Decoding Von Willebrand Factor: Fundamental Basic Science Principles

Key Takeaway
This topic focuses on Decoding Von Willebrand Factor: Fundamental Basic Science Principles, Osteoinduction recruits immature pluripotent cells, stimulating their differentiation into pre-osteoblasts. Key growth factors, such as bone morphogenetic proteins, drive this process. Factors from sources like vascular endothelium, which can include those associated with platelet function such as **von willebrand factor**, are crucial for the comprehensive regeneration needed for healthy bone formation.
Comprehensive Introduction and Patho-Epidemiology
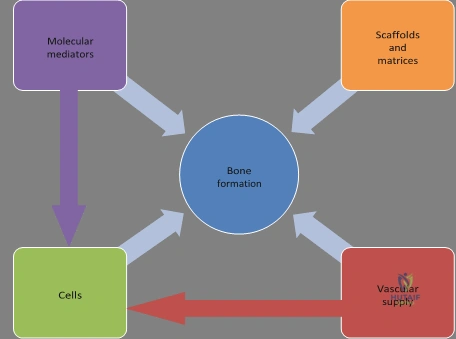
Bone regeneration is a uniquely sophisticated physiological process, distinguished from the healing of most other biological tissues by its ability to restore the structural and functional integrity of the skeleton without the formation of a fibrous scar. The fundamental basic science principles of this regenerative capacity rely on a delicate, highly orchestrated interplay of cellular and molecular events. At the core of this process is the "diamond concept" of fracture healing, which dictates that successful bone regeneration unequivocally depends on an osteoinductive stimulus, an osteoconductive matrix, a robust source of responding osteogenic cells, and a sufficient vascular supply. If one or more of these critical factors is lacking, the cascade is disrupted, and new bone formation is significantly decreased, inevitably leading to delayed union or recalcitrant nonunion.

While much of orthopedic literature focuses heavily on the structural and cellular components of the bone extracellular matrix, the paramount importance of the vascular endothelium and its associated glycoproteins cannot be overstated. Von Willebrand Factor (VWF), a massive multimeric glycoprotein synthesized primarily by vascular endothelial cells and megakaryocytes, serves as the critical initiator of the fracture healing cascade. Upon skeletal trauma and subsequent disruption of the intraosseous and periosteal microvasculature, subendothelial collagen is exposed. VWF immediately binds to this exposed collagen and facilitates the tethering and activation of circulating platelets via their glycoprotein Ib-IX-V complexes. This primary hemostatic event forms the foundational fracture hematoma, which is not merely a blood clot, but a dynamic, bioactive provisional matrix.
Following VWF-mediated aggregation, platelets degranulate, releasing a massive payload of alpha-granule contents into the local microenvironment. This payload includes Platelet-Derived Growth Factor (PDGF), Transforming Growth Factor-beta (TGF-β), and Vascular Endothelial Growth Factor (VEGF). These inflammatory cytokines and growth factors are the primary drivers of the early inflammatory phase of bone healing. They function as potent chemotactic agents, recruiting circulating mesenchymal stem cells (MSCs), polymorphonuclear leukocytes, and macrophages to the injury site. Furthermore, damage to the bone extracellular matrix (ECM) liberates matrix-bound growth factors, which synergistically induce the differentiation and proliferation of osteoprogenitor cells. Thus, the VWF-initiated hematoma is the absolute prerequisite for subsequent osteoinduction and osteoconduction.
The patho-epidemiology of impaired bone healing is deeply intertwined with disruptions to this VWF-vascular axis and the cellular microenvironment. Patient-specific risk factors such as chronic tobacco abuse, poorly controlled diabetes mellitus, and prolonged use of non-steroidal anti-inflammatory drugs (NSAIDs) profoundly impair microvascular perfusion and endothelial function. Smoking, for instance, causes profound peripheral vasoconstriction and cellular hypoxia, diminishing the synthesis of VEGF and impairing the function of the vascular endothelium. This leads to a compromised fracture hematoma, inadequate delivery of pluripotent cells, and a hostile, hypoxic environment that prevents the terminal differentiation of pre-osteoblasts. Understanding these molecular and epidemiological principles is essential for the orthopedic surgeon when evaluating a patient's intrinsic biological capacity for skeletal repair.
Detailed Cellular Anatomy and Biomechanics
The cellular anatomy of the bone microenvironment is a complex ecosystem comprising osteoblasts, osteocytes, osteoclasts, and the critical vascular endothelium. Osteoinduction is fundamentally characterized by the process of recruitment of immature, pluripotent mesenchymal stem cells (MSCs) to the site of injury or surgical intervention. Under the influence of specific molecular stimuli, these MSCs undergo subsequent stimulation that causes their terminal differentiation into pre-osteoblasts and eventually mature osteoblasts. Once differentiated, these osteoblasts are responsible for synthesizing the organic components of the bone extracellular matrix, primarily Type I collagen, and regulating the deposition of inorganic hydroxyapatite crystals.
The extracellular matrix (ECM) itself is not merely a static structural support; it is a highly active biological reservoir. Hydroxyapatite, the major inorganic component of the bone ECM synthesized by osteocytes and osteoblasts, contributes fundamentally to the density and compressive strength of bone. Beyond its mechanical role, hydroxyapatite is bioactive, playing a role in the initiation of osteoblastic differentiation from local MSC populations. When bone is fractured or surgically manipulated, the degradation of the ECM releases stored growth factors, including those from the TGF-β superfamily, directly into the local milieu. This creates a powerful autocrine and paracrine feedback loop that sustains the osteoinductive drive necessary for robust callus formation and eventual remodeling.
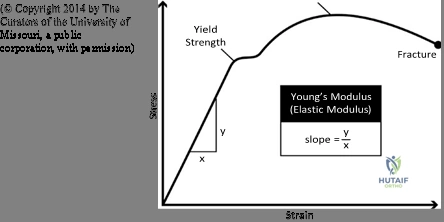
Biomechanically, the local mechanical environment will influence bone growth in strict accordance with Wolff’s Law. This foundational principle dictates that bone models and remodels in direct response to the mechanical stresses placed upon it. At the cellular level, this mechanotransduction is mediated primarily by osteocytes, which are terminally differentiated osteoblasts embedded within the mineralized matrix. Osteocytes possess an extensive dendritic network residing within the canaliculi of the bone. Mechanical loading generates fluid shear stress within these canaliculi, which is sensed by the osteocyte cilia. This mechanical signal is transduced into biochemical responses, modulating the release of sclerostin and regulating the RANKL/Osteoprotegerin (OPG) axis, thereby dictating whether bone is deposited by osteoblasts or resorbed by osteoclasts.
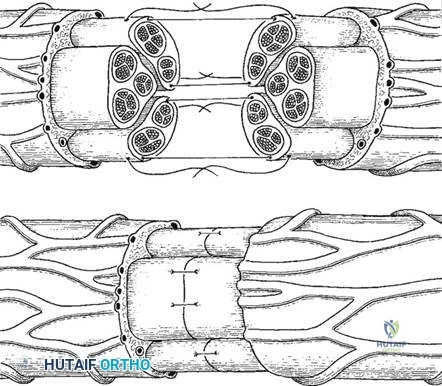
The coupling of these biomechanical forces with the cellular anatomy determines the pathway of bone regeneration. Under conditions of absolute mechanical stability—typically achieved through rigid internal fixation with interfragmentary compression—primary (haversian) bone healing occurs without the formation of an intermediate cartilaginous callus. Osteoclasts form cutting cones that traverse the fracture site, immediately followed by osteoblasts laying down lamellar bone. Conversely, under conditions of relative stability (e.g., intramedullary nailing or bridge plating), secondary bone healing occurs via endochondral ossification. Here, the VWF-mediated hematoma transitions into a soft cartilaginous callus, which is subsequently mineralized into a hard woven bone callus, and finally remodeled into organized lamellar bone according to the stress lines dictated by Wolff's Law.
Exhaustive Indications and Contraindications
The decision to augment bone healing using biologic adjuvants relies on a thorough understanding of when the intrinsic physiological cascade is insufficient. Biologic interventions are primarily designed to provide either an osteoinductive stimulus, an osteoconductive scaffold, or both. Osteoconduction is the process of bone growth on a surface or scaffold; the initial deposit of osteoid and its conversion to woven and lamellar bone is a classic example of this process. Implantation of structural bone grafts, cancellous allografts, and synthetic ceramic scaffolds are primary examples of osteoconduction utilized in clinical practice. These are indicated when there is a significant structural void or cavitary defect that requires a physical matrix for the host's native cells to migrate across and populate.

Osteoinduction relies heavily on the delivery of exogenous growth factors, the most widely studied of which are those in the bone morphogenetic protein (BMP) family. Over 20 BMPs have been identified, all functioning as members of the TGF-β superfamily of growth hormones. BMP-2 and BMP-7 have undergone the most rigorous development for therapeutic applications. BMP supplementation is currently FDA-approved for specific, highly regulated indications: to augment spinal arthrodesis (specifically anterior lumbar interbody fusion with specific cages), and to treat recalcitrant nonunions or acute open fractures of long bones (specifically tibial shaft fractures treated with intramedullary nailing). Absorbable collagen sponges have been commercialized as a highly effective method of local delivery of BMP to surgical sites, acting to retain the water-soluble protein at the target location while providing a transient osteoconductive framework.

While the indications for biologic augmentation are expanding, the contraindications are equally critical and must be strictly respected to prevent catastrophic complications. The use of powerful osteoinductive agents like rhBMP-2 is absolutely contraindicated in the setting of active surgical site infection, as the hypervascularity and cellular proliferation induced by the protein can exacerbate the infectious process. Furthermore, because BMPs are potent mitogens that stimulate cellular differentiation and proliferation, their use is strictly contraindicated in the vicinity of a resected or active malignancy due to the theoretical risk of inducing tumor growth or metastasis.

Additionally, the use of recombinant growth factors is contraindicated in skeletally immature patients, as the powerful osteoinductive stimulus can cause premature physeal closure and subsequent catastrophic limb length discrepancies or angular deformities. Hypersensitivity to the active protein or the bovine Type I collagen sponge carrier also precludes their use. Understanding the precise molecular actions of these agents—such as how Osteoprotegerin (a TNF-α superfamily member) blocks RANK ligand interaction with the RANK receptor to halt osteoclastic differentiation—allows the surgeon to manipulate the local environment safely and effectively.
Indications and Contraindications for Biologic Augmentation
| Biologic Agent / Strategy | Primary Mechanism | Indications for Use | Absolute Contraindications |
|---|---|---|---|
| Autologous Bone Graft (ICBG) | Osteogenic, Osteoinductive, Osteoconductive | Gold standard for nonunions, arthrodesis, segmental defects | Active infection, inadequate donor site stock, severe medical comorbidities |
| Cancellous Allograft | Osteoconductive | Cavitary defect filling, structural support, impaction grafting | Active local infection, severe host immunocompromise |
| rhBMP-2 (Infuse) | Osteoinductive | ALIF (with LT-Cage), acute open tibial shaft fractures | Active malignancy, active infection, skeletal immaturity, hypersensitivity to bovine collagen |
| rhBMP-7 (OP-1) | Osteoinductive | Recalcitrant long bone nonunions (Humanitarian Device Exemption) | Active malignancy, active infection, skeletal immaturity |
| Synthetic Ceramics (HA/TCP) | Osteoconductive | Metaphyseal void filling, extender for autograft | Diaphyseal segmental defects (lacks structural shear strength), highly contaminated fields |
Pre-Operative Planning, Templating, and Host Optimization
Meticulous pre-operative planning is the cornerstone of successful surgical management of bone defects and nonunions. The surgeon must first comprehensively evaluate the host's physiological capacity for healing. This requires a systemic assessment of the patient's vascular status, as the entire osteoinductive and osteoconductive cascade is entirely dependent on a robust blood supply. Endothelial dysfunction, characterized by impaired release of VWF, VEGF, and FGF-18, severely blunts the healing response. Clinical evaluation of peripheral pulses, capillary refill, and, when necessary, advanced vascular imaging (CT angiography or formal arteriography) must be performed to ensure that the surgical bed possesses the requisite perfusion to support cellular recruitment and graft incorporation.
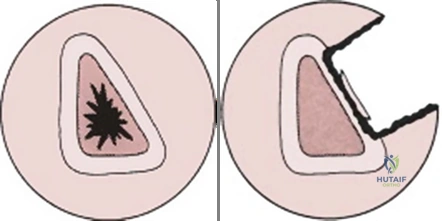
Radiographic templating is essential to define the mechanical and biological nature of the skeletal pathology. High-resolution orthogonal radiographs and fine-cut computed tomography (CT) scans are utilized to precisely quantify the volume of bone loss and assess the morphology of the nonunion. A hypertrophic nonunion, characterized by abundant "elephant foot" callus formation, possesses excellent intrinsic biology and vascularity but lacks mechanical stability; the pre-operative plan here focuses on rigid internal fixation rather than biologic augmentation. Conversely, an atrophic nonunion presents with tapered, sclerotic bone ends and no evidence of callus. This indicates a complete failure of the biological cascade, necessitating a surgical plan that includes aggressive debridement, rigid fixation, and the introduction of both osteoinductive stimuli (e.g., BMPs) and osteoconductive scaffolds.
Host optimization is arguably the most critical, yet frequently overlooked, phase of pre-operative planning. The surgeon must act as a physician, aggressively managing modifiable risk factors that impair the vascular endothelium and cellular metabolism. Smoking cessation is mandatory, as nicotine and carbon monoxide directly inhibit the proliferation of macrophages, disrupt the VWF-mediated hematoma, and downregulate the synthesis of essential inflammatory cytokines. Glycemic control in diabetic patients must be optimized, targeting a Hemoglobin A1c of less than 7.0%, to mitigate advanced glycation end-products (AGEs) that stiffen the collagen matrix and impair osteoblast function.
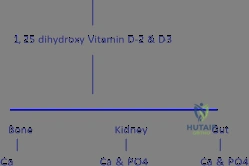
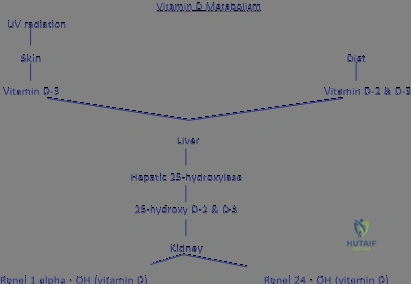
Furthermore, systemic nutritional parameters must be maximized prior to elective intervention. Vitamin D (25-hydroxyvitamin D) levels must be assessed and repleted to ensure adequate intestinal calcium absorption and proper mineralization of the newly formed osteoid. Parathyroid hormone (PTH) levels and thyroid function should be normalized to maintain the delicate balance of the RANKL/OPG axis. Only when the host's physiological milieu is optimized can the surgeon confidently proceed with complex reconstructions, knowing that the cellular machinery required to respond to osteoinductive signals and populate osteoconductive matrices is fully functional.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of bone regeneration procedures demands meticulous soft tissue handling and a profound respect for the local vascular anatomy. The surgical approach must be designed to utilize internervous and intermuscular planes, minimizing iatrogenic trauma to the delicate periosteal blood supply. The periosteum is a critical source of mesenchymal stem cells and vascular endothelial cells; its preservation is paramount for secondary bone healing. Once the nonunion site or bone defect is exposed, the first and most critical surgical step is radical debridement. All intervening fibrous tissue, necrotic cartilage, and sclerotic, avascular bone must be systematically excised until healthy, punctate bleeding bone is encountered—clinically referred to as the "paprika sign."

This aggressive debridement is not merely mechanical; it is a profound biological intervention. By exposing healthy, vascularized bone marrow and cortical haversian systems, the surgeon intentionally traumatizes the local microvasculature. This triggers the immediate release of endogenous Von Willebrand Factor, initiating platelet aggregation and the subsequent massive release of PDGF, VEGF, and endogenous BMPs directly into the surgical site. This endogenous release of inflammatory cytokines and damage to the bone ECM liberates matrix-bound growth factors, which potently induce the differentiation and proliferation of osteoprogenitor cells. Without this foundational step, any applied biologic adjunct will be rendered inert in an avascular, biologically dead space.

Following adequate preparation of the host bed, the surgeon must address the biological void. If an osteoconductive scaffold is required, materials such as cancellous allograft, structural cortical struts, or synthetic hydroxyapatite/tricalcium phosphate ceramics are meticulously packed into the defect. It is critical to eliminate all dead space, as fluid collections can serve as a nidus for infection and physically block cellular migration. If an osteoinductive stimulus is indicated, recombinant BMPs are prepared. When utilizing rhBMP-2, the protein is reconstituted and applied to the absorbable collagen sponge. The sponge is allowed to bind the protein for a mandatory minimum time before being delicately placed into the defect. The sponge must not be squeezed or tightly compressed, as this will prematurely extrude the water-soluble growth factor away from the target site.

The final step is the application of biomechanically appropriate fixation. The choice of implant dictates the mechanical strain at the fracture site, which in turn governs the biological pathway of healing. If the defect is grafted with structural allograft or if primary haversian remodeling is desired, absolute stability must be achieved. This is typically accomplished using dynamic compression plates and lag screws to generate high interfragmentary compression, effectively reducing strain to near zero. Alternatively, if secondary healing via callus formation is the goal—often the case in diaphyseal defects augmented with cancellous autograft—relative stability is provided using intramedullary nails or long bridge plates. This allows for controlled micro-motion, stimulating the piezoelectric effect and promoting the robust endochondral ossification cascade.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique and optimal host biology, the utilization of profound biologic agents and complex fixation strategies carries a distinct profile of severe complications. The introduction of highly concentrated, recombinant growth factors fundamentally alters the local physiological environment. rhBMP-2, while profoundly osteoinductive, is associated with a significant incidence of robust inflammatory reactions. Because BMPs are potent chemotactic agents, they induce a massive influx of macrophages and leukocytes. Clinically, this manifests as significant post-operative soft tissue swelling and seroma formation. In the appendicular skeleton, this is often manageable, but in confined anatomical spaces, the consequences can be dire.
The most infamous complication profile associated with rhBMP-2 occurred during its off-label use in anterior cervical discectomy and fusion (AC
Clinical & Radiographic Imaging Archive