Osteonecrosis and Joint Preservation: Comprehensive Surgical Strategies
Key Takeaway
Osteonecrosis of the femoral head is a progressive pathology leading to joint deterioration if untreated. This guide details the etiology, Ficat classification, and diagnostic modalities including MRI. It provides step-by-step surgical protocols for joint preservation, emphasizing core decompression for early-stage femoral head necrosis and ulnohumeral arthroplasty for advanced elbow osteoarthritis, optimizing outcomes for orthopedic surgeons and residents.
OSTEONECROSIS OF THE FEMORAL HEAD
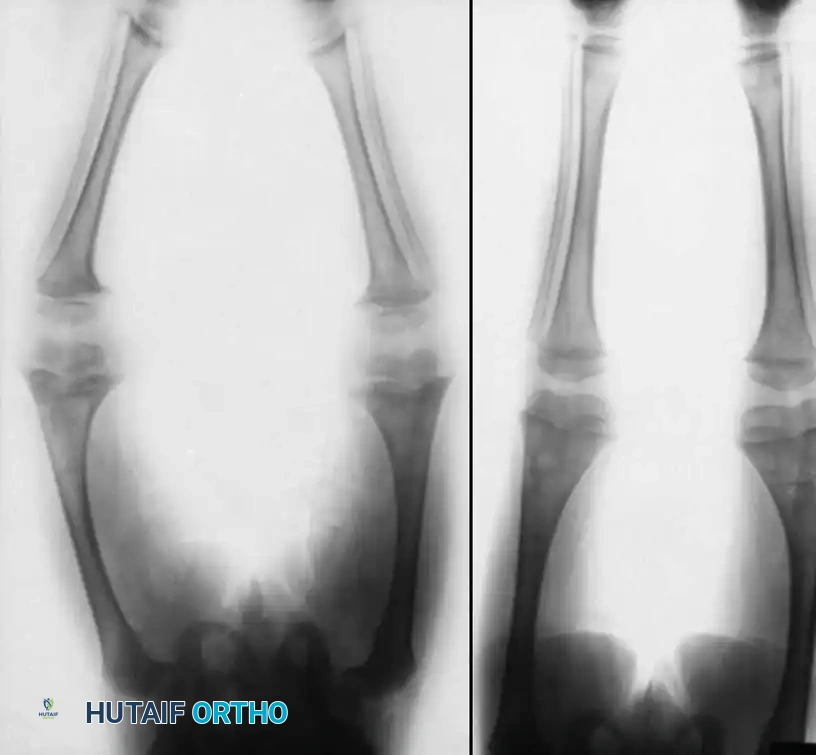
Osteonecrosis of the femoral head is a debilitating, progressive disease that predominantly affects patients in the third through fifth decades of life. If left untreated, the natural history inevitably leads to subchondral collapse, complete deterioration of the hip joint, and the premature need for total hip arthroplasty (THA). Epidemiological data estimates that 20,000 new cases of osteonecrosis are diagnosed annually in the United States, and the condition currently accounts for approximately 18% of all THAs performed nationwide.
Formerly referred to as "avascular necrosis" (AVN), the term osteonecrosis is now the preferred academic nomenclature. Simply defined, osteonecrosis translates to "dead bone." The avascular state of the necrotic bone is the terminal result of a critical loss of microcirculation, which can stem from numerous potential etiologies. Therefore, osteonecrosis should be viewed not as a single disease entity, but as an end-stage condition resulting from multiple pathogenic pathways.
Etiology and Pathogenesis
The list of risk factors and associated conditions for osteonecrosis is extensive. Recognized causes include:
* Alcohol abuse
* Systemic corticosteroid use
* Trauma (e.g., femoral neck fractures, hip dislocations)
* Hypercoagulable states (e.g., Factor V Leiden, antiphospholipid syndrome)
* Sickle cell anemia and other hemoglobinopathies
* Caisson disease (dysbarism)
* Gaucher disease
* Renal osteodystrophy
* Gout
In a significant percentage of cases, no definitive cause can be identified; these patients are classified as having idiopathic osteonecrosis.
Several theories regarding the exact pathogenesis have been proposed. Current hypotheses include direct cellular toxicity (often implicated in alcohol and steroid use), coagulopathic states leading to venous thrombosis, hyperlipidemia resulting in intraosseous fat emboli, direct vascular interruption (trauma), and elevated bone marrow pressure causing compartment syndrome of the bone. None of these theories independently accounts for the vast variety of causes. Because many patients with known risk factors never develop the disease, and others without identifiable risks do, the pathogenic process is widely accepted to be multifactorial.
Clinical Diagnosis and Imaging
Patients are typically asymptomatic during the early stages of osteonecrosis. As the ischemic cascade progresses and microfractures accumulate, patients eventually develop deep, throbbing groin pain that is exacerbated by ambulation and weight-bearing. A thorough history and physical examination are paramount to identify potential risk factors and assess the clinical status of the hip.
Plain radiography remains the initial imaging modality of choice. Standard anteroposterior (AP) and frog-leg lateral views of the pelvis and hip should be obtained. Radiographic changes are highly dependent on the stage of the disease. In early stages, plain films may appear entirely normal. As the disease progresses, subtle changes such as increased patchy sclerosis or cystic lucencies appear in the femoral head.
With further progression, the pathognomonic "crescent sign" becomes visible, representing a subchondral fracture. This is often best visualized on the frog-leg lateral view.

In the end stages of the disease, gross femoral head collapse occurs, leading to secondary degenerative changes on both the femoral and acetabular sides of the joint.
While bone scanning (technetium-99m) can be useful—especially for assessing multiple joints—its sensitivity is variable. Miki et al. reported that uptake is decreased in the very early stages but becomes variable or increased once symptoms occur.
Magnetic Resonance Imaging (MRI) has revolutionized the early diagnosis of osteonecrosis. MRI is the gold standard, allowing for the detection of the disease long before radiographic changes occur. It accurately determines the exact stage and volumetric extent of the necrotic lesion. A single-band or double-line sign on T2-weighted images is highly specific. When plain radiographs show changes in only one joint, an MRI of the entire pelvis is strictly indicated to evaluate the asymptomatic contralateral hip, as bilateral involvement is common.
Ficat and Arlet Classification
The most widely utilized classification scheme for osteonecrosis of the femoral head was developed by Ficat and Arlet, based primarily on radiographic and clinical findings:
- Stage 0: Patient is asymptomatic. Plain radiographs are normal. MRI or bone biopsy shows evidence of ischemia/necrosis.
- Stage I: Patient has mild pain. Plain radiographs remain normal. MRI and bone scans are positive.
- Stage II: Mild to moderate pain. Radiographs show density changes (sclerosis or cysts) in the femoral head.
- Stage IIA: Sclerosis or cysts with a normal joint line and normal head contour.
- Stage IIB: Flattening of the femoral head or the presence of a crescent sign (subchondral fracture) without gross collapse.
- Stage III: Moderate to severe pain. Radiographs show loss of sphericity, gross subchondral collapse, and compaction of the necrotic segment.
- Stage IV: Severe pain. Radiographs demonstrate joint space narrowing and secondary osteoarthritic changes in the acetabulum.
Clinical Pearl: The critical threshold in the natural history of osteonecrosis is subchondral collapse (transition from Stage II to Stage III). Once collapse occurs, joint-preserving procedures have a drastically reduced success rate, and progressive osteoarthritis is generally inevitable.
Surgical Management: Joint Preservation
The natural history of early-stage osteonecrosis (before subchondral collapse) suggests a high rate of progression, especially in symptomatic patients. Conservative treatment, such as restricted weight-bearing or bed rest, is universally ineffective at halting disease progression. Therefore, surgical intervention is indicated for symptomatic patients with pre-collapse disease.
Core Decompression
The theoretical advantage of core decompression is based on the principle of relieving elevated intraosseous pressure caused by venous congestion. By drilling a core tract into the necrotic lesion, the procedure decompresses the bone marrow compartment, theoretically restoring vascularity and halting the ischemic cascade.
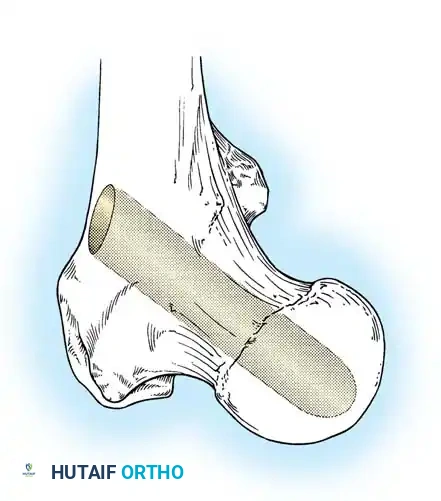
Indications: Core decompression is most effective for Ficat Stage I and Stage IIA lesions that are small to medium in size, particularly in young, non-obese patients who are not actively taking high-dose corticosteroids.
Surgical Technique:
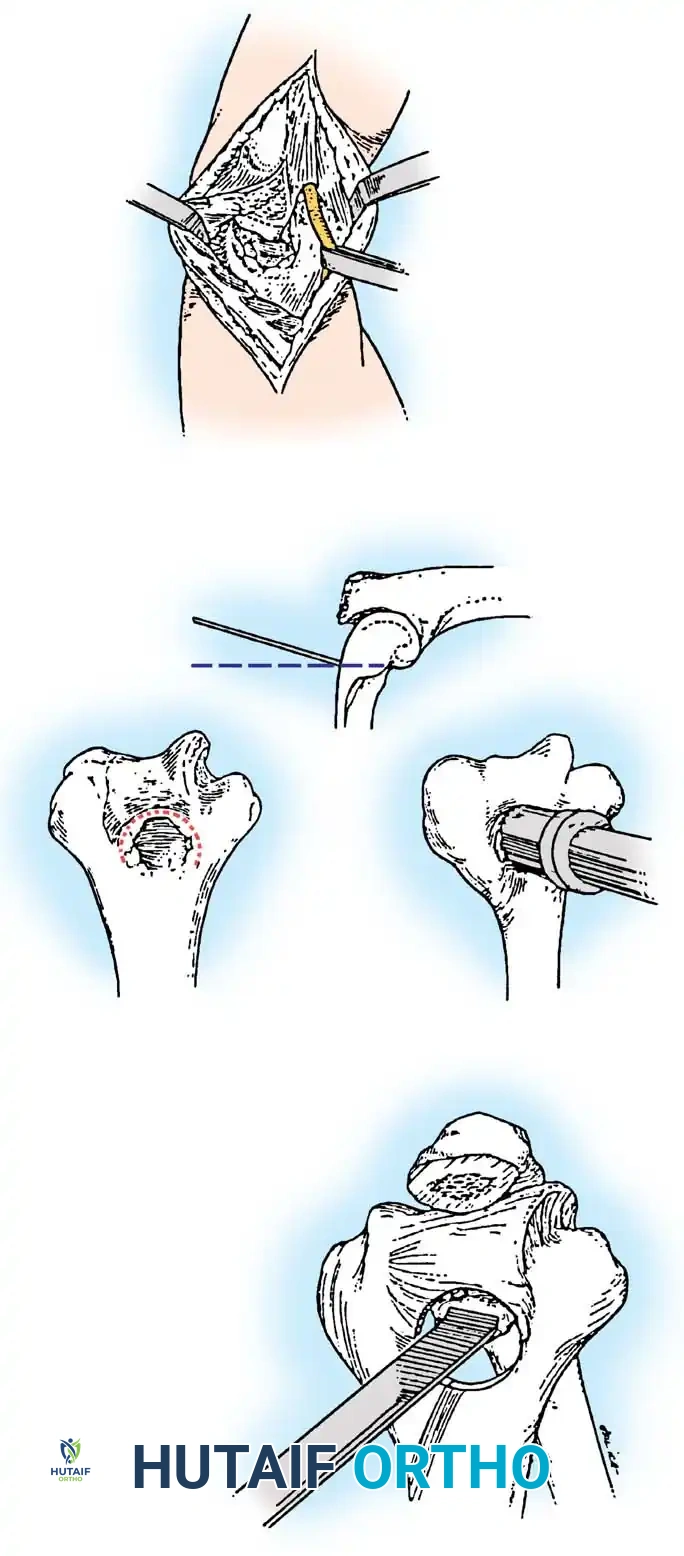
1. Positioning: The patient is placed supine on a fracture table or a radiolucent flat table.
2. Fluoroscopy: An image intensifier is positioned to allow seamless AP and lateral views of the femoral head and neck.
3. Approach: A small lateral incision is made over the proximal subtrochanteric region of the femur. The fascia lata is split, and the vastus lateralis is elevated or split to expose the lateral cortex.
4. Guide Pin Placement: Under strict fluoroscopic guidance, a 3.2-mm guide pin is introduced through the lateral cortex, distal to the vastus ridge. It is advanced through the femoral neck and directly into the center of the anterolateral necrotic lesion in the femoral head. The pin must stop 5 mm short of the subchondral plate to prevent joint penetration.
5. Reaming: A cannulated reamer (typically 8 to 10 mm) is passed over the guide pin to create the core tract. Alternatively, multiple smaller drill holes (3.2 mm) can be used to decompress the lesion while minimizing the risk of creating a stress riser that could lead to a postoperative subtrochanteric fracture.
6. Biologic Augmentation: Modern techniques often involve injecting autologous bone marrow aspirate concentrate (BMAC) or bone morphogenetic proteins (BMPs) into the cored void to stimulate osteogenesis.
Advanced Grafting and Osteotomy Techniques
For larger lesions or those approaching Stage IIB, simple core decompression may be insufficient. In these cases, structural support is required.
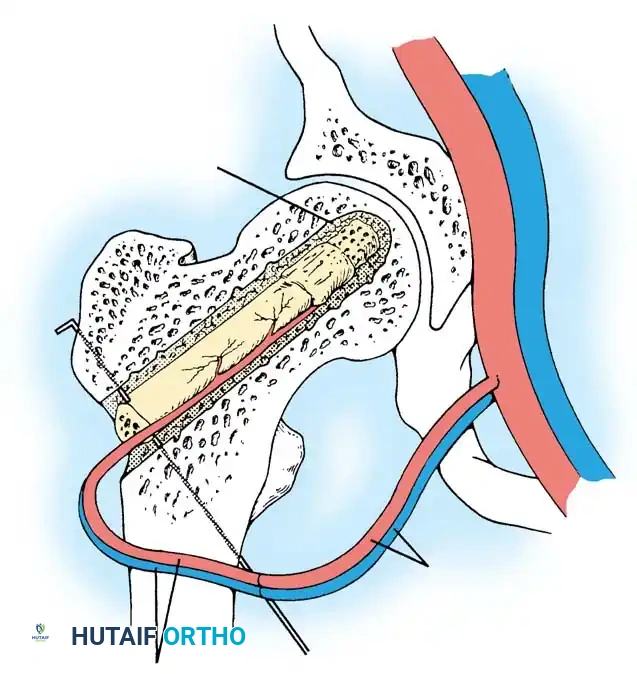
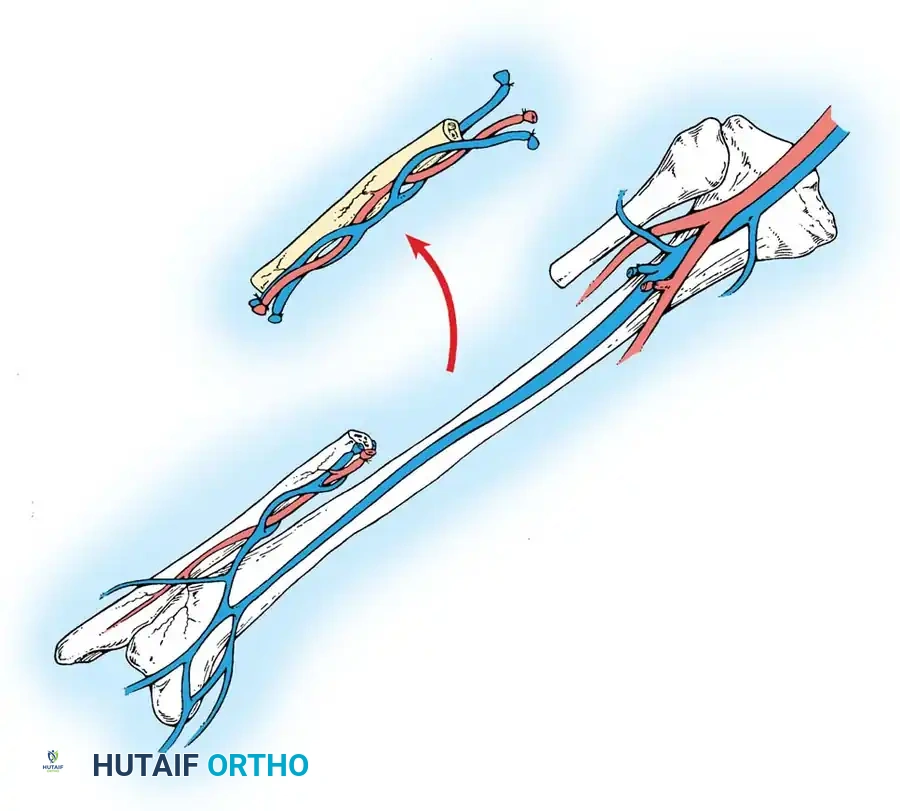
Vascularized Fibular Grafting:
This complex microsurgical procedure involves harvesting the patient's fibula along with its vascular pedicle (peroneal artery and vein). The necrotic bone in the femoral head is aggressively debrided through a core tract. The fibular graft is then inserted into the tract to provide immediate structural support to the subchondral plate, while the vascular pedicle is anastomosed to the lateral circumflex femoral vessels to restore active blood supply to the femoral head.
Rotational Osteotomies:
In highly selected young patients with small, anterosuperior lesions, a proximal femoral osteotomy (such as the Sugioka transtrochanteric rotational osteotomy) can be performed. This procedure rotates the necrotic segment away from the primary weight-bearing dome, replacing it with healthy, viable posterior articular cartilage.
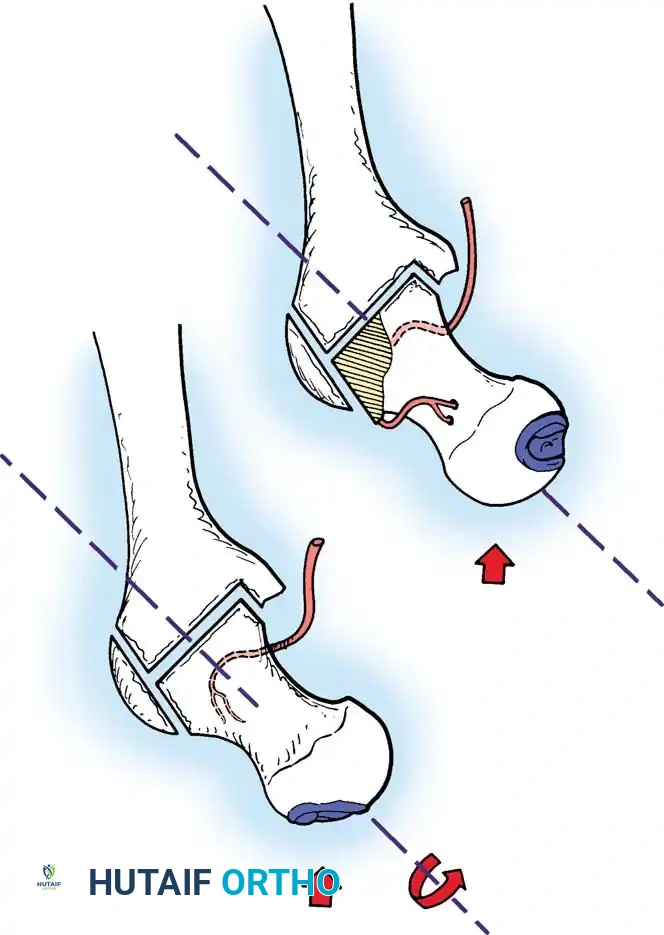
Surgical Warning: Rotational osteotomies are technically demanding and carry a high complication rate. If they fail, the altered proximal femoral anatomy can make subsequent total hip arthroplasty significantly more difficult.
ADVANCED JOINT PRESERVATION: THE ELBOW
While osteonecrosis most famously affects the femoral head, the principles of joint preservation, debridement, and decompression extend to other articulations suffering from advanced degenerative changes. In the elbow, advanced primary osteoarthritis or post-traumatic arthritis can lead to severe pain, loss of motion, and mechanical locking due to osteophyte formation and loose bodies.
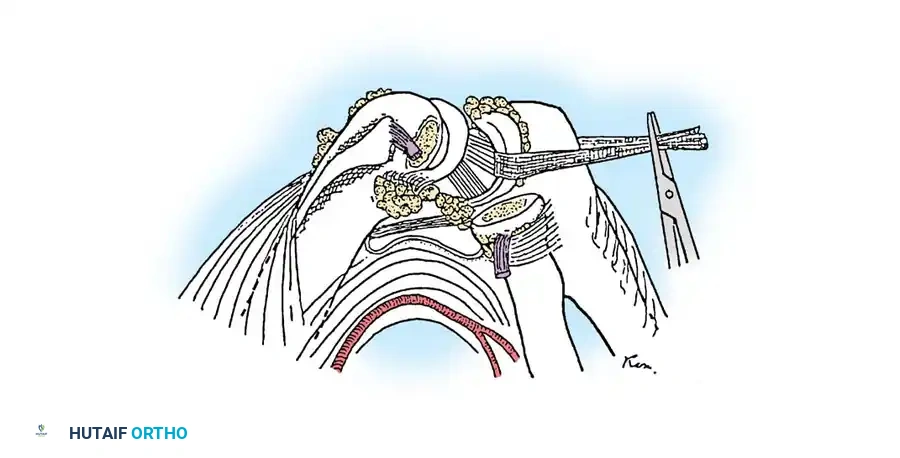
Ulnohumeral Arthroplasty (Outerbridge-Kashiwagi / Morrey Procedure)
For patients with painful limitation of motion in the elbow due to osteoarthritis, Morrey described a highly effective technique termed "ulnohumeral arthroplasty." This is a modern modification of the classic "O-K" (Outerbridge-Kashiwagi) procedure.
The hallmark of this technique is the fenestration of the olecranon fossa. By creating a communication of 1 cm or more in diameter between the anterior and posterior compartments of the elbow joint, the surgeon can effectively wash out loose bodies, debride anterior osteophytes from a posterior approach, and decompress the joint to improve the arc of motion.
Indications:
* Severe osteoarthritis with extensive ossification of the olecranon and coronoid fossae.
* Painful mechanical locking or impingement at the extremes of flexion and extension.
* Concomitant ulnar nerve involvement requiring decompression.
Contraindications:
* Excessive loss of motion (e.g., > 60 degrees of extension lost, and < 110 degrees of flexion). In such severe cases, a more extensive "column" procedure or total elbow arthroplasty may be required.
Surgical Technique: Step-by-Step
- Positioning and Approach: The patient is positioned in the lateral decubitus or prone position with the arm draped over a bolster. A posterior midline incision is utilized. The ulnar nerve is routinely identified, decompressed, and protected (or transposed if clinically indicated).
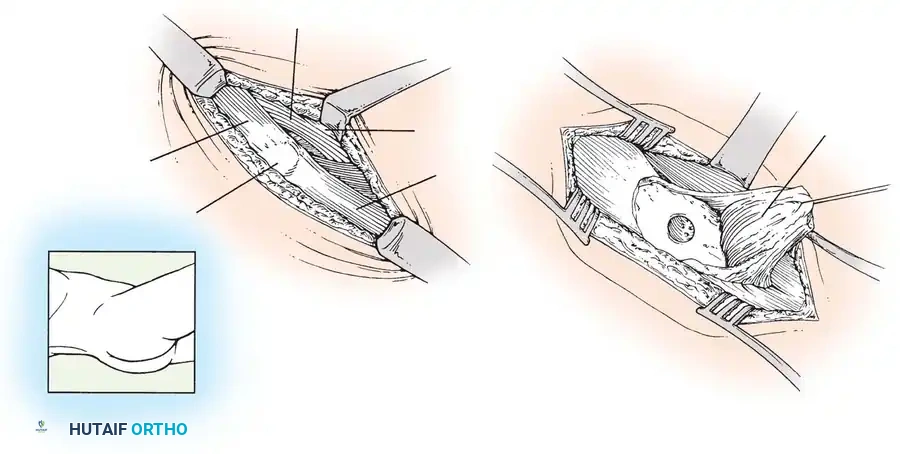
- Triceps Management: Unlike the original O-K procedure which split the triceps, Morrey’s modification involves elevating the triceps tendon from the olecranon to provide superior exposure of the posterior compartment.
-
Olecranon Debridement: The tip of the olecranon is often hypertrophied and impinges within the fossa during extension. This osteophytic tip is excised using an osteotome or oscillating saw.
-
Fenestration of the Olecranon Fossa: A trephine or high-speed burr is directed slightly proximally into the olecranon fossa to create a foramen. This fenestration must be at least 1 cm in diameter to allow adequate access to the anterior compartment.
-
Anterior Debridement: Through the newly created olecranon foramen, the anterior joint capsule can be visualized. Loose bodies are irrigated and extracted. Coronoid osteophytes, which block terminal flexion, are carefully excised using a small osteotome or pituitary rongeur passed through the defect.
-
Closure and Postoperative Protocol: The joint is thoroughly irrigated. The triceps is repaired securely to the proximal ulna through transosseous drill holes. Postoperatively, a drain is often placed to prevent hematoma. Early active and active-assisted range of motion is initiated within 24 to 48 hours to prevent capsular scarring and preserve the newly acquired arc of motion.
Clinical Outcomes
Results of the ulnohumeral arthroplasty are highly satisfactory in 80% to 90% of appropriately selected patients. Motion is usually restored to a functional range (typically 30 to 130 degrees of flexion). Long-term studies, such as those by Wada et al., demonstrate that open debridement provides lasting pain relief and maintenance of motion gains even at 10-year follow-ups, allowing a significant majority of manual laborers to return to their previous occupations.
Whether addressing the ischemic collapse of the femoral head or the osteophytic impingement of the elbow, meticulous surgical technique, strict adherence to indications, and a deep understanding of joint biomechanics are essential for successful joint preservation.
===```
📚 Medical References
- osteonecrosis, Clin Orthop Relat Res 303:38, 1994.
- Friedlaender GE: Current concepts review: bone-banking, J Bone Joint Surg 64A:307, 1982.
- Friedlaender GE: Current concepts review: bone grafts: the basic science rationale for clinical applications, J Bone Joint Surg 69A:786, 1987.
- [Friedlaender GE, Tomford W, Galloway M, et al: Tissue transplantation. In Starzl TE, Shapiro R, Simmons RL, eds: Atlas of organ transplantation, New York, 1992, Raven Press.
Froimson AI, Cummings AG Jr: Iliac hernia following](https://pubmed.ncbi.nlm.nih.gov/?term=Friedlaender%20GE%2C%20Tomford%20W%2C%20Galloway%20M%2C%20et%20al%3A%20Tissue%20transplantation.%20In%20Starzl%20TE%2C%20Shapiro%20R%2C%20Simmons%20RL%2C%20eds%3A%20Atlas%20of%20organ%20transplantation%2C%20New%20York%2C%201992%2C%20Raven%20Press.%0A%0AFroimson%20AI%2C%20Cummings%20AG%20Jr%3A%20Iliac%20hernia%20following)
You Might Also Like