Surgical Treatment of Dupuytren Disease
Introduction and Epidemiology
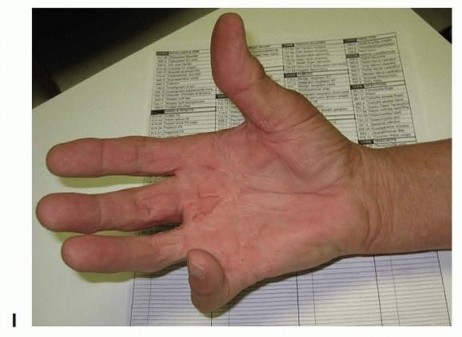
Dupuytren disease is a benign, progressive fibroproliferative disorder that primarily affects the palmar fascia of the hand, leading to debilitating digital contractures. Clinically, it is characterized by the abnormal proliferation of myofibroblasts and the excessive deposition of extracellular matrix proteins, specifically characterized by a pathologic shift toward type III collagen. While the condition predominantly manifests in the palmar and digital fascia, it is widely recognized as a systemic fibroproliferative diathesis. Secondary involvement of remote connective tissues can occur, presenting clinically as plantar fibromatosis (Ledderhose disease), penile fibromatosis (Peyronie disease), and dorsal proximal interphalangeal (PIP) joint nodules (Garrod nodes). The presence of these ectopic manifestations defines a more aggressive clinical phenotype known as "Dupuytren diathesis," which correlates with earlier onset and higher recurrence rates.

The etiology of Dupuytren disease is multifactorial but possesses a profound genetic foundation, supported by extensive population studies, family clustering, and genome-wide association studies (GWAS). Current genetic evidence points toward alterations in the Wnt signaling pathway, with chromosomes 6, 11, and 16 harboring primary susceptibility genes. The condition exhibits an autosomal dominant inheritance pattern with variable penetrance. It is most prevalent in individuals of Northern European descent, leading to its historical moniker, the "Viking disease." Epidemiologically, it exhibits a distinct male predominance (ratio of approximately 3:1 to 10:1 depending on the cohort), with men typically affected a decade earlier and experiencing a more severe, rapidly progressive clinical course than women.
Pathophysiologically, Dupuytren disease represents a sui generis condition that resembles no other known disorder. While it shares fundamental cellular mechanisms with normal wound healing—specifically the activation and proliferation of myofibroblasts—the critical pathophysiologic divergence lies in the perpetual, uninhibited progression of this proliferation. Normal wound healing eventually undergoes apoptosis and downregulates; Dupuytren disease features continuous, abnormal collagen deposition, leading to progressive tissue contracture. Luck's historical classification divides the disease into three distinct phases: the proliferative phase (nodule formation with high cellularity), the involutional phase (alignment of myofibroblasts along lines of tension), and the residual phase (acellular, dense collagenous cords). Historical attempts to classify this condition under inflammatory, infectious, or neoplastic categories have been definitively disproven.
Cellular and Molecular Pathogenesis
The biological core of the disease process is the myofibroblast, a specialized, differentiated cell exhibiting ultrastructural characteristics of both fibroblasts and smooth muscle cells. These cells express alpha-smooth muscle actin (α-SMA), allowing them to generate significant, sustained contractile forces via intracellular microfilaments that connect to the extracellular matrix through fibronexus junctions. As the disease progresses, the extracellular matrix undergoes a fundamental structural shift. The normal fascial architecture, composed predominantly of organized type I collagen, is rapidly replaced by disorganized type III collagen, altering the mechanical properties of the palmar fascia.
Cytokines and localized growth factors play a pivotal role in driving this fibroproliferative cascade. Transforming growth factor-beta (TGF-β), basic fibroblast growth factor (bFGF), and platelet-derived growth factor (PDGF) are heavily implicated in the recruitment, proliferation, and differentiation of fibroblasts into myofibroblasts. Specifically, TGF-β1 has been identified as the primary profibrotic cytokine, upregulating the expression of α-SMA and inhibiting matrix metalloproteinases (MMPs), thereby preventing the degradation of the newly synthesized extracellular matrix. Furthermore, localized microvascular ischemia within the palmar fascia leads to the generation of oxygen free radicals, which stimulate fibroblast proliferation and further exacerbate the fibrotic cascade.
Recent molecular analyses have also highlighted the dysregulation of the Wnt/β-catenin signaling pathway. In normal tissue homeostasis, β-catenin is continuously degraded. In Dupuytren tissue, aberrant Wnt signaling leads to the intracellular accumulation and nuclear translocation of β-catenin, where it acts as a transcription factor driving the expression of fibrogenic genes. Understanding these molecular pathways is critical, as they represent potential targets for future pharmacologic interventions aimed at arresting or reversing the disease process at a cellular level.
Surgical Anatomy and Biomechanics
A profound understanding of the normal and pathologic anatomy of the palmar fascia is the cornerstone of safe and effective surgical intervention in Dupuytren disease. The palmar fascia is a complex, three-dimensional network of connective tissue that stabilizes the palmar skin, protects underlying neurovascular structures, and compartmentalizes the hand. In Dupuytren disease, normal fascial structures (bands and ligaments) undergo hypertrophy and contracture to become pathologic cords.
Normal Fascial Anatomy
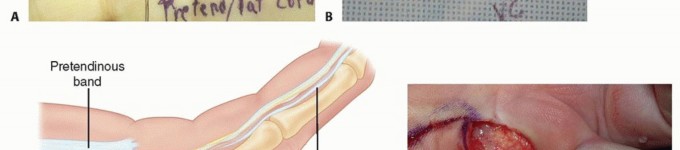
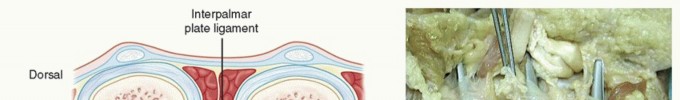
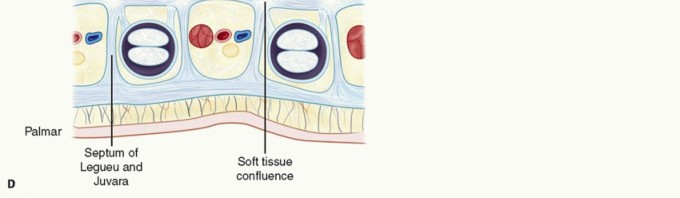
The normal palmar aponeurosis consists of longitudinal, transverse, and vertical fibers. The longitudinal fibers, known as the pretendinous bands, originate from the palmaris longus tendon (when present) and the transverse carpal ligament, radiating distally toward the digits. Deep to these lie the transverse fibers, forming the superficial transverse palmar ligament (Skoog's ligament), which notably is rarely involved in the pathologic process of Dupuytren disease and serves as a critical landmark for identifying the neurovascular bundles in the palm.
Within the digits, the fascial anatomy becomes highly intricate. Key structures include:
* Natatory Ligaments: Transverse fibers in the web spaces that contribute to digital abduction.
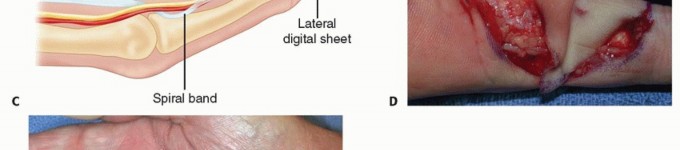
* Spiral Bands: Fibers that originate from the pretendinous band, pass deep to the neurovascular bundle, and insert onto the lateral digital sheet.
* Lateral Digital Sheets: Fascial layers running longitudinally along the lateral aspects of the digits, receiving contributions from the natatory ligaments and spiral bands.
* Grayson Ligaments: Volar to the neurovascular bundle, connecting the flexor tendon sheath to the skin.
* Cleland Ligaments: Dorsal to the neurovascular bundle, connecting the phalanges to the skin. (Like the superficial transverse palmar ligament, Cleland ligaments are classically spared in Dupuytren disease).
Pathologic Anatomy and Cord Formation
When normal bands undergo fibroproliferative changes, they become cords, which dictate the specific pattern of digital contracture.
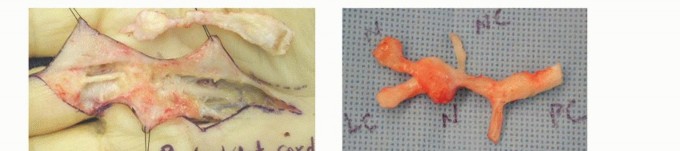
The Pretendinous Cord arises from the pretendinous band and is responsible for metacarpophalangeal (MCP) joint contractures. Because this cord lies entirely superficial to the neurovascular bundle, surgical excision at the level of the palm is generally straightforward, provided the bundle is identified proximally.
The Central Cord is a continuation of the pretendinous cord into the digit, attaching to the base of the middle phalanx or the flexor tendon sheath, leading to proximal interphalangeal (PIP) joint contracture.
The Spiral Cord is the most anatomically treacherous structure in Dupuytren surgery. It develops from the sequential contracture of four normal structures: the pretendinous band, the spiral band, the lateral digital sheet, and Grayson's ligament. As this composite cord contracts, it produces a severe PIP joint flexion contracture and simultaneously displaces the neurovascular bundle centrally, volarly, and proximally. The nerve may be found wrapped around the cord or lying directly beneath the dermis, making it highly susceptible to iatrogenic transection during the initial skin incision.
The Natatory Cord arises from the natatory ligaments, causing web space contractures and preventing digital abduction.
The Retrovascular Cord is a less common pathologic structure that runs dorsal to the neurovascular bundle and can contribute to distal interphalangeal (DIP) joint contractures or recurrent PIP joint contractures.
Indications and Contraindications
The mere presence of a palmar nodule or a non-contracting cord is not an indication for intervention. Because Dupuytren disease is characterized by an unpredictable clinical course, prophylactic surgery is contraindicated; operating too early can precipitate a flare reaction, accelerating the disease process and worsening the ultimate functional outcome.
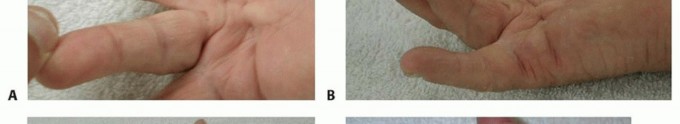
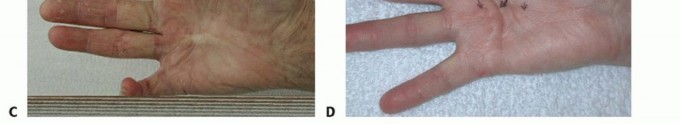
Intervention is indicated when the contracture significantly impairs hand function and activities of daily living. The clinical threshold for intervention is traditionally evaluated using Hueston's tabletop test. The patient is asked to place their hand flat on a hard surface; if the hand cannot lie flat due to digital contracture, the test is positive, and intervention may be warranted.
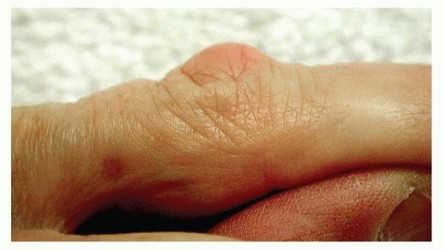
Specific angular thresholds guide surgical decision-making. Metacarpophalangeal (MCP) joint contractures are generally well-tolerated up to 30 degrees. Because the MCP joint is highly amenable to surgical release with excellent long-term outcomes, intervention is typically recommended when the contracture exceeds 30 degrees. Proximal interphalangeal (PIP) joint contractures are more problematic. The PIP joint is prone to capsular contracture and collateral ligament shortening; therefore, PIP contractures are notoriously difficult to correct fully and have a high rate of recurrence. Intervention for the PIP joint is usually considered at 15 to 20 degrees of contracture.
Operative vs Non Operative Indications Summary
| Modality | Primary Indications | Contraindications |
|---|---|---|
| Observation | Palmar nodules without contracture; painless cords; negative tabletop test. | Rapidly progressive contracture; functional impairment. |
| Percutaneous Needle Aponeurotomy (PNA) | Well-defined, palpable pretendinous cords; primarily MCP joint contractures; elderly patients with multiple comorbidities. | Diffuse disease; severe PIP joint contracture; recurrent disease; inability to palpate the cord. |
| Collagenase Injection (CCH) | Palpable cords causing MCP or PIP joint contracture in adults; patients seeking to avoid surgery. | Hypersensitivity to CCH; cords intimately involved with the neurovascular bundle; active infection. |
| Open Regional Fasciectomy | Progressive MCP contracture > 30°; PIP contracture > 15-20°; diffuse disease; failure of minimally invasive techniques. | Medically unfit for surgery; active local infection; poor soft tissue envelope (relative). |
| Dermatofasciectomy & Skin Grafting | Severe recurrent disease; Dupuytren diathesis; skin intimately involved with the cord; salvage procedures. | Inadequate donor site; severe vascular compromise of the digit. |
| Joint Arthrodesis / Amputation | End-stage fixed PIP contracture > 90°; chronic digital ischemia; multi-operated digit with neurovascular compromise. | Patient refusal; functional potential remains with soft tissue release. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is essential to mitigate complications and optimize outcomes. A detailed history must assess the rate of contracture progression, previous interventions, and the presence of diathesis factors (family history, ectopic lesions, early age of onset). Medical optimization is critical; patients with poorly controlled diabetes mellitus or those who actively smoke are at a significantly higher risk for postoperative wound complications, skin necrosis, and delayed healing.
Anesthesia and Tourniquet Considerations
Surgical intervention for Dupuytren disease can be performed under various anesthetic modalities, including general anesthesia, regional block (axillary or supraclavicular), or Wide Awake Local Anesthesia No Tourniquet (WALANT).
The WALANT technique utilizes a mixture of lidocaine and epinephrine injected directly into the surgical field. This approach offers several distinct advantages: it eliminates the systemic risks of general anesthesia, avoids the ischemic pain associated with a pneumatic tourniquet, and allows the surgeon to assess active digital motion and the adequacy of the contracture release intraoperatively. However, in cases of severe recurrent disease where the anatomy is heavily distorted, many surgeons still prefer a regional block with a proximal pneumatic tourniquet to ensure a completely bloodless field for meticulous neurovascular dissection.
Patient Positioning and Equipment
The patient is positioned supine with the operative extremity extended on a radiolucent hand table. If a tourniquet is utilized, it is placed on the proximal arm and typically inflated to 250 mm Hg after exsanguination with an Esmarch bandage.
Surgical loupe magnification (minimum 2.5x, preferably 3.5x to 4.5x) is mandatory for this procedure. The neurovascular bundles in a digit afflicted by Dupuytren disease are often encased in fibrotic tissue and aberrantly displaced; attempting this dissection without adequate magnification is a breach of standard surgical principles. Fine-tipped bipolar electrocautery, delicate skin hooks, and specialized retractors (such as the Senn or Ragnell retractors) must be available on the sterile field.
Detailed Surgical Approach and Technique
The surgical management of Dupuytren disease encompasses a spectrum of interventions, ranging from minimally invasive cord disruption to radical excision of diseased fascia and overlying skin. The choice of procedure is dictated by the severity of the contracture, the specific joints involved, the primary vs. recurrent nature of the disease, and the patient's overall physiological status.
Percutaneous Needle Aponeurotomy
Percutaneous needle aponeurotomy (PNA) is a minimally invasive technique aimed at mechanically dividing the pathologic cord. It is most efficacious for isolated, well-defined pretendinous cords causing metacarpophalangeal (MCP) joint contractures.
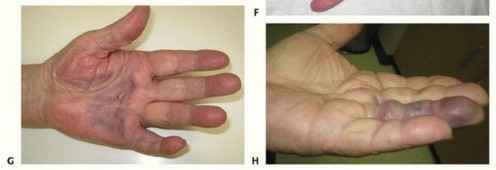
Under local anesthesia, a 25-gauge or 22-gauge needle is introduced percutaneously. The bevel of the needle is used in a sweeping motion to score and divide the tense cord at multiple levels. Concurrently, the surgeon applies passive extension force to the digit until the cord ruptures. While PNA offers rapid recovery and minimal wound morbidity, it does not remove the diseased tissue. Consequently, the recurrence rate is high, often exceeding 50% within 3 to 5 years. Extreme caution must be exercised when utilizing PNA for PIP joint contractures due to the risk of iatrogenic injury to a displaced neurovascular bundle.
Open Regional Fasciectomy
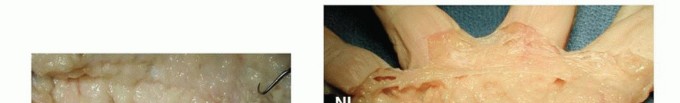
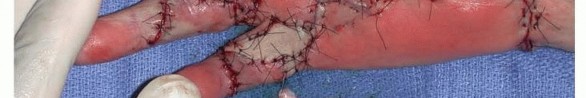
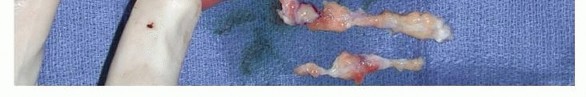
Open regional fasciectomy remains the gold standard for the surgical treatment of Dupuytren disease. The objective is the macroscopic excision of all diseased fascial tissue while preserving the neurovascular structures and normal tendinous anatomy.
Incision Design
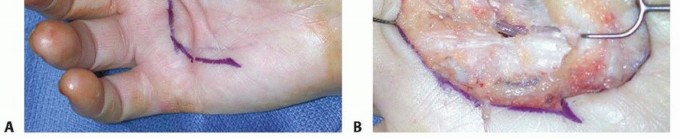
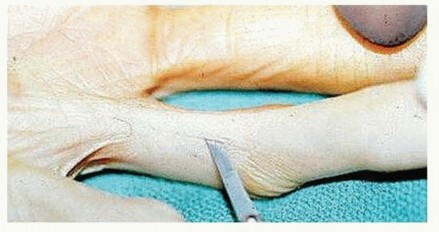
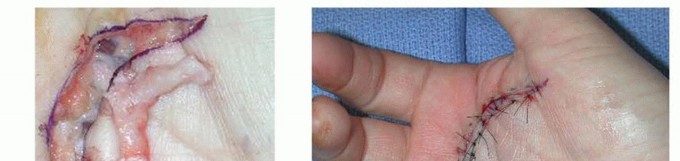
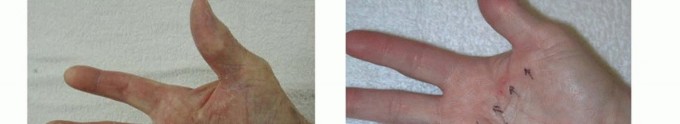
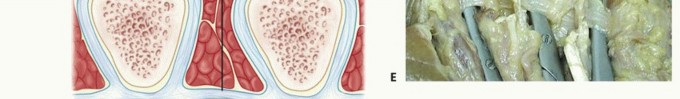
The surgical incision must provide extensile exposure of the cord while preventing postoperative linear scar contracture. The most widely utilized incisions are the Brunner zigzag incision and the longitudinal incision with subsequent Z-plasty closure.
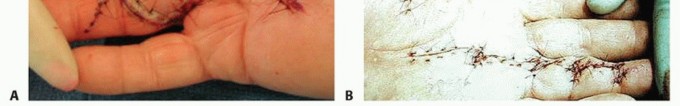
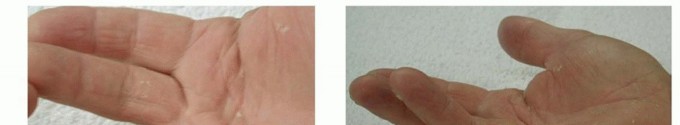
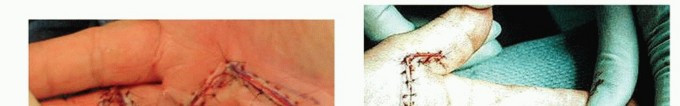
The Brunner incision consists of volar V-shaped flaps with the apices located at the flexion creases of the joints. This design ensures that the resulting scar does not cross the flexion creases perpendicularly, thereby mitigating scar contracture. Alternatively, a straight longitudinal incision can be made directly over the palpable cord. After the fasciectomy is complete, the longitudinal wound is closed utilizing multiple Z-plasties to lengthen the volar skin and break up the linear scar.
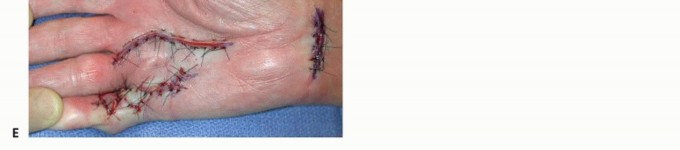
Dissection and Neurovascular Protection
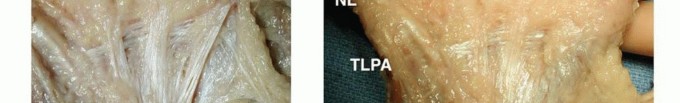
The fundamental principle of Dupuytren surgery is to identify the neurovascular bundle in an area of normal, un-diseased anatomy before tracing it into the pathologic tissue.
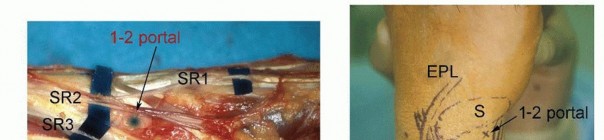
Dissection commences proximally in the palm. The pretendinous cord is identified, and the dissection proceeds deep to the cord. The superficial transverse palmar ligament is an invaluable landmark; it lies deep to the pretendinous cord and superficial to the neurovascular bundles. By locating this ligament, the surgeon can definitively identify the digital nerves and arteries in their normal anatomic position before they enter the diseased digit.
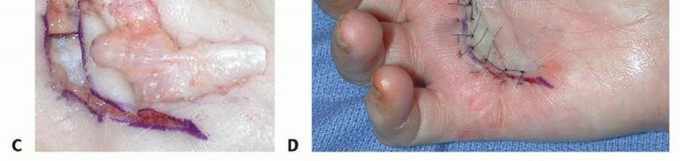
As the dissection progresses distally into the digit, the surgeon must be acutely aware of the spiral cord. If a spiral cord is present, the neurovascular bundle will be pulled centrally and volarly, often lying immediately deep to the dermis. The surgeon must meticulously dissect the fibrotic tissue off the nerve using tenotomy scissors or a scalpel under loupe magnification. The bundle must be continuously visualized and protected with vessel loops as the cord is excised.
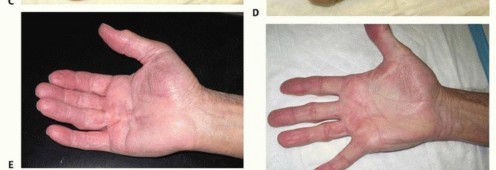
Excision and Joint Release
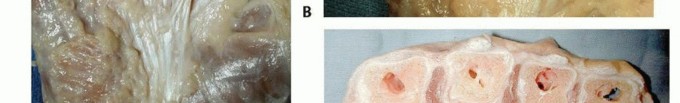
Once the neurovascular bundles are secured, the diseased fascia is systematically excised. The pretendinous cord, central cord, lateral cords, and natatory cords are resected. Care is taken to avoid injury to the underlying flexor tendon sheath.
Following fasciectomy, the degree of residual contracture is assessed. If a PIP joint contracture persists despite complete removal of the diseased fascia, the pathology likely involves secondary joint capsular contracture. Sequential release may be required, progressing from release of the flexor tendon sheath, to checkrein ligament excision, to volar plate release, and occasionally accessory collateral ligament release. However, aggressive joint release must be balanced against the risk of postoperative joint instability and profound stiffness.
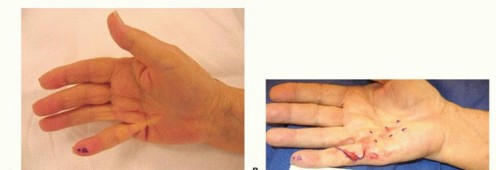
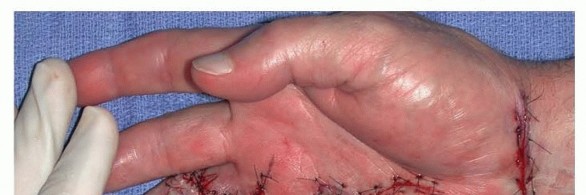
Skin Closure and the McCash Technique
After tourniquet deflation, meticulous hemostasis is achieved using bipolar electrocautery to prevent postoperative hematoma. The skin is typically closed with non-absorbable sutures.
In cases of severe, chronic contracture, the volar skin may be significantly shortened and unable to cover the extended digit without excessive tension. In such scenarios, the McCash open palm technique is highly effective. The transverse palmar incision is left entirely open to heal by secondary intention. This technique eliminates skin tension, dramatically reduces the risk of hematoma, and allows for immediate postoperative mobilization. Despite the open wound, infection rates are remarkably low, and the wound typically epithelializes within 4 to 6 weeks, leaving a supple, non-contractile scar.

You Might Also Like