompartmental), and metastasis (M); and the American Joint Committee on Cancer (AJCC) System, which incorporates tumor size, depth, grade, and nodal/distant metastasis.
Common Benign Soft-Tissue Tumors
- Fatty Tumors (Lipoma): The most common soft-tissue tumor. Treated with marginal excision if symptomatic.
- Nerve Sheath Tumors (Schwannoma, Neurofibroma): Schwannomas are eccentric to the nerve and can often be enucleated while preserving nerve function. Neurofibromas are central and intimately involved with nerve fascicles, making resection without neurologic deficit difficult.
- Vascular Lesions (Hemangioma): Often present with pain exacerbated by exercise or dependency. Treated conservatively; surgery is reserved for refractory cases due to high recurrence rates.
- Fibrous Lesions (Desmoid Tumors / Aggressive Fibromatosis): Benign but locally highly aggressive. They lack metastatic potential but infiltrate surrounding tissues. Management is shifting toward observation or medical therapy (NSAIDs, anti-estrogens, tyrosine kinase inhibitors), reserving wide resection for progressive, symptomatic lesions.
Common Malignant Soft-Tissue Tumors (Sarcomas)
- Undifferentiated Pleomorphic Sarcoma (UPS): Formerly known as Malignant Fibrous Histiocytoma (MFH). It is the most common soft-tissue sarcoma in older adults, typically presenting in the proximal extremities (thigh).
- Liposarcoma: Ranges from atypical lipomatous tumors (low-grade, locally aggressive) to dedifferentiated and pleomorphic variants (high-grade, high metastatic potential). Myxoid liposarcomas uniquely metastasize to the spine and retroperitoneum.
- Synovial Sarcoma: Misnamed, as it rarely arises from actual synovium. It typically occurs near joints in young adults (15-40 years) and is characterized by the t(X;18) chromosomal translocation. It has a high propensity for lymph node metastasis.
- Rhabdomyosarcoma: The most common soft-tissue sarcoma in children. Treated with a multimodal approach heavily reliant on systemic chemotherapy.
- Malignant Peripheral Nerve Sheath Tumor (MPNST): Highly aggressive, often arising in the setting of Neurofibromatosis Type 1 (NF1). Prognosis is generally poor.
Surgical Management: Operative Techniques
The primary goal of sarcoma surgery is complete eradication of the tumor with negative microscopic margins, while maximizing limb function.
Surgical Margins Defined
- Intralesional: Macroscopic tumor left behind (unacceptable for sarcomas).
- Marginal: Resection through the reactive zone/pseudocapsule. Microscopic disease remains. Acceptable only for benign lesions.
- Wide: Resection of the tumor with a continuous cuff of normal, healthy tissue in all directions. This is the standard of care for soft-tissue sarcomas.
- Radical: Removal of the entire anatomic compartment containing the tumor. Rarely performed today due to the efficacy of wide resection combined with radiation.
Preoperative Preparation and Positioning
- Positioning: The patient must be positioned to allow access to the entire compartment and potential flap donor sites. A sterile tourniquet is applied but generally not inflated unless catastrophic hemorrhage occurs, as ischemia can obscure the distinction between normal and reactive tissues.
- Incision Planning: The incision must incorporate the previous biopsy tract as an ellipse. It should be longitudinal and extensile.
Step-by-Step Surgical Approach (Wide Resection)
- Skin and Flap Elevation: Incise the skin, incorporating the biopsy tract. Raise full-thickness fasciocutaneous flaps away from the tumor. Do not dissect down to the tumor pseudocapsule.
- Identifying the Barrier: The concept of anatomic barriers is critical. Fascia, periosteum, and epineurium are strong barriers to tumor spread. Fat and muscle are poor barriers. The resection plane must be established in normal tissue, at least 1-2 cm away from the tumor in muscle/fat, or separated by a pristine fascial layer.
- Vascular Control: Identify and isolate the major neurovascular bundles proximal and distal to the tumor mass. If the tumor is intimately adherent to the adventitia of major vessels, vascular resection and bypass grafting (using reversed saphenous vein or synthetic grafts) may be required to achieve negative margins.
- En Bloc Resection: Dissect circumferentially around the tumor, maintaining the cuff of normal tissue. The tumor must not be visualized during the dissection. If the tumor is seen, the margin has been breached.
- Specimen Orientation: Once excised, the specimen must be oriented for the pathologist using sutures or surgical clips (e.g., short stitch superior, long stitch lateral) to allow precise mapping of any close or positive margins.
- Reconstruction and Closure: Meticulous hemostasis is achieved. Dead space must be obliterated using muscle flaps (e.g., gastrocnemius or latissimus dorsi flaps) to prevent seroma formation and provide healthy, vascularized tissue over exposed neurovascular structures or bone. Closed suction drains are placed in-line with the incision.
Adjuvant Therapies and Controversies
The management of high-grade soft-tissue sarcomas is inherently multidisciplinary. While surgery is the cornerstone, adjuvant therapies are critical for local control and systemic disease management.
Radiation Therapy
Radiation is indicated for high-grade sarcomas, large tumors (>5 cm), and deep lesions to improve local control rates, which approach 90% when combined with wide resection.
* Preoperative (Neoadjuvant) Radiation: Typically 50 Gy delivered over 5 weeks.
* Advantages: Lower total dose, smaller radiation field, sterilizes the reactive zone (making the pseudocapsule thicker and easier to resect), and the tumor is well-oxygenated (increasing radiosensitivity).
* Disadvantages: Significantly higher rate of major postoperative wound complications (up to 35%).
* Postoperative Radiation: Typically 60-66 Gy.
* Advantages: Lower risk of acute wound healing complications.
* Disadvantages: Higher total dose, larger radiation field (must include the entire surgical bed and drain tracts), leading to higher rates of long-term morbidities such as joint contracture, lymphedema, and radiation-induced fractures.
* Brachytherapy: The placement of radioactive seeds (e.g., Iridium-192) directly into the tumor bed via catheters placed at the time of surgery. It delivers a highly localized dose while sparing surrounding healthy tissue. Its use is highly institution-dependent.
Chemotherapy
The role of systemic chemotherapy in adult soft-tissue sarcomas remains one of the most debated topics in orthopedic oncology.
* Unlike pediatric sarcomas (Ewing, Osteosarcoma, Rhabdomyosarcoma) where chemotherapy dramatically improves survival, adult STS response rates are variable.
* Adjuvant chemotherapy (typically Doxorubicin and Ifosfamide-based regimens) is generally considered for young, healthy patients with large (>5 cm), deep, high-grade sarcomas at high risk for systemic metastasis. However, definitive proof of an overall survival benefit remains elusive in large randomized controlled trials, and the decision is made on a case-by-case basis at specialized tumor boards.
Postoperative Protocols and Surveillance
Immediate Postoperative Care
- Immobilization of the extremity may be required if major muscle groups were resected or if vascular/nerve repairs were performed.
- Drains are maintained until output is minimal (<30 cc/day) to prevent hematoma/seroma, which can act as a nidus for infection or delay adjuvant therapies.
- Aggressive deep vein thrombosis (DVT) prophylaxis is instituted, as cancer patients are in a hypercoagulable state.
Long-Term Surveillance
Patients with soft-tissue sarcomas require rigorous, long-term follow-up to detect local recurrences and pulmonary metastases early.
* Years 1-2: Physical examination, local MRI of the surgical bed, and non-contrast chest CT every 3 to 4 months.
* Years 3-5: Clinical exam, local MRI, and chest CT every 6 months.
* Years 5-10: Annual clinical exam and chest radiography/CT.
> CLINICAL PEARL: The majority of local recurrences and pulmonary metastases occur within the first two years following definitive resection. However, late recurrences (beyond 5 years) are well-documented, particularly in low-grade sarcomas and certain subtypes like myxoid liposarcoma, mandating a minimum of 10 years of oncologic surveillance.
📚 Medical References
- soft-tissue tumors, Philadelphia, 1998, Lippincott-Raven. Skolnick AA: Early data suggest clot-dissolving drug may help save frostbitten limbs from amputation, JAMA 267:2008, 1992.
- Smith D, Assal M, Reiber G, et al: Minor environmental trauma and lower extremity amputation in high-risk patients with diabetes: incidence, pivotal events, etiology, and amputation level in a prospectively followed cohort, Foot Ankle Int 24:690, 2003.
- Smith DG: Principles of partial
You Might Also Like