Pathophysiology and Clinical Evaluation of Peripheral Nerve Injuries
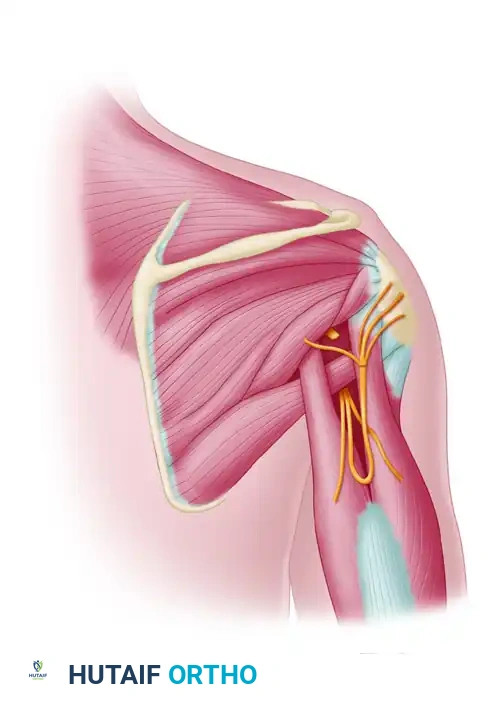
Key Takeaway
Peripheral nerve injuries initiate a complex cascade of motor, sensory, and autonomic deficits. Accurate clinical evaluation requires a profound understanding of Wallerian degeneration, electromyographic timelines, and sensory recovery sequences. This guide details the British Medical Research Council (BMRC) grading systems, the pathophysiology of muscle atrophy, and the critical distinction between collateral sensory sprouting and true axonal regeneration to guide surgical decision-making and postoperative rehabilitation.
PATHOPHYSIOLOGY OF PERIPHERAL NERVE INJURIES
The severance or severe crush of a peripheral nerve initiates a profound and highly predictable sequence of physiological, histological, and macroscopic changes in the distal target organs. Understanding the precise timeline of these changes—encompassing motor, sensory, and autonomic domains—is paramount for the orthopedic surgeon. It dictates the timing of electrodiagnostic testing, guides the indications for surgical exploration, and establishes the framework for postoperative rehabilitation.
When a peripheral nerve is severed at a given level (neurotmesis), all motor, sensory, and sympathetic function of the nerve distal to that level is immediately abolished. The distal axonal segment undergoes Wallerian degeneration, a process of axonal breakdown and myelin clearance mediated by Schwann cells and macrophages, which prepares the endoneurial tubes for potential future axonal regeneration.
Motor System Deficits and Muscle Atrophy
Following complete nerve transection, all muscles supplied by branches of the nerve distal to the level of injury are paralyzed and become acutely atonic. The denervated muscle undergoes a rapid and progressive cascade of atrophic changes.
Electromyographic (EMG) Timeline:
* 0 to 7 Days: Immediately following injury, the muscle is paralyzed, but the distal axon remains electrically excitable for a few days until Wallerian degeneration is complete. EMG during this hyperacute phase will show an absence of voluntary motor unit action potentials (MUAPs), but Wallerian degeneration has not yet altered the resting muscle membrane stability.
* 8 to 14 Days: Significant changes on electromyography (EMG) first become apparent. Transient fibrillation potentials upon needle insertion may be observed, indicating early denervation hypersensitivity of the muscle membrane.
* 2 to 4 Weeks: Spontaneous fibrillations and positive sharp waves become clearly evident at rest. This electrophysiological milestone coincides with the onset of profound histological atrophic changes within the muscle fibers themselves.
Macroscopic and Histological Muscle Changes:
Atrophy of muscle bulk progresses aggressively, reaching a 50% to 70% reduction in total mass by the end of approximately 2 months. Following this initial rapid phase, atrophy continues at a much slower, insidious rate. Concurrently, the connective tissue component (fibrosis) within the muscle interstitium increases, replacing the lost contractile tissue.
Surgical Warning: The "Race Against Time" in Nerve Repair
Striations and motor endplate configurations are retained for slightly longer than 12 months. However, the empty endoneurial tubes shrink to about one-third of their normal diameter. If reinnervation does not occur within 12 to 18 months, the motor endplates degrade irreversibly. Complete disruption and fibrotic replacement of muscle fibers may become absolute after 3 years, rendering any subsequent nerve repair functionally useless for motor recovery.
CLINICAL EVALUATION OF MOTOR RETURN
Accurate documentation of motor recovery is essential for determining whether a nerve is regenerating conservatively or if surgical intervention (e.g., neurolysis, nerve grafting, or tendon transfers) is required.
Several modalities are utilized to evaluate motor return after peripheral nerve injuries. The foundation of this assessment involves testing muscle strength against gravity and against graded resistance. Adjunctive quantitative tools, such as pinch meters and grip dynamometers, alongside the evaluation of endurance, speed of movement, and isolated individual muscle function, help to objectively document the progress of motor return.
The BMRC Motor Grading System
The British Medical Research Council (BMRC) established the universally accepted system for assessing the return of muscle function after peripheral nerve injuries.
- M0: No contraction has returned.
- M1: Return of perceptible contraction in proximal muscles.
- M2: Return of perceptible contraction in proximal and distal muscles.
- M3: Return of function in proximal and distal muscles of such a degree that all important muscles are sufficiently powerful to act against resistance.
- M4: Return of function as in stage M3; in addition, all synergistic and independent movements are possible.
- M5: Complete recovery of muscle strength and function.
(Note: In the hand, "proximal muscles" are defined as extrinsic muscles originating in the forearm, whereas "distal muscles" are defined as the intrinsic muscles of the hand.)
Clinical Pearl: Beware of "Trick Movements"
When assessing motor grades, inexperienced examiners may be fooled by trick movements. For example, a patient with a radial nerve palsy may appear to actively extend their digits by vigorously flexing their wrist, utilizing the tenodesis effect of the extensor digitorum communis. Always isolate the joint and palpate the specific muscle belly or tendon during testing.
SENSORY SYSTEM DEFICITS AND ZONAL ANATOMY
Sensory loss following a peripheral nerve injury usually follows a definite, predictable anatomical pattern. However, the factor of sensory overlap from adjacent, uninjured nerves can frequently confuse inexperienced surgeons during the clinical examination.
To accurately map sensory deficits, the surgeon must understand the three distinct zones of sensory innervation:
- The Autonomous Zone (Isolated Zone): After severance of a peripheral nerve, only a relatively small area of complete sensory loss is found. This specific area is supplied exclusively by the severed nerve.
- The Intermediate Zone: A larger area of tactile and thermal anesthesia can be delineated, which corresponds more closely to the gross anatomical distribution of the nerve. This area features overlapping innervation from adjacent nerves.
- The Maximal Zone: When a nerve is intact, but all adjacent nerves are blocked or sectioned, the area of sensibility that remains exceeds the gross anatomical distribution of the intact nerve. This represents the absolute maximal territory that the nerve can supply.

The Illusion of Early Sensory Recovery
It has long been recognized that the autonomous zone becomes smaller during the first few days or weeks after an injury. This occurs long before true axonal regeneration is physiologically possible (axons regenerate at approximately 1 mm per day).
Historically, Livingston suggested this shrinkage was caused by the physical ingrowth of adjacent nerves. However, modern neurophysiology indicates that the resumption of, or increase in, function within pre-existing anastomotic branches from adjacent nerves is a much more plausible explanation.
Pitfall: Misinterpreting Autonomous Zone Shrinkage
This early decrease in the area of absolute sensory loss might be misinterpreted by an inexperienced surgeon as evidence of rapid nerve regeneration or as proof of an incomplete injury (axonotmesis/neuropraxia). This misinterpretation is dangerous, as it can be responsible for a needless and detrimental delay in the surgical exploration of a completely transected nerve.
CLINICAL EVALUATION OF SENSORY RETURN
The evaluation of sensory return after peripheral nerve injuries is critical regardless of the site of the injury, but it is of paramount importance in the upper extremity, where fine sensibility in the hand dictates overall limb function.
The BMRC Sensory Grading System
Similar to motor grading, the British Medical Research Council established a six-level grading scale for sensory return:
- S0: Complete absence of sensibility in the autonomous area.
- S1: Recovery of deep cutaneous pain within the autonomous area.
- S2: Return of some superficial cutaneous pain and tactile sensibility within the autonomous area.
- S3: Return of superficial cutaneous pain and tactile sensibility throughout the autonomous area, with the disappearance of overreaction (hypersensitivity/hyperpathia).
- S3+: Some recovery of two-point discrimination within the autonomous area.
- S4: Complete sensory recovery.
The Sequence of Sensory Recovery
Sensory modalities do not return simultaneously. They follow a highly specific sequence dictated by fiber diameter, myelination status, and the maturation of specific distal sensory receptors. Dellon, Curtis, and Edgerton outlined a predictable sequence of sensory recovery:
- Pain and Temperature: Mediated by small-diameter, unmyelinated (C fibers) and thinly myelinated (A-delta) fibers. These regenerate the fastest.
- 30 cps Vibratory Stimulus: Mediated by Meissner corpuscles.
- Moving Touch: Mediated by quickly adapting fibers and Pacinian corpuscles.
- Constant Touch: Mediated by slowly adapting fibers and Merkel discs.
- 256 cps Vibratory Stimulus: The final modality to return, requiring high-level receptor maturation.
Pathophysiological Rationale for the Sequence:
In injuries to the median and ulnar nerves, studies confirm that pinprick (pain) is the first perception to return. Investigators infer that this early return results from the faster regeneration rate of the small-diameter pain fibers. The larger-diameter touch fibers regenerate much more slowly.
Furthermore, the return of moving touch perception (Pacinian corpuscles) before the return of constant touch (Merkel discs) is explained by the differential maturation rates of the respective end-organ receptors, rather than by the diameter of the nerve fibers alone.
Advanced Sensory Testing Modalities
To accurately stage a patient's recovery and guide sensory reeducation, several advanced tests are employed:
- Two-Point Discrimination (Weber Test): This test has been shown to directly correlate with the return of functional hand use and object identification. Static two-point discrimination tests slowly adapting fibers, while moving two-point discrimination tests quickly adapting fibers.
- The Pick-Up Test (Moberg): A timed test designed to measure fine motor control, manual dexterity, and tactile gnosis. It requires the patient to pick up various small objects and place them in a container, first with eyes open, then closed.
- Triketohydrindene Hydrate (Ninhydrin) Printing Test: An objective test of sudomotor (sympathetic) function. Denervated skin does not sweat. The Ninhydrin test detects amino acids in sweat; a lack of color change indicates anhidrosis and, by proxy, complete sensory denervation.
TROPHIC AND AUTONOMIC CHANGES
Peripheral nerves carry sympathetic postganglionic fibers. Therefore, complete nerve injuries result in profound autonomic and trophic changes in the denervated territory.
- Sudomotor and Vasomotor Changes: The skin initially becomes warm and flushed due to the loss of sympathetic vasoconstrictor tone. Later, it becomes cold, mottled, and cyanotic. Sweating ceases entirely (anhidrosis), leading to dry, smooth skin that loses its normal papillary ridges and wrinkles.
- Osteoporosis: Severe nerve injuries are frequently associated with localized osteoporosis, often resembling Complex Regional Pain Syndrome (CRPS) or Sudeck's atrophy. These osteopenic changes occur most prominently in the distal phalanges of the thumb, index, and long fingers following median nerve injuries.
- Joint Stiffness and Ankylosis: Partial ankylosis resulting from the fibrosis of periarticular structures frequently develops. While these changes share similarities with the atrophy of disuse, they are significantly more severe and rapid in onset due to the complete loss of protective sensation and spontaneous nocturnal joint movements.
SURGICAL INDICATIONS AND POSTOPERATIVE PROTOCOLS
The comprehensive understanding of the aforementioned physiological effects directly dictates surgical decision-making.
Indications for Surgical Exploration
- Open Injuries with Known Transection: Immediate primary repair (within 72 hours) is indicated to align fascicles before retraction and scarring occur.
- Closed Injuries (e.g., Traction/Crush): These are typically observed for 3 months. If serial clinical examinations and EMG at 12 weeks show no evidence of motor unit action potentials (MUAPs) or advancing Tinel's sign across the zone of injury, surgical exploration, neurolysis, or nerve grafting is indicated.
Postoperative Sensory Reeducation
The system of evaluating moving touch, constant touch, vibratory stimulus, pinprick, and Weber two-point discrimination is not merely diagnostic; it is a screening method to determine specific exercises for sensory reeducation.
- Phase 1 (Early): Focuses on protective sensation. Patients are taught to visually compensate for the lack of pain and temperature perception to avoid burns and pressure sores.
- Phase 2 (Late): Once 30 cps vibration and moving touch return, cortical remapping exercises begin. Patients handle objects of varying textures (e.g., velvet, Velcro, sandpaper) with their eyes closed, attempting to identify them, thereby forcing the brain to interpret the newly arriving, albeit distorted, sensory signals.
You Might Also Like