Subacute Hematogenous Osteomyelitis: Comprehensive Surgical Guide
Key Takeaway
Subacute hematogenous osteomyelitis presents an insidious clinical challenge, often mimicking primary bone neoplasms. Characterized by an indolent course and minimal systemic symptoms, diagnosis relies heavily on advanced imaging and high clinical suspicion. This guide details the pathophysiology, the Roberts classification system, and evidence-based surgical interventions—including cortical windowing and intramedullary debridement—essential for eradicating persistent localized bone infections like the Brodie abscess.
Introduction to Subacute Hematogenous Osteomyelitis
Subacute hematogenous osteomyelitis represents a distinct clinical entity within the spectrum of bone infections. Compared with acute osteomyelitis, the subacute variant is characterized by a markedly more insidious onset and a conspicuous absence of severe systemic symptoms. This indolent presentation frequently confounds initial clinical evaluations, making the diagnosis of this disorder notoriously difficult and often delayed.
Despite its subtle presentation, subacute osteomyelitis is relatively common in orthopedic practice. Epidemiological studies, such as those by Jones et al., have reported that up to 35% of patients presenting with primary bone infections are ultimately diagnosed with subacute osteomyelitis. Because of its protracted and indolent course, the diagnosis is typically delayed for more than two weeks from the onset of initial, often vague, symptoms.
Clinical Pearl: The hallmark of subacute hematogenous osteomyelitis is the dissociation between the patient's mild clinical symptoms and the potentially aggressive appearance of the lesion on radiographic imaging. Always maintain a high index of suspicion when evaluating a patient with persistent, localized bone pain but normal systemic inflammatory markers.
Pathophysiology and Microbiology
The indolent course of subacute osteomyelitis is generally understood to be the result of a delicate equilibrium between the host's immune system and the invading pathogen. This balance is typically achieved through one of three primary mechanisms:
1. Increased Host Resistance: A robust and highly competent immune response that successfully walls off the infection, preventing systemic dissemination.
2. Decreased Bacterial Virulence: Infection by organisms that inherently possess lower virulence factors, leading to a localized, low-grade inflammatory response.
3. Prior Antibiotic Administration: The empirical administration of antibiotics for unrelated or misdiagnosed conditions prior to the onset of definitive orthopedic symptoms, which partially suppresses the infection without fully eradicating it.
It is widely speculated that the combination of an organism of low virulence with a strong host response allows the inflammation to persist within the rigid confines of the bone without producing significant systemic signs or symptoms.
Microbiologically, Staphylococcus aureus and Staphylococcus epidermidis are the predominant organisms identified in subacute osteomyelitis. However, even with an adequate bone aspirate or a meticulously obtained open biopsy specimen, a causative pathogen is successfully identified only 60% of the time. This high rate of culture-negative results underscores the necessity of obtaining multiple tissue samples for aerobic, anaerobic, fungal, and acid-fast bacilli (AFB) cultures, alongside definitive histological examination.
Clinical Presentation and Diagnostic Evaluation
Systemic signs and symptoms in subacute hematogenous osteomyelitis are characteristically minimal. The patient's core body temperature is usually normal or only mildly elevated. Mild-to-moderate, deep, aching pain is often the only consistent sign suggesting the diagnosis.
Laboratory Findings
Laboratory investigations are frequently unhelpful in establishing a definitive diagnosis:
* White Blood Cell (WBC) Count: Generally within normal limits.
* Erythrocyte Sedimentation Rate (ESR): Elevated in only approximately 50% of patients.
* C-Reactive Protein (CRP): May be mildly elevated but is often normal.
* Blood Cultures: Almost universally negative due to the localized nature of the infection.
Radiographic Imaging
Because laboratory markers are unreliable, correct diagnosis largely depends on clinical suspicion and radiographic findings. Plain radiographs and technetium-99m bone scans are generally positive and serve as the first-line imaging modalities.

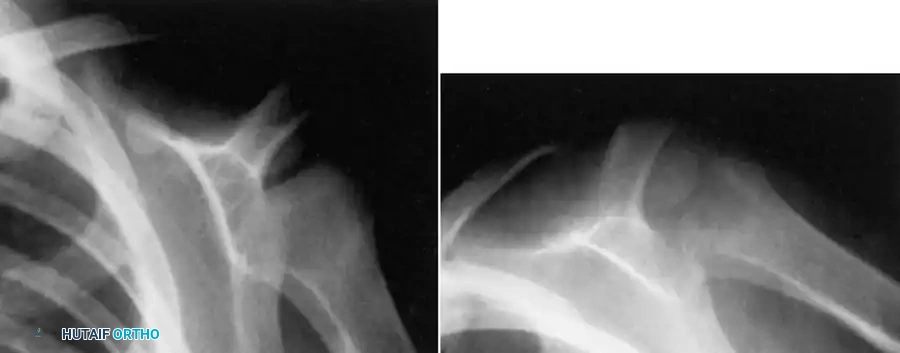
Fig. 16-7 Subacute osteomyelitis of proximal humerus. Note the localized radiolucency and surrounding sclerosis, which can easily be mistaken for a benign bone cyst or a primary bone neoplasm.
Differentiating these infectious lesions from primary bone tumors can be exceedingly difficult based on plain radiographs alone. While there are no massive series reporting Magnetic Resonance Imaging (MRI) findings exclusively in subacute osteomyelitis, MRI is invaluable. Hempfing et al. demonstrated that MRI is critical not only for diagnosing the condition by identifying bone marrow edema, soft tissue extension, and the "penumbra sign" (a rim of vascularized granulation tissue lining the abscess cavity), but also for precise preoperative planning of the surgical approach.
Radiographic Classification: The Roberts Modification
A comprehensive radiographic classification of subacute hematogenous osteomyelitis was originally described by Gledhill and subsequently modified by Roberts et al. This classification system is vital for categorizing the lesion, guiding the differential diagnosis, and determining the appropriate treatment algorithm.
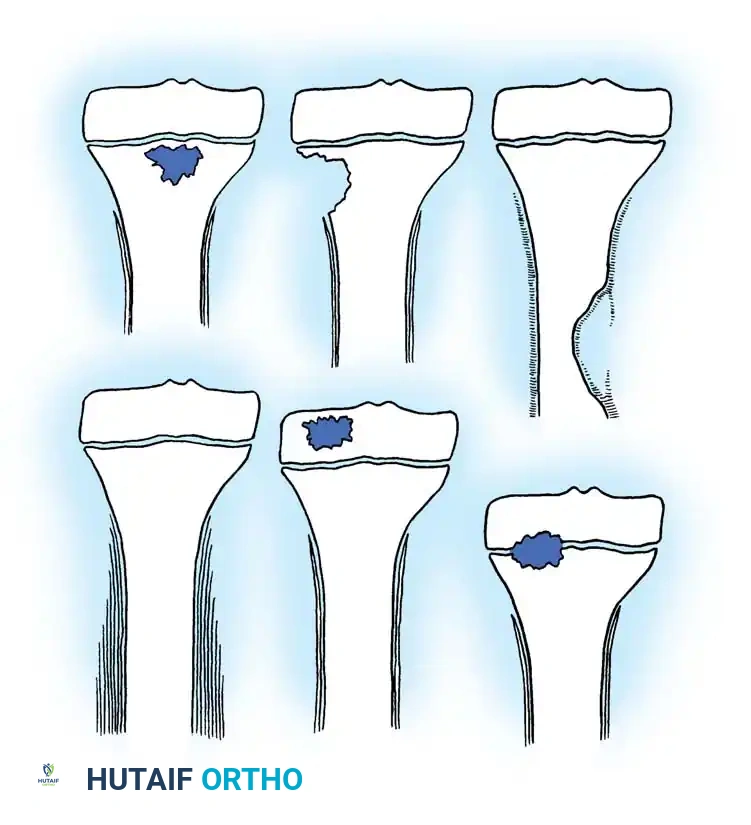
Fig. 16-8 Classification of subacute osteomyelitis: type 1, central metaphyseal lesion; type 2, eccentric metaphyseal lesion with cortical erosion; type 3, diaphyseal cortical lesion; type 4, diaphyseal lesion with periosteal new bone formation, but without definite bony lesion; type 5, primary subacute epiphyseal osteomyelitis; and type 6, subacute osteomyelitis crossing physis to involve metaphysis and epiphysis. (Modified from Roberts JM, Drummond DS, Breed AL, et al: Subacute hematogenous osteomyelitis in children: a retrospective study, J Pediatr Orthop 2:249, 1982.)
Table 16-1: Subacute Osteomyelitis Classification and Differential Diagnosis
- Type I: Solitary localized zone of radiolucency.
- Type Ia: Punched-out radiolucency surrounded by reactive new bone formation. (Differential: Langerhans’ cell histiocytosis, Eosinophilic granuloma).
- Type Ib: Punched-out radiolucent lesion with a dense sclerotic margin. (Differential: Brodie abscess).
- Type II: Metaphyseal radiolucencies with cortical erosion. (Differential: Osteogenic sarcoma).
- Type III: Cortical hyperostosis in the diaphysis; no onion skinning. (Differential: Osteoid osteoma).
- Type IV: Subperiosteal new bone and onion skin layering. (Differential: Ewing sarcoma).
- Type V: Central radiolucency in the epiphysis. (Differential: Chondroblastoma).
- Type VI: Destructive process involving the vertebral body. (Differential: Tuberculosis, Osteogenic sarcoma).
Surgical Warning: Type II and Type IV lesions mimic highly aggressive, malignant primary bone tumors (Osteosarcoma and Ewing Sarcoma, respectively). In these cases, an open biopsy is absolutely mandatory to rule out malignancy before initiating any definitive treatment.
The Brodie Abscess
A Brodie abscess is a specific, highly localized form of subacute osteomyelitis. It occurs most frequently in the long bones of the lower extremities (particularly the distal tibia and proximal femur) of young adults and children.
Anatomical Distribution
- Before Physeal Closure: The metaphysis is the most frequently affected anatomical region.
- After Physeal Closure (Adults): The lesion typically involves the metaphyseal-epiphyseal junction.
Clinical and Radiographic Profile
The presenting complaint is almost universally intermittent pain of long duration, accompanied by localized tenderness over the affected bone. On plain radiographs, a Brodie abscess generally appears as a well-circumscribed lytic lesion surrounded by a distinct, thick rim of sclerotic reactive bone. However, its appearance can be markedly varied. Careful evaluation of plain films is mandatory because a Brodie abscess can easily be mistaken for a variety of benign or malignant neoplasms.
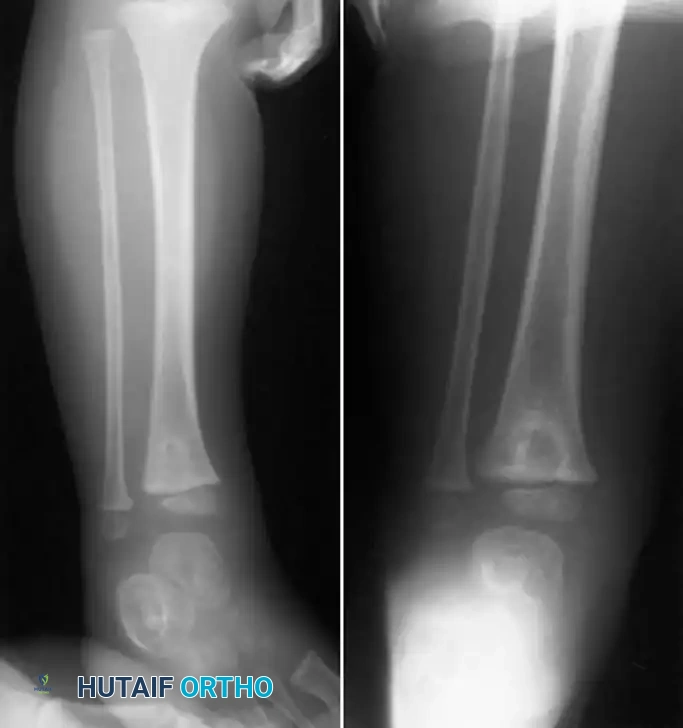
Fig. 16-9 Brodie abscess in right distal tibial epiphysis of 3-year-old child. The dense sclerotic rim is a classic hallmark of the host's attempt to wall off the low-virulence infection.
Organisms of low virulence are believed to cause the lesion. S. aureus is cultured in approximately 50% of patients, while in 20% of cases, the cultures remain completely sterile despite meticulous sampling. Because of the diagnostic uncertainty and the dense sclerotic bone that prevents antibiotic penetration, this condition frequently requires an open biopsy combined with thorough surgical curettage to establish the diagnosis and eradicate the infection.
Treatment Algorithms: Conservative vs. Surgical Management
The management of subacute hematogenous osteomyelitis requires a nuanced approach, balancing the efficacy of modern antimicrobial therapy against the necessity of surgical debridement.
Ross and Cole established a widely accepted treatment algorithm. They recommended open biopsy and curettage, followed by treatment with appropriate culture-directed antibiotics, for all lesions that appear radiographically aggressive (e.g., Types II, IV, and VI).
Conversely, for lesions that appear to be a simple, well-contained abscess in the epiphysis or metaphysis (e.g., Type I lesions without aggressive periosteal reaction), immediate biopsy is not strictly recommended. These lesions, which are highly characteristic of subacute hematogenous osteomyelitis, can be treated empirically. The recommended regimen is intravenous antibiotics for 48 hours, followed by a comprehensive 6-week course of oral antibiotics. Ross and Cole reported an impressive 87% success rate with this conservative treatment regimen.
Further supporting this, Hamdy et al. reviewed 44 patients with subacute osteomyelitis and found no statistically significant difference in clinical outcomes between conservative and surgical treatment for benign-appearing lesions. They concluded that open biopsy and curettage should be reserved strictly for aggressive-appearing lesions, or for lesions that fail to respond to an initial trial of appropriate antibiotic treatment.
Surgical Technique 16-1: Open Biopsy and Cortical Windowing
When surgical intervention is indicated—either to rule out malignancy in an aggressive-appearing lesion or to eradicate a recalcitrant Brodie abscess—meticulous surgical technique is paramount. The goal is to obtain diagnostic tissue, evacuate purulence, and debride necrotic bone while minimizing iatrogenic damage to the remaining osseous structures.
Preoperative Preparation and Positioning
- Imaging Review: Thoroughly review orthogonal plain radiographs and MRI to localize the lesion precisely. Measure the distance from the joint line or physis to avoid iatrogenic physeal arrest in pediatric patients.
- Positioning: Position the patient on a radiolucent operating table. Ensure that a C-arm fluoroscope can easily access the operative field in both anteroposterior (AP) and lateral planes.
- Tourniquet: Apply a pneumatic tourniquet to provide a bloodless field, which is critical for identifying the subtle transition between necrotic tissue and healthy, bleeding bone.
Step-by-Step Surgical Approach
-
Surgical Approach and Periosteal Incision:
- Utilize an internervous, intermuscular approach appropriate for the specific anatomical location.
- Once the bone is reached, incise the periosteum longitudinally over the center of the lesion.
- Note: The periosteum may already be elevated from the underlying cortex by a subperiosteal abscess. If so, compressed pus will escape immediately upon incision. Obtain aerobic, anaerobic, and AFB cultures immediately.
-
Periosteal Elevation (Minimizing Vascular Damage):
- If no frank abscess is found upon incision, gently elevate the periosteum approximately 1.5 cm on each side of the longitudinal incision using a Cobb elevator.
- CRITICAL STEP: Try to strip as little periosteum as possible. The cortical bone's blood supply is already severely compromised by the underlying endosteal infection and increased intramedullary pressure. The more periosteum that is stripped, the more this fragile blood supply is damaged, increasing the risk of postoperative bone necrosis and chronic osteomyelitis.
-
Creating the Cortical Window:
- Under fluoroscopic guidance, localize the center of the lesion.
- Use a 4-mm drill bit to create several holes through the near cortex and into the medullary canal. Do this regardless of whether a subperiosteal abscess was present.
- If pus escapes through these pilot holes, use the drill to outline a cortical window measuring approximately 1.3 × 2.5 cm.
- Connect the drill holes using a sharp osteotome or a high-speed burr to remove the cortical window.
Biomechanical Pearl: When creating a cortical window, never leave sharp, 90-degree corners. Sharp corners act as severe stress risers, significantly increasing the risk of a postoperative torsional pathologic fracture. Always use a burr to round the corners of the cortical defect, creating an oval or elliptical window.
-
Intramedullary Debridement and Curettage:
- Evacuate all intramedullary pus.
- Use sharp curettes to gently but thoroughly remove all necrotic granulation tissue, the sclerotic lining of the abscess cavity (the penumbra), and any sequestra.
- Send multiple distinct samples for microbiology (cultures) and surgical pathology (to definitively rule out malignancy). Purulent material is not always obtained, but thick, inflammatory granulation tissue is a common finding.
-
Irrigation and Lavage:
- Once healthy, punctate bleeding bone is reached (the "paprika sign"), irrigate the cavity copiously.
- Use at least 3 Liters of sterile normal saline delivered via a pulsatile lavage system to mechanically wash out residual planktonic bacteria and debris.
- Depending on institutional protocols and the surgeon's preference, local antibiotics (e.g., vancomycin or tobramycin powder) may be placed in the irrigation solution or packed into the defect using antibiotic-impregnated calcium sulfate beads.
-
Closure:
- Place a closed-suction drain deep into the wound bed.
- Close the deep fascial layers and the skin loosely over the drains.
- Do not close the wound tightly if doing so produces excessive tension on the skin, as postoperative edema can lead to wound necrosis and secondary infection. If necessary, leave the wound open for delayed primary closure or negative pressure wound therapy (NPWT).
Postoperative Protocol and Aftertreatment
The postoperative rehabilitation and medical management are just as critical as the surgical debridement in ensuring the eradication of subacute osteomyelitis.
Immobilization and Weight Bearing
- Immediately postoperatively, apply a well-padded, long leg posterior plaster splint (for lower extremity lesions).
- Positioning is critical to prevent contractures: the foot must be in a neutral position, the ankle at exactly 90 degrees, and the knee at 20 degrees of flexion.
- The patient is kept strictly non-weight bearing initially.
- Once the surgical wound has healed (typically 10 to 14 days), the splint is removed. The patient is then transitioned to protected weight bearing with crutches, advancing as tolerated and guided by radiographic evidence of bone consolidation.
Antimicrobial Therapy
- Postoperative antibiotic therapy must be guided by culture sensitivities and directed by an infectious disease specialist.
- While awaiting final cultures, broad-spectrum intravenous antibiotics are initiated.
- Generally, a comprehensive 6-week course of intravenous antibiotics is required to penetrate the sclerotic bone and eradicate residual microscopic disease. In select cases with highly susceptible organisms, an early transition to highly bioavailable oral antibiotics may be considered.
Long-Term Follow-Up
- Orthopaedic and infectious disease follow-up must be continued for at least 1 year postoperatively.
- Serial radiographs should be obtained at 6 weeks, 3 months, 6 months, and 1 year to monitor for the resolution of the lytic lesion, incorporation of any bone graft (if used subsequently), and to ensure there is no recurrence of the infection or signs of physeal arrest in pediatric patients.
Transition to Chronic Osteomyelitis: The Compromised Host
While subacute osteomyelitis can often be eradicated with the protocols described above, failure of treatment or delayed presentation can lead to chronic osteomyelitis. Surgery is not always the best option in these advanced stages, especially in physiologically compromised patients.
Consider an ambulatory, immunocompromised host with multiple medical comorbidities presenting with chronic osteomyelitis of the femur. For this patient, who might not survive the extensive surgical stress required to achieve a complete marginal resection of the infected bone, less aggressive alternatives must be considered. Limited surgical debridement combined with lifelong suppressive antibiotics and aggressive nutritional support may be the most appropriate path to limit the frequency of sinus drainage and manage pain. The treatment course and the definition of a "successful outcome" must be highly individualized.
Cierny-Mader Classification
To guide these complex decisions, Cierny and Mader developed a comprehensive classification system for chronic osteomyelitis based on physiological and anatomical criteria.
Physiological Criteria (Host Status):
* Class A: Normal hosts with a normal physiological, immune, and vascular response to infection and surgery.
* Class B: Compromised hosts with deficient wound healing capabilities (e.g., diabetics, smokers, immunocompromised).
* Class C: Hosts for whom the results of the required surgical treatment are potentially more damaging or life-threatening than the presenting condition itself.
Anatomical Criteria:
* Type I: Medullary lesion, characterized primarily by endosteal disease.
* Type II: Superficial lesion, typically involving the outer cortex.
* Type III: Localized lesion, involving full-thickness cortical sequestration but maintaining bone stability.
* Type IV: Diffuse lesion, involving circumferential disease requiring segmental resection and resulting in mechanical instability.
Understanding this transition from subacute to chronic disease underscores the importance of prompt, accurate diagnosis and meticulous surgical execution when managing subacute hematogenous osteomyelitis.
You Might Also Like
