Deciphering Syndrome Autosomal Recessive: Genetics Explained
Key Takeaway
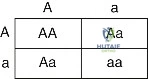
In this comprehensive guide, we discuss everything you need to know about Deciphering Syndrome Autosomal Recessive: Genetics Explained. An autosomal recessive syndrome is a genetic disorder manifested when an individual inherits two copies of a mutant recessive allele (aa). Such conditions typically involve biochemical or enzymatic defects. Parents are usually unaffected heterozygotes, with a 25% chance of transmitting the syndrome to offspring. This inheritance pattern affects males and females equally.
Introduction and Epidemiology
The intersection of molecular genetics and orthopedic surgery represents a critical frontier in the understanding and management of musculoskeletal pathology. Autosomal recessive syndromes, skeletal dysplasias, rheumatologic conditions, and connective tissue disorders frequently present with profound orthopedic manifestations that necessitate complex surgical intervention. A rigorous comprehension of cellular biology, chromosomal architecture, and molecular genetics is paramount for the academic orthopedic surgeon. This foundational knowledge not only facilitates accurate diagnosis but also dictates preoperative optimization, surgical technique, and postoperative rehabilitation in syndromic patients.
Autosomal recessive inheritance requires the presence of two mutated alleles for the phenotypic expression of the disorder. In the realm of orthopedics, autosomal recessive conditions encompass a wide spectrum of pathologies, including diastrophic dysplasia, spinal muscular atrophy, sickle cell disease, and various mucopolysaccharidoses (such as Hurler and Morquio syndromes). The epidemiology of these conditions varies significantly based on geographic and ethnic demographics, often demonstrating higher prevalence in populations with higher rates of consanguinity. Understanding the underlying molecular mechanisms—ranging from DNA transcription errors to aberrant cell cycle regulation—provides the basis for targeted biologic therapies, advanced genetic screening, and tailored surgical management.
Molecular Anatomy and Cellular Biomechanics
To comprehend the pathophysiology of autosomal recessive syndromes and associated connective tissue disorders, one must first master the molecular anatomy of the human cell. The biomechanics of cellular replication and protein synthesis dictate tissue integrity, bone healing, and immune response.
Chromosomal Architecture and Deoxyribonucleic Acid
The human genome is organized into 46 chromosomes, comprising 23 distinct pairs. This includes 22 pairs of autosomes and one pair of sex chromosomes (XX or XY). The structural integrity of these chromosomes is maintained by the intricate coiling of deoxyribonucleic acid (DNA) around histone protein octamers, forming nucleosomes. These nucleosomes further condense into chromatin, allowing meters of genetic material to be packaged within the microscopic confines of the cellular nucleus.
DNA itself possesses a highly stable double-helix structure. It is constructed from a sugar-phosphate backbone linking specific nucleotide bases. The purines (adenine and guanine) strictly pair with the pyrimidines (thymine and cytosine, respectively). Specifically, adenine links to thymine via two hydrogen bonds, while guanine links to cytosine via three hydrogen bonds, conferring greater thermal stability to GC-rich regions of the genome.

Transcription Mechanisms and Rheumatologic Biomarkers
A gene is fundamentally defined as a specific sequence of DNA that encodes the information requisite for the synthesis of a single protein. The conversion of this genomic blueprint into a functional protein begins with transcription—the process of synthesizing messenger RNA (mRNA) from the DNA template.
Transcription initiates with the unwinding of the DNA double helix, a mechanically demanding process mediated by the enzyme DNA topoisomerase. Topoisomerase relieves the torsional strain and supercoiling ahead of the replication or transcription fork by creating transient single-strand breaks. In the context of rheumatologic pathology, Topoisomerase-1 is a critical autoantigen. Antibodies directed against Topoisomerase-1, clinically identified as scl-70 antibodies, are highly specific markers for systemic sclerosis (scleroderma) and the diffuse cutaneous variant of CREST syndrome (calcinosis, Raynaud phenomenon, sclerodactyly, telangiectasia).
Once the DNA is unwound, RNA polymerase traverses the template strand, synthesizing a complementary mRNA molecule. RNA differs structurally from DNA; it is typically single-stranded, utilizes a ribose sugar rather than deoxyribose, and substitutes the pyrimidine uracil for thymine. Consequently, during transcription, uracil in the nascent RNA strand pairs with adenine on the DNA template.

Posttranscriptional Modification and Spliceosome Pathology
The initial mRNA transcript, known as pre-mRNA, undergoes rigorous posttranscriptional modification before it can be translated. The human genome is highly complex; approximately 97 percent consists of noncoding DNA. Within a given gene, the coding sequences are termed exons (which are expressed), while the intervening noncoding sequences are termed introns.
Splicing is the critical cellular process whereby introns are excised and exons are ligated together to form the mature mRNA transcript. This highly regulated process is catalyzed by the spliceosome, a macromolecular complex composed of small nuclear ribonucleoproteins (snRNPs). These snRNPs are RNA-protein complexes that recognize specific splice site sequences.
Pathologies of the immune system frequently target these splicing mechanisms. Autoantibodies directed against specific snRNP components serve as pathognomonic markers for severe autoimmune connective tissue diseases. For instance, anti-Smith antibodies target core proteins of the snRNP complex and are highly specific for systemic lupus erythematosus (SLE). Similarly, anti-U1-RNP antibodies, which target the U1 specific ribonucleoprotein, are the defining serological hallmark of mixed connective tissue disease (MCTD).

Translation and Ribosomal Function
Translation is the process by which the mature mRNA transcript is utilized as a template to build a polypeptide chain. This occurs in the cytoplasm, specifically at the ribosomes. The genetic code is read in triplets known as codons, with each three-nucleotide codon corresponding to a single, specific amino acid. A sequential string of these codons dictates the primary structure of the resulting protein.
Transfer RNA (tRNA) molecules are responsible for delivering the correct amino acids to the ribosome. Each tRNA possesses an anticodon that is complementary to the mRNA codon, ensuring high-fidelity protein synthesis. The attachment of the appropriate amino acid to its corresponding tRNA is catalyzed by a family of enzymes known as aminoacyl-tRNA synthetases.
In the clinical setting of inflammatory myopathies, autoantibodies can develop against these critical translational enzymes. Antibodies directed against histidyl-tRNA synthetase, known clinically as anti-Jo-1 antibodies, are highly associated with polymyositis and dermatomyositis, particularly in patients presenting with the antisynthetase syndrome characterized by interstitial lung disease, myositis, and polyarthritis.

Cell Cycle Regulation and Tumor Suppressor Genetics
Cellular proliferation is governed by the cell cycle, a highly regulated sequence of events culminating in DNA duplication and cell division. Ploidy refers to the number of sets of chromosomes within a cell, typically annotated as XN. The standard somatic cell is diploid (2N).
The cell cycle is divided into distinct phases:
1. Growth 0 (G0): A quiescent, stable phase where cells maintain diploid (2N) DNA content without active division.
2. Growth 1 (G1): Upon receiving mitogenic stimuli, cells initiate growth and organelle duplication but remain diploid (2N).
3. Synthesis (S): The critical period of DNA replication, resulting in a tetraploid (4N) DNA content.
4. Growth 2 (G2): A secondary phase of cellular growth and protein synthesis, maintaining tetraploidy (4N) in preparation for division.
5. Mitosis (M): The complex sequence of chromosomal segregation and cytokinesis resulting in two identical, diploid (2N) daughter cells.
During mitosis, the precise separation of genetic material relies on the attachment of spindle fibers to centromeres, the specialized DNA sequences that link sister chromatids. Autoantibodies targeting centromeric proteins (anticentromere antibodies) are a primary diagnostic marker for limited cutaneous systemic sclerosis, historically referred to as CREST syndrome.
Progression through these cell cycle phases is strictly gated by regulatory proteins, most notably cyclins, cyclin-dependent kinases (CDKs), and tumor suppressor genes. Genetic defects or somatic alterations in these tumor suppressors predispose cells to dysregulated, unchecked growth, leading to oncogenesis. A classic example is the retinoblastoma protein (pRb-1). pRb undergoes progressive, cell cycle-regulated phosphorylation. In its hypophosphorylated state, pRb binds and sequesters the E2F transcription factor, arresting the cell in the G1 phase. Upon phosphorylation by CDK complexes, pRb releases E2F, allowing the cell to transition into the S phase. Mutations inactivating both alleles of the RB1 gene lead to familial retinoblastoma and confer a significantly elevated risk of secondary primary malignancies, most notably osteosarcoma.

Indications and Contraindications
Surgical intervention in patients with autosomal recessive syndromes and associated genetic connective tissue disorders requires meticulous patient selection. The indications for surgery often differ substantially from those in the general population due to altered bone biomechanics, compromised soft tissue healing, and systemic comorbidities.
Genetic profiling and molecular diagnostics are indicated for any pediatric or young adult patient presenting with atypical fractures, disproportionate short stature, multiple joint dislocations, or progressive spinal deformity. Identifying the specific molecular defect (e.g., SMN1 mutation in spinal muscular atrophy, or specific enzyme deficiencies in mucopolysaccharidoses) directly influences surgical timing and implant selection.
Contraindications to elective orthopedic surgery in these populations often involve severe, unoptimized cardiopulmonary pathology. For instance, restrictive lung disease in thoracic insufficiency syndrome or severe cardiomyopathy in Friedreich's ataxia may present absolute contraindications to extensive reconstructive procedures until medical optimization is achieved.
| Intervention Category | Operative Indications | Non Operative Indications | Absolute Contraindications |
|---|---|---|---|
| Spinal Deformity | Progressive scoliosis > 50 degrees; Coronal/sagittal imbalance; Neurogenic claudication | Curve < 40 degrees; Skeletally immature without progression; High surgical risk | Unoptimized cor pulmonale; Active surgical site infection; Severe malnutrition |
| Joint Reconstruction | End-stage secondary osteoarthritis; Intractable pain failing conservative management | Mild to moderate joint space narrowing; Asymptomatic deformity | Charcot arthropathy (relative); Active systemic bacteremia |
| Fracture Management | Displaced intra-articular fractures; Impending pathologic fractures; Long bone nonunions | Nondisplaced fractures amenable to casting; Terminal illness | Inability to tolerate anesthesia (malignant hyperthermia risk unmitigated) |
| Soft Tissue Contractures | Fixed contractures severely limiting ADLs or ambulation | Flexible deformities responsive to serial casting or orthotics | Severe overlying skin compromise (e.g., severe scleroderma skin changes) |

Pre Operative Planning and Patient Positioning
Preoperative planning for the syndromic orthopedic patient is highly complex and mandates a multidisciplinary approach involving orthopedics, medical genetics, pulmonology, cardiology, and specialized anesthesiology.
Advanced imaging is non-negotiable. In many autosomal recessive skeletal dysplasias (such as Morquio syndrome), odontoid hypoplasia and atlantoaxial instability are prevalent. Flexion-extension cervical spine radiographs and magnetic resonance imaging (MRI) of the craniocervical junction are mandatory prior to any procedure requiring endotracheal intubation. Failure to recognize cervical instability can result in catastrophic spinal cord injury during neck extension for intubation.
Bone quality assessment is another critical component of preoperative planning. Conditions characterized by altered collagen synthesis or osteoclast dysfunction (such as autosomal recessive malignant osteopetrosis) present extreme challenges. In osteopetrosis, the bone is profoundly dense yet brittle, lacking a medullary canal. Preoperative planning must include the availability of specialized high-speed diamond burrs, as standard orthopedic drills will rapidly overheat and fail. Conversely, in conditions with severe osteopenia, planning must involve strategies for augmented fixation, such as cement-augmented pedicle screws or the use of locking plate constructs with multiple points of fixation to prevent implant pullout.
Patient positioning requires extraordinary care. Patients with connective tissue disorders, such as scleroderma or severe joint contractures typical of diastrophic dysplasia, are at high risk for positioning-related nerve palsies and pressure necrosis. The use of specialized gel pads, meticulous padding of all bony prominences, and avoidance of extreme joint positioning during the procedure are imperative. In patients with severe kyphoscoliosis, standard prone positioning frames (e.g., Jackson table) may need custom modifications to accommodate the rigid thoracic deformity without compressing the abdomen or compromising ventilation.

Detailed Surgical Approach and Technique
The surgical approach in patients with underlying genetic and molecular defects must respect the altered anatomy and biology of the tissues. Whether addressing a spinal deformity in spinal muscular atrophy or performing an osteotomy in a patient with a skeletal dysplasia, the principles of meticulous soft tissue handling and rigid fixation apply, albeit with significant modifications.
Soft Tissue Dissection and Internervous Planes
Surgical exposure must utilize standard internervous planes to minimize denervation, but the surgeon must anticipate abnormal fascial planes and muscle hypoplasia. In patients with underlying inflammatory myopathies (such as dermatomyositis associated with anti-Jo-1 antibodies), the muscle tissue may be fibrotic, friable, and prone to excessive bleeding. Electrocautery should be used judiciously to maintain hemostasis without causing excessive thermal necrosis to already compromised muscular beds.
In patients with scleroderma (associated with scl-70 or anticentromere antibodies), the skin and subcutaneous tissues are thickened, inelastic, and poorly vascularized. Incisions should be planned to avoid areas of severe skin tethering if possible. Undermining of the skin edges must be strictly avoided to prevent postoperative wound necrosis and dehiscence.
Osseous Preparation and Reduction
The technique for osseous preparation is dictated by the specific molecular defect. In conditions leading to hyperdense bone (e.g., osteopetrosis), the absence of a medullary canal makes standard intramedullary nailing nearly impossible. If intramedullary fixation is absolutely required, the canal must be meticulously created using sequential high-speed burring under continuous cold saline irrigation to prevent thermal osteonecrosis.
In conditions characterized by osteomalacia or severe osteopenia, reduction maneuvers must be gentle to prevent iatrogenic fractures. The use of reduction forceps with broad footprints is preferred over pointed reduction clamps, which can easily crush dysplastic bone.
Fixation Strategies
Implant selection is critical. In the pediatric syndromic patient, growth-friendly constructs are often required. For spinal deformity in early-onset scoliosis associated with autosomal recessive conditions, magnetically controlled growing rods (MCGR) or vertical expandable prosthetic titanium ribs (VEPTR) allow for continued spinal and thoracic growth while controlling the deformity.
When performing internal fixation for fractures or osteotomies in mechanically inferior bone, the principle of load-sharing versus load-bearing must be carefully evaluated. Locking plate technology, which acts as an internal fixator, is highly advantageous in poor-quality bone as it does not rely on friction between the plate and bone for stability. Furthermore, maximizing the working length of the construct and utilizing bicortical fixation can help dissipate stress and reduce the risk of peri-implant fractures.

Complications and Management
The complication profile for orthopedic procedures in patients with autosomal recessive syndromes and systemic genetic disorders is significantly higher than in the general population. The molecular defects that define these syndromes often impair the normal physiologic responses to surgical trauma, leading to delayed healing, increased infection rates, and hardware failure.
A deep understanding of the patient's specific genetic profile allows the surgical team to anticipate and mitigate these risks. For example, patients with certain muscular dystrophies or myopathies are at a known risk for malignant hyperthermia or anesthesia-induced rhabdomyolysis. Preoperative coordination with anesthesiology to employ non-triggering agents (avoiding volatile anesthetics and succinylcholine) is a life-saving necessity.
| Complication | Estimated Incidence in Syndromic Cohorts | Pathophysiologic Mechanism | Salvage Strategy and Management |
|---|---|---|---|
| Hardware Pullout / Failure | 15 - 30% | Altered bone mineral density; Defective collagen cross-linking | Revision with augmented fixation (cement, larger diameter screws, locking technology); Extended immobilization |
| Pseudoarthrosis / Nonunion | 10 - 25% | Impaired osteoblast/osteoclast coupling; Poor vascularity | Revision surgery with autologous bone grafting; Application of orthobiologics (BMP-2); Optimization of metabolic parameters |
| Wound Dehiscence / Infection | 5 - 15% | Compromised microcirculation (e.g., scleroderma); Immunosuppression | Aggressive surgical debridement; Negative pressure wound therapy; Soft tissue flap coverage by plastic surgery |
| Cervical Spine Instability / Myelopathy | Variable (Syndrome specific) | Odontoid hypoplasia; Ligamentous laxity | Urgent closed reduction and halo immobilization; Posterior occipitocervical fusion |
| Malignant Hyperthermia | Rare but catastrophic | Ryanodine receptor mutations; Altered calcium channel dynamics | Immediate cessation of triggering agents; Administration of Dantrolene; Active cooling; ICU level care |
Post Operative Rehabilitation Protocols
Postoperative rehabilitation in the syndromic patient cannot follow standard, protocolized timelines. The rehabilitation strategy must be highly individualized, taking into account the patient's baseline functional status, the specific molecular pathology affecting tissue healing, and the biomechanical stability of the surgical construct.
Weight-bearing restrictions are frequently extended in patients with connective tissue disorders or skeletal dysplasias due to anticipated delays in osseous union. Radiographic evidence of bridging callus may take significantly longer to appear compared to neurotypically normal, non-syndromic patients.
Orthotic management plays a substantial role in the postoperative phase. Custom-molded orthoses (such as Thoracolumbosacral orthoses [TLSO] or Ankle-Foot Orthoses [AFO]) are often required to protect surgical repairs and prevent the recurrence of deformity. Because patients with conditions like scleroderma or epidermolysis bullosa have highly vulnerable skin, the fabrication of these orthoses requires specialized, pressure-relieving interfaces to prevent catastrophic ulceration.
Physical therapy focuses on the maintenance of joint mobility and the prevention of contractures, which are a constant threat in many autosomal recessive conditions. However, passive stretching must be performed with caution to avoid iatrogenic physeal injuries or fractures in osteopenic bone. A multidisciplinary approach, incorporating occupational therapy, physical therapy, physiatry, and orthopedic surgery, is essential to maximize functional recovery and integrate the patient back into their activities of daily living.
Summary of Key Literature and Guidelines
The integration of molecular genetics into orthopedic surgery is supported by a robust and expanding body of literature. Foundational texts, such as Medical Genetics by Jorde et al., provide the essential framework for understanding transcription, translation, and chromosomal architecture as it applies to human disease.
In the orthopedic literature, guidelines established by the American Academy of Orthopaedic Surgeons (AAOS) and the Pediatric Orthopaedic Society of North America (POSNA) strongly advocate for multidisciplinary management of skeletal dysplasias. Key literature emphasizes the necessity of preoperative cervical spine screening in all patients with mucopolysaccharidoses and the critical role of targeted genetic testing in patients presenting with atypical primary bone tumors, such as osteosarcoma, to rule out underlying pRb-1 or p53 (Li-Fraumeni syndrome) mutations.
Recent advancements in CRISPR-Cas9 gene editing and targeted biologics (such as nusinersen for spinal muscular atrophy) are rapidly altering the natural history of these previously devastating autosomal recessive conditions. As these medical therapies improve survival and motor function, the orthopedic surgeon will increasingly be called upon to manage the complex, evolving musculoskeletal manifestations of these genetic syndromes, underscoring the absolute necessity of mastering the underlying cellular and molecular biology.
Clinical & Radiographic Imaging








