Comprehensive Management of Atypical and Complicated Osteomyelitis: Pathological Fractures, CRMO, and Specific Pathogens
Key Takeaway
Pathological fractures in osteomyelitis present a complex biomechanical and biological challenge. Management requires aggressive surgical debridement, rigid external fixation, and dead space management. This guide explores the pathophysiology, surgical techniques, and evidence-based protocols for infected fractures, chronic recurrent multifocal osteomyelitis (CRMO), Pseudomonas puncture wounds, and anaerobic bone infections, providing orthopedic surgeons with a comprehensive framework for limb salvage and infection eradication.
PATHOLOGICAL FRACTURE IN OSTEOMYELITIS
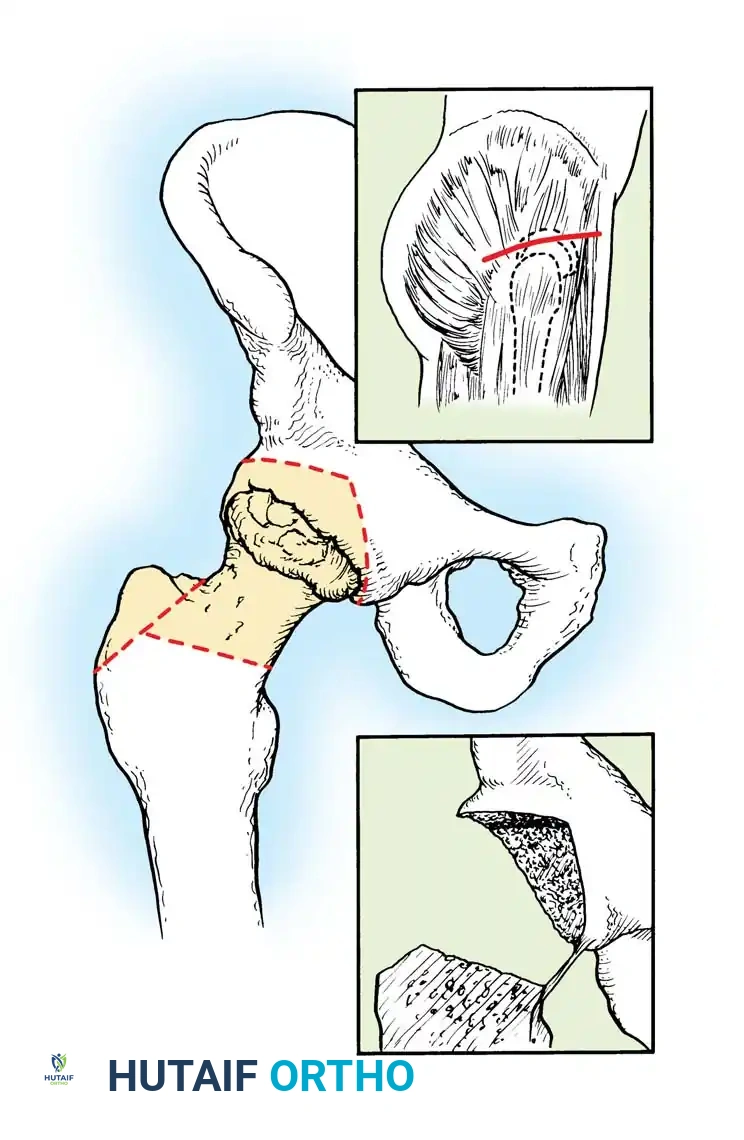
The occurrence of a pathological fracture in the setting of osteomyelitis represents a catastrophic failure of bone biomechanics secondary to infectious osteolysis and altered bone remodeling. The management of these fractures is fundamentally different from that of aseptic traumatic fractures, requiring a dual-phase approach: absolute eradication of the infection and subsequent or concurrent mechanical stabilization.
Pathophysiology and Biomechanics
Pathological fractures in osteomyelitis occur via two distinct pathophysiological mechanisms, depending on the chronicity of the infection:
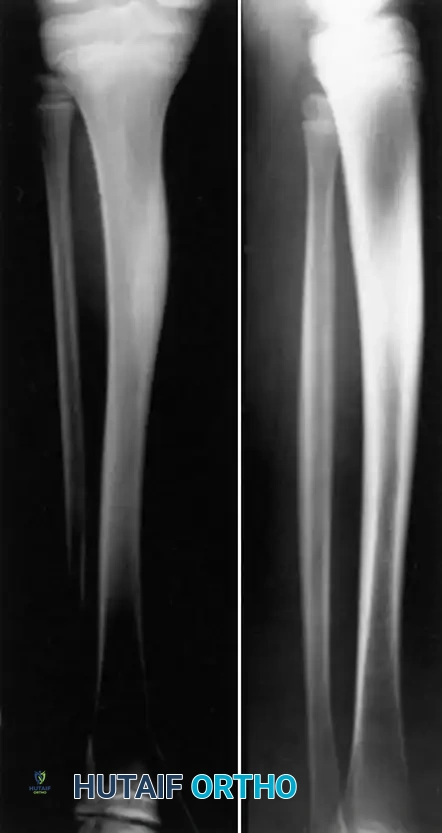
- Acute and Subacute Stages: During the early phases of pyogenic osteomyelitis, aggressive osteoclastic resorption outpaces osteoblastic bone formation. The host attempts to wall off the infection by forming an involucrum (new bone formation around the infected, necrotic diaphysis). However, before this involucrum matures and achieves sufficient cortical thickness, the structural integrity of the long bone shaft is severely compromised. Immobilization is critical during this phase; without it, physiological loading can easily result in a fracture through the weakened, hyperemic bone.
- Chronic Stage: In chronic osteomyelitis, the bone undergoes extensive sclerosis as a reactive process. While sclerotic bone appears radiographically dense, it is biomechanically inferior. The Haversian systems are obliterated, leading to avascularity, loss of elasticity, and extreme brittleness. This dense bone acts as a stress riser, and minor torsional or bending forces can propagate microcracks that rapidly culminate in a complete fracture.
Surgical Principles and Fixation Strategies
Whatever the underlying chronicity, the primary objective remains the thorough surgical eradication of the infection.
Surgical Pearl: Never compromise the thoroughness of the debridement to preserve bone stock for fixation. Dead bone is a harbor for biofilm. Radical resection of all necrotic bone (sequestrum) and infected soft tissue until punctate cortical bleeding (the "paprika sign") is observed is mandatory.
Once debridement is complete, the bone fragments must be realigned and immobilized.
Avoidance of Internal Fixation:
The use of standard internal fixation devices—such as compression plates and intramedullary nails—is generally contraindicated in the presence of active intramedullary infection. Metallic implants provide an ideal substrate for bacterial glycocalyx (biofilm) formation, which shields bacteria from systemic antibiotics and host immune responses. While antibiotic-coated intramedullary nails have seen limited use in highly specialized limb salvage scenarios, standard internal fixation should be avoided whenever possible.
Preferred Modalities:
* External Fixation: This is the gold standard for stabilizing infected pathological fractures. Monolateral frames can be used for simple diaphyseal stabilization, but circular frames (Ilizarov technique) or hexapod systems offer superior biomechanical stability, allowing for early weight-bearing which stimulates bone healing. External fixation pins should be placed outside the zone of infection to prevent pin-tract seeding.
* Cast Immobilization: In pediatric patients or non-weight-bearing upper extremity fractures with minimal displacement, rigid cast immobilization may suffice, provided the infection is adequately drained and soft tissues are accessible for wound care (e.g., via a windowed cast).
Management of Segmental Bone Defects
Because aggressive debridement is required, significant segmental bone loss is a frequent complication. The fracture will slowly heal as the infection subsides, but critical-sized defects will not bridge spontaneously. Reconstruction options include:
* Autogenous Bone Grafting: For defects less than 2-3 cm, cancellous autograft (typically from the iliac crest) can be utilized once the infection is clinically and serologically cleared.
* The Masquelet Technique (Induced Membrane): A two-stage procedure where a polymethylmethacrylate (PMMA) antibiotic spacer is placed in the defect. After 6-8 weeks, a vascularized pseudomembrane forms around the spacer. The spacer is removed, and the void is packed with autogenous bone graft.
* Bone Transport (Distraction Osteogenesis): Utilizing the Ilizarov technique, a corticotomy is performed in healthy bone adjacent to the defect, and a bone segment is gradually transported across the defect. This simultaneously addresses bone loss and soft tissue defects.
* Vascularized Osseous Grafts: Free vascularized fibular grafts are indicated for massive defects (typically >6 cm), providing immediate structural support and a robust blood supply to the infected bed.
CHRONIC RECURRENT MULTIFOCAL OSTEOMYELITIS (CRMO)
Chronic recurrent multifocal osteomyelitis (CRMO) is a rare, sterile, inflammatory bone disease that presents a significant diagnostic and therapeutic challenge. First described by Giedion et al. in 1972, it is now recognized as the pediatric equivalent of SAPHO syndrome (Synovitis, Acne, Pustulosis, Hyperostosis, Osteitis) and falls under the umbrella of autoinflammatory bone disorders.
Clinical Presentation
CRMO is characterized by an insidious onset of mild-to-moderate deep bone pain, accompanied by localized signs of inflammation (swelling, warmth, tenderness) over the affected areas. The hallmark of the disease is its relapsing-remitting nature; symptoms wax and wane unpredictably over months or years.
Systemic manifestations are generally mild, though patients may present with low-grade fevers. A highly specific associated condition is palmar-plantar pustulosis, a dermatological manifestation characterized by sterile, pustular rashes on the soles of the feet and palms of the hands.
Radiographic and Advanced Imaging Evaluation
The disease exhibits a strong predilection for the metaphyses of long bones, particularly the tibia, distal femur, and clavicle. Yu et al. also reported the frequent occurrence of vertebra plana (complete vertebral body collapse) associated with this condition.
Radiographically, early lesions are predominantly osteolytic and often bilaterally symmetrical. As the disease progresses into the chronic phase, varying degrees of reactive sclerosis and hyperostosis become prominent.

Clinical Pearl: While whole-body technetium-99m (Tc-99m) bone scanning historically demonstrated bilaterally symmetrical areas of increased radiotracer uptake, Whole-Body MRI (WB-MRI) has now superseded scintigraphy as the gold standard. MRI is highly sensitive for detecting asymptomatic multifocal marrow edema and is critical for excluding pyogenic involvement or malignant processes (e.g., Ewing sarcoma, leukemia).
Diagnosis and Biopsy Indications
CRMO is fundamentally a diagnosis of exclusion. Because the clinical and radiographic presentation can mimic subacute pyogenic osteomyelitis or primary bone malignancy, a biopsy is often considered.
However, in typical cases presenting with classic multifocal, symmetrical lesions and associated palmar-plantar pustulosis, a biopsy may not be strictly necessary. If the diagnosis is in doubt, a confirmatory open or core needle biopsy is indicated.
Histopathology: Biopsy specimens typically reveal non-specific acute and chronic inflammation, with polymorphonuclear leukocytes in early lesions and lymphocytes/plasma cells in older lesions. Crucially, microbiological cultures are almost always negative.
Evidence-Based Treatment Protocols
Because CRMO is an autoinflammatory rather than an infectious process, antibiotic therapy is strictly contraindicated if cultures are negative.
- First-Line Therapy: Nonsteroidal anti-inflammatory drugs (NSAIDs) are the cornerstone of initial treatment. They provide significant pain relief and can induce clinical remission in up to 50-60% of patients.
- Second-Line Therapy: For NSAID-refractory cases, or cases involving the spine (vertebra plana) where structural collapse is a risk, bisphosphonates (e.g., pamidronate) are highly effective. They inhibit osteoclastic activity and possess intrinsic anti-inflammatory properties.
- Biologic DMARDs: Tumor necrosis factor-alpha (TNF-α) inhibitors (e.g., etanercept, infliximab) are utilized in severe, recalcitrant cases.
- Historical Treatments: Cases of disease remission after treatment with interferon-gamma have been reported, though absolute proof of causality remains elusive due to the naturally relapsing-remitting nature of the disease.
Generally, symptoms continue to recur over a period of 2 to 5 years before the disease spontaneously "burns out." The disease is generally self-limiting, and the long-term prognosis is excellent, provided that structural complications (like physeal arrest or vertebral collapse) are prevented.
OSTEOMYELITIS AFTER PUNCTURE WOUND OF THE FOOT
Puncture wounds to the plantar aspect of the foot are ubiquitous pediatric injuries. While most heal uneventfully, a specific and highly documented complication is the development of Pseudomonas aeruginosa osteomyelitis.
Pathogenesis and Microbiology
The association between Pseudomonas and puncture wounds is directly linked to modern footwear. Pseudomonas aeruginosa thrives in the moist, warm environment of the inner foam lining of athletic sneakers. When a nail or sharp object penetrates the shoe, it inoculates the deep plantar fascia, joint spaces, or periosteum with this specific pathogen.
Clinical Evaluation
The clinical presentation is classically biphasic:
1. Initial Phase: The patient experiences acute pain from the initial puncture, which typically diminishes over the first 24 to 48 hours.
2. Infectious Phase: Pain, erythema, and swelling dramatically increase 2 to 4 days post-injury. The patient often becomes unable to bear weight.
Surgical Warning: Do not dismiss a delayed onset of pain following a puncture wound as simple soft-tissue inflammation. A high index of suspicion for deep space infection and osteomyelitis must be maintained. Plain radiographs may be normal for up to 10-14 days; MRI is the imaging modality of choice for early detection of marrow edema and deep abscesses.
Surgical Management and Antibiotic Stewardship
Superficial incision and drainage in the emergency department are inadequate for established Pseudomonas osteomyelitis.
Surgical Approach:
* The patient is positioned supine, and a tourniquet is utilized for a bloodless field.
* A longitudinal plantar incision is made, avoiding weight-bearing pads if possible, but prioritizing direct access to the puncture tract.
* The tract must be meticulously excised. Retained foreign bodies (e.g., pieces of rubber sole, sock fibers) are common and must be removed.
* If the infection has penetrated a joint (often the metatarsophalangeal joint) or bone, aggressive curettage and debridement of the necrotic cartilage and infected bone are mandatory.
* The wound is thoroughly irrigated and typically left open to heal by secondary intention or closed over a drain, depending on the extent of contamination.
Antibiotic Protocol:
Empiric therapy must cover Pseudomonas. Jacobs et al. suggested that 7 days of targeted intravenous antibiotics (e.g., ceftazidime, cefepime, or an antipseudomonal penicillin) following aggressive surgical debridement is adequate. However, contemporary literature and other authors often recommend a longer course, typically transitioning to a highly bioavailable oral fluoroquinolone (like ciprofloxacin, if age-appropriate and risks are weighed) for a total of 3 to 6 weeks, guided by clinical response and normalization of inflammatory markers (CRP/ESR).
ANAEROBIC OSTEOMYELITIS
Historically underdiagnosed, anaerobic bacteria are increasingly recognized as a critical cause of severe, tissue-destructive osteomyelitis. In a landmark series from the Mayo Clinic, 40 out of 182 patients (22%) undergoing surgery for osteomyelitis were found to harbor anaerobic bacteria in their operative cultures.
Pathogenesis and Associated Conditions
Anaerobic soft-tissue and bone infections rarely occur in healthy, well-vascularized tissue. They are opportunistic pathogens that thrive in environments with a low oxidation-reduction (redox) potential. Consequently, anaerobic osteomyelitis is most frequently associated with:
* Diabetic Gangrene and Neuropathy: Microvascular disease and ischemia create the perfect hypoxic environment.
* Severe Trauma: Crush injuries with extensive soft-tissue devitalization.
* Chronic Ischemic Extremities: Peripheral vascular disease.
Common isolates include Bacteroides fragilis, Peptostreptococcus, and Clostridium species. These infections are frequently polymicrobial, acting synergistically with aerobic bacteria to accelerate tissue destruction.
Clinical Presentation and Diagnosis
The clinical hallmarks of anaerobic infection include:
* A profoundly putrid, foul-smelling discharge.
* The presence of gas in the soft tissues (crepitus on palpation or visible on plain radiographs).
* Extensive, rapidly progressive tissue necrosis that tends to burrow aggressively through subcutaneous and fascial planes.
Microbiological Culturing Protocols:
The demonstration of anaerobic bacteria requires rigorous adherence to specific culturing protocols. Hall, Fitzgerald, and Rosenblatt strongly recommended that deep tissue specimens from all patients with osteomyelitis be cultured for anaerobes.
* Swabs are generally inadequate. Deep tissue or bone fragments must be obtained.
* Clinical material must be cultured immediately after collection.
* Specimens must be placed in specialized anaerobic transport media.
* The laboratory must use fresh media with proper anaerobic conditions, subculturing colonies immediately upon removal from the anaerobic environment to prevent oxygen toxicity to the bacteria.
Surgical and Medical Management
The treatment of anaerobic osteomyelitis is an orthopedic emergency requiring aggressive intervention.
- Radical Surgical Debridement: Antibiotics cannot penetrate the ischemic, necrotic tissue where anaerobes thrive. Wide surgical drainage, extensive fasciotomies, and radical resection of all necrotic bone and soft tissue are required. In cases of advanced diabetic gangrene with extensive midfoot or hindfoot anaerobic osteomyelitis, partial or major limb amputation (e.g., Syme or below-knee amputation) is often the most definitive and life-saving procedure.
- Dead Space Management: Following resection, the resulting dead space must be managed, often with antibiotic-impregnated PMMA beads (utilizing heat-stable antibiotics like tobramycin or vancomycin, though systemic coverage is primary for anaerobes) or negative pressure wound therapy (NPWT).
- Antimicrobial Therapy: High-dose, targeted intravenous antibiotics are initiated based on culture and sensitivity studies. Metronidazole, clindamycin, or beta-lactam/beta-lactamase inhibitor combinations (e.g., piperacillin-tazobactam) are mainstays of therapy.
- Hyperbaric Oxygen Therapy (HBOT): As an adjunct, HBOT can be utilized to increase tissue oxygen tension, which is directly bactericidal to strict anaerobes and enhances the oxidative burst of host macrophages.
You Might Also Like