Comprehensive Guide to Peripheral Nerve Anatomy and Surgical Repair
Key Takeaway
Peripheral nerve injuries present complex reconstructive challenges. Despite microsurgical advancements, functional recovery remains modest. This comprehensive guide details the gross and microscopic anatomy of spinal nerves, dermatomal architecture, and evidence-based surgical principles for nerve repair. It provides step-by-step approaches for sciatic, peroneal, and tibial nerve reconstruction, emphasizing critical timing, fascicular alignment, and postoperative rehabilitation to optimize patient outcomes in orthopedic practice.
INTRODUCTION TO PERIPHERAL NERVE INJURIES
Peripheral nerve injuries represent a formidable challenge in operative orthopedics, often resulting in profound functional impairment and debilitating neuropathic pain. Despite numerous contemporary advances in microsurgical techniques, the utilization of interfascicular nerve grafting, and high-resolution intraoperative magnification, many foundational treatment principles remain rooted in the extensive World War II experiences documented by Seddon and Woodhall.
Current research focusing on pharmacological agents, immune system modulation, neurotrophic enhancing factors, and bioabsorbable entubulation chambers shows immense promise in preclinical models. However, these innovations have thus far seen limited clinical application. Consequently, the clinical outcomes of peripheral nerve repair remain modest, with historically only about 50% of patients regaining highly useful, independent motor and sensory function.
This masterclass delineates the gross and microscopic anatomy of the peripheral nervous system, the biomechanics of nerve injury, and the definitive surgical approaches for the sciatic, peroneal, and tibial nerves.
Clinical Pearl: The ultimate success of a peripheral nerve repair is dictated not only by the precision of the microsurgical coaptation but also by the critical limit of delay before surgery, the age of the patient, and the distance the regenerating axon must travel to its target end-organ.
GROSS ANATOMY OF THE SPINAL NERVES
The architecture of the peripheral nervous system begins at the spinal cord. Each segmental spinal nerve is formed at or near its respective intervertebral foramen by the union of its dorsal (sensory) root with its ventral (motor) root.
In the majority of the thoracic segments, these mixed spinal nerves retain their strict segmental autonomy, supplying one intercostal dermatomal and myotomal segment. However, in virtually all other segments of the spinal axis, the spinal nerves arborize and join with adjacent segments to form complex plexuses. These plexuses innervate the limbs or specialized body segments that have lost their primitive myomeric pattern during embryological development.
A total of 31 mixed spinal nerves leave their respective intervertebral foramina on each side of the vertebral column to innervate the homolateral trunk and extremities:
* Cervical: 8 pairs
* Thoracic: 12 pairs
* Lumbar: 5 pairs
* Sacral: 5 pairs
* Coccygeal: 1 pair
COMPONENTS OF MIXED SPINAL NERVES
A typical mixed spinal nerve is a highly organized conduit comprising three distinct functional components: motor, sensory, and sympathetic fibers.
- Motor Component: Several rootlets leave the anterolateral sulcus of the spinal cord and unite to form each ventral motor root. The efferent fibers traversing these roots arise from the anterior horn cells of the spinal cord and are responsible for innervating skeletal musculature.
- Sensory Component: The afferent sensory fibers arise from pain, thermal, tactile, and stretch receptors located peripherally. The cell bodies for these unipolar neurons are housed within the dorsal root ganglia, with their proximal axons entering the posterolateral sulcus of the spinal cord.
- Sympathetic Component: Mixed spinal nerves, having exited the intervertebral foramina, receive their sympathetic component via the gray rami communicantes and promptly branch into anterior and posterior primary rami.

RAMI DISTRIBUTION AND PLEXUS FORMATION
The posterior primary rami are directed posteriorly to supply the paraspinal musculature and the cutaneous territories along the posterior aspect of the trunk, neck, and head. With the exception of the upper three cervical posterior rami (which are larger than their anterior counterparts to supply the posterior scalp and craniocervical musculature), posterior primary rami are relatively small.
The major portion of each spinal nerve continues laterally as the anterior primary ramus. The anterior primary rami of all cervical, the first thoracic, and all lumbosacral nerves join to form the major neural plexuses:
* Cervical Plexus: Formed by the upper four cervical anterior rami (C1-C4).
* Brachial Plexus: Formed by the lower four cervical and first thoracic anterior rami (C5-T1).
* Lumbar Plexus: Formed by the first three and a portion of the fourth lumbar anterior rami (L1-L4).
* Lumbosacral Plexus: Formed by the sacral anterior rami, the fifth lumbar, and a portion of the fourth lumbar rami (L4-S4).
Surgical Warning: Because the fibers of any single mixed spinal nerve may be distributed through several peripheral nerves via plexus formation, a proximal root avulsion will present with a complex, multi-nerve deficit pattern that must be carefully differentiated from a distal peripheral nerve trunk injury.
DERMATOMAL AND MYOTOMAL ARCHITECTURE
The specific area of skin supplied by the afferent fibers of a single spinal root is termed a dermatome. While segmental dermatomal patterns are well preserved and easily mapped in the thoracic region, they become highly complex in the appendicular skeleton.
This alteration of the metameric pattern results from the embryological migration of dermatomes and myotomes into the developing limb buds. The enlargement and prolongation of the limb bud markedly alter the myotomal pattern, resulting in the fusion of some myotomes and the extended migration of others.
Migration accounts for the displacement of midcervical dermatomes along the lateral aspect of the arm and radial aspect of the forearm, and the displacement of lower cervical and upper thoracic dermatomes along the medial aspect of the arm and ulnar aspect of the forearm. Lumbar and sacral dermatomal alignment along the lower extremities is similarly explained by this embryological rotation and extension. The distinct line separating the more rostral segmental dermatomes from the more caudal ones is known as the axial line, which can be traced proximally into the spinal axis.
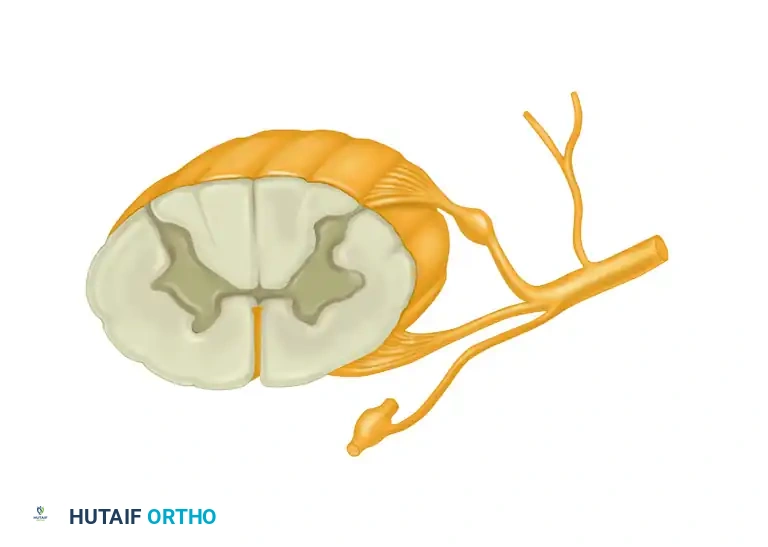
MICROSCOPIC ANATOMY AND NEURAL BIOMECHANICS
Understanding the microscopic architecture of a peripheral nerve is paramount for successful microsurgical repair. Each nerve fiber (axon) is a direct cytoplasmic extension of a dorsal root ganglion cell (sensory), an anterior horn cell (motor), or a postganglionic sympathetic neuron.
AXONAL MYELINATION AND SCHWANN CELLS
Sensory and motor nerves contain both unmyelinated and myelinated fibers, typically in a ratio of 4:1.
* Unmyelinated Fibers: Several axons are enveloped by a single Schwann cell, providing structural support but lacking a true myelin sheath.
* Myelinated Fibers: A single Schwann cell rotates around a single axon, forming a multilaminated myelin sheath. This sheath acts as an electrical insulator, facilitating rapid saltatory conduction.
The segment of a myelinated nerve fiber enclosed by a single Schwann cell is referred to as an internode, varying in length from 0.1 to 1.8 mm (heavily myelinated fibers possess longer internodes). The junction where one Schwann cell ends and the next begins is relatively devoid of myelin and is termed the node of Ranvier.
CONNECTIVE TISSUE ENVELOPES
The structural integrity and biomechanical properties of a peripheral nerve are dictated by its connective tissue layers:
1. Endoneurium: A delicate veil of fibrous tissue surrounding the individual axon and its Schwann cell. Seen longitudinally, it forms a tube encircling the sheath.
2. Perineurium: A dense, metabolically active layer of connective tissue that surrounds a cluster of endoneurial tubes to form a fascicle (or funicle, as termed by Sunderland). The perineurium provides the primary tensile strength of the nerve and acts as a vital blood-nerve barrier.
3. Epineurium: The outermost layer of loose areolar connective tissue that encases the entire group of fascicles, forming the definitive mixed peripheral nerve. It also contains the extrinsic vasa nervorum (blood supply).
Biomechanics of Nerve Repair: The perineurium is the strongest layer. During microsurgical repair, sutures must engage the epineurium (and occasionally the perineurium in group fascicular repairs) without strangulating the delicate endoneurial contents. Excessive tension across the repair site induces ischemia within the vasa nervorum, leading to profound fibroplasia and repair failure.
SURGICAL PRINCIPLES OF PERIPHERAL NERVE REPAIR
INDICATIONS AND CRITICAL LIMIT OF DELAY
The timing of nerve repair is dictated by the mechanism of injury. Sharp transections (e.g., glass lacerations) warrant immediate primary repair (within 72 hours). Blunt trauma, crush injuries, or high-velocity gunshot wounds require delayed secondary repair (at 2 to 3 weeks) to allow the zone of injury to demarcate, ensuring that resection back to healthy, bleeding fascicles is accurate.
The critical limit of delay refers to the maximum time elapsed between injury and repair before irreversible motor end-plate degradation and muscle atrophy occur. For most major nerves, motor recovery drops precipitously if repair is delayed beyond 6 to 12 months. Sensory receptors (e.g., Meissner and Pacinian corpuscles) can survive longer, allowing for sensory recovery even with repairs performed up to 24 months post-injury.
METHODS OF CLOSING GAPS
When a nerve is transected, the intrinsic elasticity of the nerve and muscle retraction create a gap. Closing this gap without tension is the most critical step in nerve surgery.
1. Mobilization: Extensive proximal and distal neurolysis can provide several centimeters of length.
2. Joint Positioning: Flexing adjacent joints (e.g., the knee for the sciatic/tibial nerve) can reduce tension, though extreme flexion should be avoided to prevent postoperative stretch-induced ischemia during rehabilitation.
3. Nerve Grafting: If a tension-free primary coaptation is impossible, interfascicular nerve grafting (typically utilizing the reversed sural nerve) is the gold standard.
OPERATIVE APPROACHES TO SPECIFIC NERVES
THE SCIATIC NERVE
The sciatic nerve is the largest nerve in the body, formed by the L4-S3 roots. It is highly susceptible to injury during posterior approach total hip arthroplasty, pelvic trauma, or deep gluteal penetrating wounds.
Positioning and Approach:
* The patient is positioned prone.
* The surgical incision begins at the posterior superior iliac spine, curves distally and laterally toward the greater trochanter, and extends distally down the posterior midline of the thigh.
* The gluteus maximus is split in line with its fibers or reflected from its femoral insertion to expose the short external rotators.
* The sciatic nerve is identified as it exits the greater sciatic foramen, typically inferior to the piriformis muscle.
Results of Suture and Critical Limit of Delay:
Suture of the sciatic nerve yields variable results. Because the nerve is a composite of the tibial and common peroneal divisions, the tibial division consistently demonstrates superior motor recovery compared to the peroneal division. The critical limit of delay for sciatic nerve repair is strictly 6 to 9 months; beyond this, the distance to the distal musculature of the leg precludes functional motor reinnervation before irreversible muscle fibrosis occurs.
THE COMMON, SUPERFICIAL, AND DEEP PERONEAL NERVES
The common peroneal nerve is uniquely vulnerable as it winds around the subcutaneous neck of the fibula. Injuries here result in a profound "foot drop" (loss of ankle dorsiflexion and toe extension).
Examination and Treatment:
Clinical examination reveals an inability to dorsiflex the ankle (deep peroneal nerve) and loss of eversion (superficial peroneal nerve), accompanied by sensory loss over the dorsum of the foot.
Surgical Approach:
* The patient is positioned supine with a bump under the ipsilateral hip to internally rotate the leg.
* An incision is made along the biceps femoris tendon, curving anteriorly around the fibular neck.
* The nerve is decompressed by releasing the superficial fascia of the peroneus longus.
Results of Suture and Tendon Transfer:
The results of primary suture or grafting of the peroneal nerve are notoriously poor, often cited as the least successful of all major lower extremity nerves. Due to this unpredictable recovery, Tendon Transfer for Peroneal Nerve Paralysis is frequently indicated. The standard reconstructive protocol involves transferring the posterior tibial tendon (PTT) through the interosseous membrane to the dorsum of the foot (often inserted into the lateral cuneiform or split between the tibialis anterior and peroneus brevis) to restore active dorsiflexion and provide a balanced foot.
THE TIBIAL NERVE
The tibial nerve provides critical motor innervation to the posterior compartment of the leg (plantar flexion) and intrinsic muscles of the foot, as well as vital protective sensation to the plantar aspect of the foot.
Approach to the Tibial Nerve in the Popliteal Fossa:
* The patient is positioned prone.
* A classic S-shaped incision is utilized across the popliteal fossa to avoid scar contracture across the flexion crease. The incision starts posterolaterally in the distal thigh, crosses the joint line transversely, and extends posteromedially down the leg.
* The deep fascia is incised, and the nerve is identified superficial and lateral to the popliteal vein and artery.
* Distal exposure requires splitting the two heads of the gastrocnemius and carefully dividing the soleal sling.
Results of Suture of the Tibial Nerve:
Unlike the peroneal nerve, the tibial nerve exhibits excellent potential for functional recovery following microsurgical repair. Restoration of protective plantar sensation is highly achievable and is critical for preventing neuropathic ulcerations. Motor recovery of the gastrocnemius-soleus complex is generally robust if repaired within the critical limit of delay (ideally within 6 months of injury).
POSTOPERATIVE PROTOCOLS AND REHABILITATION
The postoperative management of peripheral nerve repairs is designed to protect the delicate micro-coaptation while preventing debilitating joint contractures.
- Immobilization Phase (0-3 Weeks): The limb is immobilized in a well-padded orthosis or cast in a position that minimizes tension on the repair site. For example, following a sciatic or tibial nerve repair, the knee is immobilized in 30 to 45 degrees of flexion.
- Graduated Mobilization (3-6 Weeks): A hinged orthosis is applied. The joint is gradually extended by 10 to 15 degrees per week. This controlled elongation induces a gradual viscoelastic creep in the nerve, preventing sudden traction failure of the epineurial sutures.
- Neuromuscular Re-education (6+ Weeks): Once full range of motion is achieved, aggressive physical therapy commences. Electrical stimulation of denervated muscle remains controversial but may be used to maintain muscle bulk. Biofeedback and sensory re-education are critical as regenerating axons reach their target end-organs.
- Monitoring: Tinel's sign is monitored monthly. An advancing Tinel's sign (approximately 1 mm per day or 1 inch per month) indicates successful axonal sprouting and progression down the endoneurial tubes. Failure of Tinel's sign to advance after 3 to 4 months may indicate a neuroma-in-continuity requiring surgical re-exploration.
Clinical Pearl: Never substitute hope for clinical evidence. If a primary nerve repair fails to show clinical or electromyographic (EMG) evidence of reinnervation by 4 to 6 months, early re-exploration or definitive tendon transfer should be aggressively pursued to optimize the patient's functional outcome.
You Might Also Like

