Chordoma and Adamantinoma: Comprehensive Surgical Management and Pathology
Key Takeaway
Chordoma is a rare, slow-growing malignant bone neoplasm originating from embryological notochord remnants. Most commonly arising in the sacrococcygeal region and skull base, it requires aggressive en bloc surgical resection with wide margins to prevent high rates of local recurrence. This guide covers the clinical presentation, advanced imaging, histopathology, and step-by-step surgical management of chordoma, alongside a brief review of tibial adamantinoma.
CHORDOMA: PATHOPHYSIOLOGY AND EPIDEMIOLOGY
Chordoma is a rare, locally aggressive, and malignant neoplasm that originates from the embryological remnants of the notochord. Despite its slow growth rate, it is notoriously difficult to manage due to its critical anatomical locations and high propensity for local recurrence. In extensive institutional reviews, such as the Mayo Clinic series, chordomas account for approximately 4% of all primary malignancies of bone.
Within the axial skeleton, chordoma represents the second most common primary malignancy of the spine (surpassed only by multiple myeloma) and stands as the absolute most common primary malignancy of the sacrum.
The anatomical distribution of chordomas directly correlates with the developmental path of the notochord:
* Sacrococcygeal Region: > 50% of cases.
* Sphenooccipital Region (Skull Base): > 30% of cases.
* Mobile Spine (Cervical, Thoracic, Lumbar): The remaining 15-20%, distributed evenly, though cervical lesions are slightly more frequent than thoracic or lumbar.
Demographics and Incidence
The peak incidence of chordoma varies significantly depending on the anatomical site of the lesion. Sacrococcygeal chordomas typically present in older adults, with a peak incidence in the fifth to seventh decades of life. Conversely, sphenooccipital (skull base) lesions tend to present earlier, peaking in the fourth to sixth decades.
Most epidemiological series demonstrate a marked male predominance, with a male-to-female ratio of approximately 3:1. This gender disparity is especially pronounced in sacrococcygeal tumors.
CLINICAL PRESENTATION
Because the vast majority of chordomas are indolent and slow-growing, patients frequently experience a prolonged duration of symptoms—often exceeding a year—before a definitive diagnosis is established. The presenting signs and symptoms are highly variable and are dictated entirely by the anatomical site of the lesion and the adjacent structures subjected to mass effect.
Site-Specific Symptomatology
- Sacrococcygeal Tumors: The most common presenting complaint is deep, unrelenting low back pain. As the anterior soft-tissue mass expands into the presacral space, patients frequently develop bowel and bladder disturbances (e.g., constipation, tenesmus, urinary retention) and sciatic pain due to compression of the lumbosacral plexus.
- Sphenooccipital Tumors: Patients typically report severe headaches, visual disturbances, or symptoms related to direct cranial nerve compression (e.g., diplopia, facial numbness).
- Mobile Spine Tumors: Symptoms are primarily driven by nerve root or spinal cord compression, presenting as radiculopathy or myelopathy.
Clinical Pearl: If a large anterior mass exists in association with a cervical spine chordoma, the patient may present with dysphagia, airway compromise, or a palpable anterior neck mass. Clinically, these symptoms can closely mimic those caused by a retropharyngeal abscess.
Surgical Warning: A digital rectal examination (DRE) is a mandatory component of the physical examination for any patient presenting with intractable low back pain and bowel/bladder changes. A palpable, firm, presacral mass is frequently present on rectal examination in patients with sacrococcygeal chordoma.
DIAGNOSTIC IMAGING
The radiographic evaluation of a suspected chordoma requires a multimodal approach to accurately stage the local extent of the tumor and plan for complex en bloc resection.
Plain Radiography
Radiographically, chordomas appear as destructive, osteolytic lesions that virtually always arise from the midline of the axial skeleton.

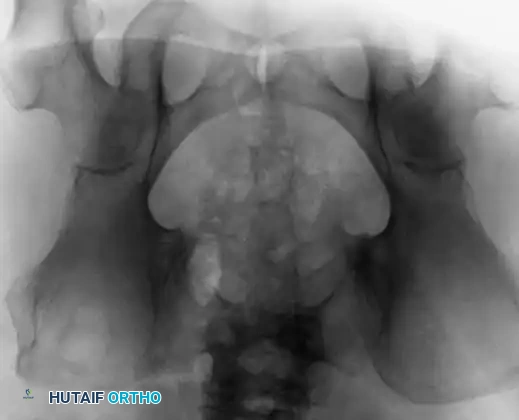
Fig. 22-11 A and B: Anteroposterior and lateral views of the sacrum of a patient with a sacrococcygeal chordoma. This lesion could be missed easily on the AP view because of overlying bowel gas.
Sacrococcygeal lesions are notoriously difficult to visualize on standard anteroposterior (AP) radiographs of the pelvis due to the superimposition of overlying bowel gas and fecal matter. They are usually seen much more clearly on a dedicated lateral view of the sacrum, which may demonstrate cortical destruction, anterior displacement of the presacral soft tissues, or a calcified mass.
Advanced Cross-Sectional Imaging
- Computed Tomography (CT): CT is highly sensitive for detecting intralesional calcification, which is present in more than 50% of chordomas. CT is also invaluable for assessing the degree of cortical bone destruction and planning osteotomy trajectories.
- Magnetic Resonance Imaging (MRI): MRI is the gold standard for determining the full anatomical extent of the lesion, evaluating marrow replacement, and delineating the tumor's relationship to critical neurovascular structures (e.g., the rectum, iliac vessels, and sacral nerve roots). Chordomas typically exhibit high signal intensity on T2-weighted images due to their high mucin content.
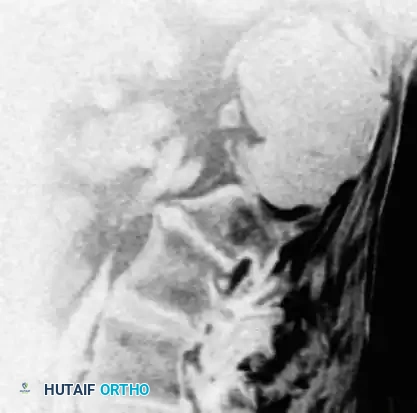
Fig. 22-11 C: Sagittal MRI clearly shows the extensive presacral lesion, demonstrating the characteristic high T2 signal and anterior displacement of pelvic viscera.
Diagnostic Pitfall: A common and potentially devastating pitfall in the evaluation of a patient with low back pain is ordering an MRI of only the lumbar spine. Standard lumbar MRI protocols typically end at the L5-S1 disc space or the S1 body. Because most sacrococcygeal chordomas arise below S3, a standard lumbar MRI will completely miss the tumor. If clinical suspicion exists, a dedicated MRI of the sacrum or a full pelvic MRI must be ordered.
- Bone Scintigraphy: Technetium-99m bone scans may show variable uptake. However, radioisotope accumulation in the urinary bladder can easily obscure a sacral tumor, limiting the utility of this modality for local staging.
HISTOPATHOLOGY
A definitive diagnosis of chordoma requires careful histopathological examination, often obtained via a CT-guided core needle biopsy. The biopsy tract must be meticulously planned so that it can be excised en bloc with the definitive surgical specimen to prevent tumor seeding.
Microscopically, chordoma is characterized by a lobular architecture. The tumor cells are arranged in lobules separated by prominent fibrous septa.
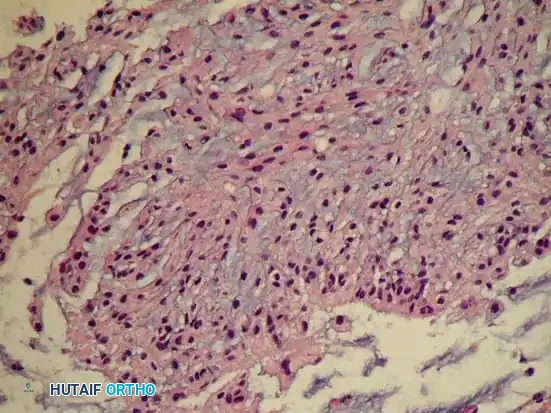
Fig. 22-11 D: Typical microscopic appearance of chordoma. Cells with abundant vacuolated cytoplasm (physaliferous cells) are arranged in cords with a mucinous background.
Cytological Features
The hallmark of a chordoma is the presence of physaliferous cells. These are large cells containing abundant, highly vacuolated cytoplasm, giving them a "bubbly" appearance. The cells are typically arranged in long strands, or "cords," set within a rich, myxoid or mucinous background stroma.
Immunohistochemically, chordomas are uniquely positive for Brachyury, a transcription factor crucial for notochordal development. They also typically express epithelial markers (cytokeratin, EMA) and S-100 protein.
Dedifferentiated Chordoma
While the vast majority of chordomas are histologically low-grade, a variant known as dedifferentiated chordoma exists. These tumors contain abrupt transitions from conventional low-grade chordoma to areas of high-grade sarcoma—most frequently resembling a malignant fibrous histiocytoma (MFH) or undifferentiated pleomorphic sarcoma. Dedifferentiated chordomas behave in a highly aggressive manner, with rapid growth and a high propensity for early systemic metastasis.
SURGICAL MANAGEMENT
The primary, curative treatment for chordoma is aggressive surgical resection with wide (negative) margins. Because chordomas are relatively radioresistant and chemoresistant, the initial surgical resection offers the patient the only realistic chance for a cure.
Principles of Resection
The goal is en bloc resection. Intralesional excision or tumor violation during surgery leads to a near 100% rate of local recurrence. The surgeon must achieve wide margins, even if this creates a permanent neurological deficit. Progressive growth of an incompletely resected tumor will inevitably create the same neurological deficits, accompanied by intractable pain, fungating local recurrence, and a higher risk of metastatic disease.
Sacrectomy: Step-by-Step Approach
For sacrococcygeal chordomas, a partial or total sacrectomy is required. The level of the sacral osteotomy dictates the postoperative neurological function.
* Preservation of S1-S3: Unilateral preservation of S1-S3 roots generally maintains normal bowel and bladder continence.
* Bilateral Sacrifice of S2-S4: Results in permanent neurogenic bowel and bladder, requiring self-catheterization and a colostomy.
1. Preoperative Preparation:
Extensive multidisciplinary planning involving orthopedic oncology, neurosurgery, colorectal surgery, and plastic surgery is required. Preoperative mechanical bowel preparation and prophylactic ureteral stenting are highly recommended.
2. Patient Positioning and Approach:
Depending on the proximal extent of the tumor, a combined anterior-posterior approach may be necessary.
* Anterior Phase (Supine): A laparotomy is performed to mobilize the rectum, ligate the internal iliac vessels (to reduce intraoperative hemorrhage), and release the anterior sacral attachments.
* Posterior Phase (Prone): A midline posterior incision is made. The gluteus maximus muscles are reflected laterally.
3. Osteotomy and En Bloc Delivery:
Laminectomies are performed proximal to the tumor to identify and protect the thecal sac and exiting nerve roots. The sacroiliac joints are exposed. Using a threadwire (Gigli saw) or osteotomes, the sacrum is transected at the planned level (e.g., between S1 and S2). The sacrotuberous and sacrospinous ligaments are divided, and the tumor is delivered en bloc from the surgical field.
Biomechanics and Spinopelvic Reconstruction
The biomechanical stability of the pelvic ring depends heavily on the sacroiliac (SI) joints.
* If the osteotomy is performed below the S1 level (preserving the S1 body and the upper half of the SI joints), the pelvic ring remains biomechanically stable, and instrumental reconstruction is rarely needed.
* If a Total Sacrectomy is performed (sacrificing the entire SI joint), the continuity between the axial spine and the appendicular pelvis is completely lost. This requires complex spinopelvic reconstruction, typically utilizing lumbar pedicle screws connected to iliac screws or rods, often augmented with structural allografts or custom 3D-printed titanium implants to prevent superior migration of the pelvis and allow for eventual weight-bearing.
Soft Tissue Coverage
Following a massive sacral resection, a large dead space is created, and the overlying skin is at high risk for necrosis. Plastic surgery consultation is vital. A pedicled Vertical Rectus Abdominis Myocutaneous (VRAM) flap or bilateral gluteal advancement flaps are frequently utilized to obliterate the dead space, provide healthy vascularized tissue over the spinal hardware, and prevent postoperative wound dehiscence and infection.
ADJUVANT THERAPIES
- Radiation Therapy: If wide margins cannot be obtained due to anatomical constraints, or if inadvertent tumor contamination occurs intraoperatively, adjuvant radiation therapy may be beneficial. High-dose proton beam therapy or carbon ion therapy is preferred over conventional photon radiation, as it allows for the delivery of tumoricidal doses while sparing adjacent critical structures (e.g., the rectum and spinal cord). Radiation may also be used for palliation in patients with unresectable disease, although a cure is rarely, if ever, achieved in these scenarios.
- Chemotherapy: Conventional cytotoxic chemotherapy is of no proven benefit in the treatment of classic chordoma. Targeted therapies (e.g., imatinib, EGFR inhibitors) are currently under investigation for advanced disease but remain experimental.
PROGNOSIS AND SURVEILLANCE
The clinical course of chordoma is characterized by a high rate of local recurrence, primarily due to the anatomical difficulties encountered in achieving wide surgical margins.
- Survival Rates: The 5-year overall survival rate for patients with chordomas is approximately 60% to 80%. However, the survival rate continues to decline steadily with longer follow-up due to late recurrences, dropping to a 25% to 40% survival rate at 10 years.
- Prognostic Factors: Male gender, younger age at diagnosis, and a more distal (caudal) location for sacral lesions are associated with a more favorable prognosis. The most critical prognostic factor remains the adequacy of the surgical margin.
- Metastasis: Distant metastases are rare at initial presentation (< 5%) but may occur later in the disease course in approximately 30% to 60% of patients. Metastases most commonly affect the lungs, but chordoma has a unique propensity to metastasize to bone, skin, eyelids, brain, liver, and other internal organs. Isolated distant metastases should be treated with aggressive surgical resection when feasible.
DIFFERENTIAL DIAGNOSIS: ADAMANTINOMA
While discussing rare, low-grade malignancies of bone, it is pertinent to briefly review adamantinoma, another distinct entity that requires aggressive surgical management.
Adamantinoma is a rare, low-grade malignant bone tumor that almost exclusively arises in the diaphysis of the tibia (often presenting alongside osteofibrous dysplasia). It typically presents in young adults with indolent, progressive anterior leg pain or a palpable mass.
Radiographic and Histological Features
Radiographically, adamantinoma presents as a multiloculated, expansile, eccentric radiolucent lesion in the anterior tibial cortex, often described as having a "soap-bubble" appearance.
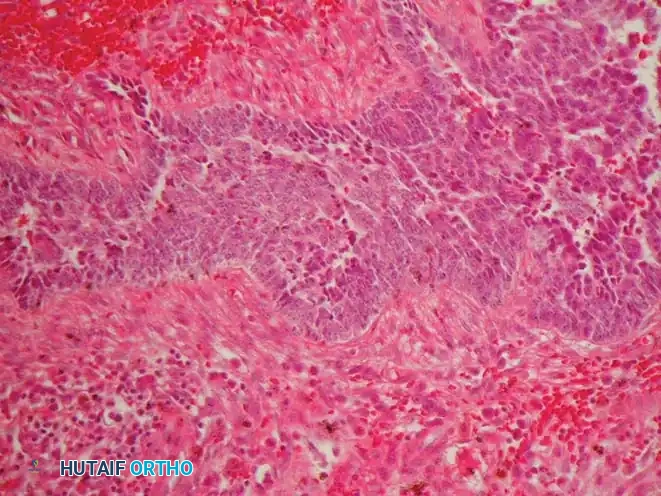
Fig. 22-12 C: Typical microscopic appearance of adamantinoma. Islands of epithelioid cells are seen within a fibrous background.
Microscopically, adamantinoma is characterized by islands, nests, or cords of epithelioid cells surrounded by a bland, spindle-cell fibrous stroma. Immunohistochemically, the epithelioid cells are strongly positive for cytokeratins and vimentin, confirming their biphasic nature. Histological features (e.g., mitotic rate) are generally not predictive of the tumor's clinical behavior.
Treatment and Prognosis of Adamantinoma
Similar to chordoma, adamantinoma is generally radioresistant and chemoresistant. The optimal and only definitive treatment is surgical: wide en bloc resection (often requiring intercalary allograft or vascularized fibular graft reconstruction) or amputation.
Compared with patients who undergo marginal or intralesional surgical procedures, those treated with wide resection have significantly better outcomes. Local recurrence occurs in approximately 25% of patients, and amputation should be strongly considered for these salvage cases. Metastases are rare at presentation but may occur later in up to 30% of patients, primarily to the lungs or regional lymph nodes. Overall survival is excellent if adequately resected, approaching 85% at 10 years. Prognosis depends almost entirely on the adequacy of the initial surgical margin.
📚 Medical References
- chordoma, Ann Surg 166:394, 1967.
- Longhi A, Fabbri N, Donati D, Capanna R, et al: Neoadjuvant chemotherapy for patients with synchronous multifocal osteosarcoma: results in eleven cases, J Chemother 13:324, 2001.
- Lukens JA, McLeod RA, Sim FH: Computed tomographic evaluation of primary osseous malignant neoplasms, AJR Am J Roentgenol 139:45, 1982.
- MacCarty CS, Waugh JM, Coventry MB, et al: Surgical treatment of sacral and presacral tumors other than sacrococcygeal chordoma, J Neurosurg 22:458, 1965.
- MacCarty CS, Waugh JM, May CW, et al: The surgical treatment of presacral tumors: a combined problem, Mayo Clin Proc 27:73, 1952.
- Mackinnon J, Aziz T, Dixon JH: Interscapulothoracic resection: a case report and review of the literature, J Bone Joint Surg 70B:791, 1988.
- Maeda M, Tateishi H, Takaiwa H, et al: High-energy, low-dose radiation therapy for aneurysmal bone cyst: report of a case, Clin Orthop Relat Res 243:200, 1989.
- Majhail N, Urbain J, Albani J, et al: F-18 fl uorodeoxyglucose positron emission tomography in the evaluation of distant metastases from renal cell carcinoma, J Clin Oncol 21:3995, 2003.
- Malawer MM: Surgical management of aggressive and malignant tumors of the proximal fi bula, Clin Orthop Relat Res 186:172, 1984.
- Malawer MM: Tumors of the shoulder girdle: technique of resection and description of a surgical classifi cation, Orthop Clin North Am 22:7, 1991.
- Malawer MM, Buch R, Reaman G, et al: Impact of two cycles of preoperative chemotherapy with intraarterial Cisplatin and intravenous Doxorubicin on the choice of surgical procedure for highgrade bone sarcomas of the extremities, Clin Orthop Relat Res 270:214, 1991.
- Malawer MM, Chou LB: Prosthetic survival and clinical results with use of large-segment replacements in the treatment of high-grade bone sarcomas, J Bone Joint Surg 77A:1154, 1995.
- Malawer MM, Dunham W: Cryosurgery and acrylic cementation as surgical adjuncts in the treatment of aggressive (benign) bone tumors, Clin Orthop Relat Res 262:42, 1991.
- Malawer MM, Dunham WK: Skip metastases in osteosarcoma: recent experience, J Surg Oncol 22:236, 1983.
- Malawer MM, Marks MR, McChesney D, et al: The effect of cryosurgery and polymethylmethacrylate in dogs with experimental bone defects comparable to tumor defects, Clin Orthop Relat Res 226:299, 1988.
- Malawer MM, McHale KA: Limb-sparing surgery for high-grade malignant tumors of the proximal tibia: surgical technique and a method of
You Might Also Like