Introduction & Epidemiology
Acute tophaceous gout presenting as hand pain in the elderly population poses a significant diagnostic challenge for orthopedic surgeons. While classically associated with monoarticular involvement of the first metatarsophalangeal (MTP) joint, gout's presentation in the elderly is often atypical, polyarticular, and commonly affects the upper extremities, mimicking various inflammatory and degenerative arthropathies. The prevalence of gout increases with age, affecting approximately 7-10% of men and 3-5% of women over 65 years. In this demographic, polypharmacy, comorbidities such as renal insufficiency, hypertension, and heart failure, and less pronounced inflammatory responses can obscure the classical presentation, leading to misdiagnosis and delayed appropriate management.
Tophaceous gout, characterized by the deposition of monosodium urate (MSU) crystals in soft tissues, joints, and bones, represents a chronic, often undertreated stage of the disease. In the hand, tophi can develop in periarticular soft tissues, tendon sheaths, bursae, and even within bone, leading to significant pain, deformity, functional impairment, nerve compression syndromes (e.g., carpal tunnel syndrome, cubital tunnel syndrome), and skin ulceration. The acute exacerbation of pain in established tophaceous disease, often referred to as an acute flare within a tophus or a newly inflamed tophus, necessitates precise diagnosis to differentiate it from septic arthritis, rheumatoid arthritis (RA), calcium pyrophosphate deposition disease (CPPD), osteoarthritis (OA) with inflammatory flares, or even neoplastic processes. The stakes are particularly high in the elderly due to the potential for severe morbidity and the need to avoid inappropriate surgical intervention for non-gout pathology or, conversely, to intervene appropriately for gout-related complications.
Early and accurate diagnosis of acute tophaceous gout in the hand is paramount. It guides prompt initiation of anti-inflammatory treatment and long-term urate-lowering therapy (ULT), preventing irreversible joint damage and systemic complications. This review provides a comprehensive guide for orthopedic surgeons, residents, and medical students on the diagnostic approach and management considerations for this challenging clinical entity.
Surgical Anatomy & Biomechanics
Understanding the intricate anatomy and biomechanics of the hand and wrist is crucial for both diagnosing and managing tophaceous gout. The hand's complex architecture, comprising numerous small joints (metacarpophalangeal, interphalangeal), tendons, tendon sheaths, ligaments, and neurovascular bundles, provides multiple potential sites for MSU crystal deposition and subsequent tophus formation.
- Joints: While the distal interphalangeal (DIP) joints are classically associated with OA and psoriatic arthritis, and the metacarpophalangeal (MCP) joints with RA, tophaceous gout can involve any joint in the hand and wrist. The synovium, being a highly vascularized tissue, is a primary site for crystal deposition. Chronic inflammation leads to synovial hypertrophy, pannus formation, and progressive erosion of articular cartilage and subchondral bone.
- Tendons and Tendon Sheaths: The flexor and extensor tendon sheaths are common sites for tophus formation, often leading to tenosynovitis, trigger digit, or even spontaneous tendon rupture. The presence of tophi within a tendon can alter its excursion and mechanical properties, predisposing it to attritional wear.
- Ligaments and Bursae: Periarticular ligaments and bursae (e.g., olecranon bursa, prepatellar bursa, wrist bursae) can also accumulate MSU crystals, resulting in localized swelling, pain, and reduced joint stability in chronic cases.
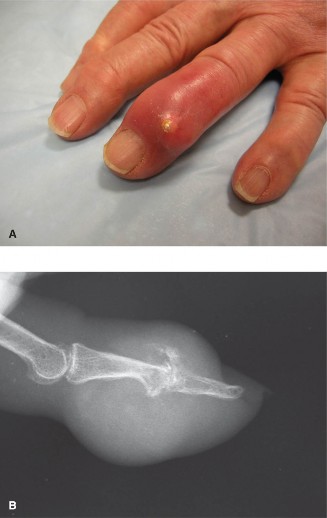
- Bone and Cartilage: MSU crystals have a predilection for relatively avascular tissues, including articular cartilage. Over time, chronic tophaceous deposits within bone lead to characteristic "punched-out" erosions, often with sclerotic margins and an "overhanging edge" sign on plain radiographs, which is highly suggestive of gout. These erosions can be extra-articular or intra-articular and often occur adjacent to an intact joint space until very late stages, distinguishing them from other inflammatory arthropathies where joint space narrowing occurs earlier.
- Nerve Compression Syndromes: Tophi can expand within confined anatomical spaces, such as the carpal tunnel or Guyon's canal, leading to compression neuropathies (e.g., median nerve or ulnar nerve compression). This mechanical compression adds a neurological component to the patient's pain and dysfunction.
From a biomechanical perspective, the presence of tophi mechanically impedes joint motion, creates impingement, and compromises tendon gliding. The inflammatory process itself degrades cartilage and subchondral bone, leading to joint instability and secondary OA. The crystalline deposition alters the elastic properties of tissues, making them more susceptible to injury and rupture under normal physiological stresses. Differentiating true biomechanical failure from an inflammatory flare is critical for surgical planning.
Indications & Contraindications
While the primary focus is on diagnosis, understanding the indications and contraindications for surgical intervention in tophaceous gout is essential for the orthopedic surgeon. Surgical management is typically reserved for complications or diagnostic necessity when conservative measures fail.
Indications for Surgical Intervention in Tophaceous Gout
-
Diagnostic Uncertainty:
- Failure to confirm diagnosis via synovial fluid aspiration or when aspiration is not feasible.
- Need for open biopsy to differentiate gout from other conditions (e.g., infection, malignancy, other crystal arthropathies).
-
Complications of Tophi:
- Nerve Compression Syndromes: Clinically significant carpal tunnel syndrome, cubital tunnel syndrome, or other peripheral neuropathies caused by tophaceous compression refractory to medical management.
- Skin Ulceration and Impending Rupture: Large, rapidly expanding tophi causing skin necrosis, ulceration, or imminent skin breakdown, increasing the risk of secondary infection. This is particularly crucial in the elderly with fragile skin.
- Infection: Infected tophi or joints where surgical debridement and drainage are necessary in conjunction with antimicrobial therapy. Distinguishing an infected tophus from a sterile gout flare is a critical indication for urgent aspiration and potentially surgical intervention.
- Tendon Rupture: Spontaneous or attritional tendon rupture caused by tophaceous invasion or severe tenosynovitis, requiring surgical repair or reconstruction.
- Severe Joint Destruction and Instability: Advanced erosive changes leading to disabling pain, instability, or loss of function, potentially necessitating arthroplasty (e.g., MCP joint arthroplasty) or arthrodesis.
- Mass Effect/Functional Impairment: Large tophi causing significant mechanical obstruction, limiting range of motion, or interfering with activities of daily living, despite adequate urate-lowering therapy.
- Cosmetic Deformity: While generally a secondary indication, exceptionally large and disfiguring tophi, particularly in exposed areas like the hands, may warrant surgical debulking if functional impairment is also present or imminent.
Contraindications for Surgical Intervention
- Acute Gout Flare: Surgical intervention is generally contraindicated during an acute inflammatory flare. Surgery can exacerbate the inflammatory response and potentially trigger a new flare. Medical management (NSAIDs, colchicine, corticosteroids) should be optimized to quiescent the inflammation before elective surgery.
- Uncontrolled Hyperuricemia: While not an absolute contraindication for urgent surgical issues (e.g., infection, severe nerve compression), elective surgical intervention should ideally be deferred until serum uric acid (SUA) levels are adequately controlled by urate-lowering therapy (typically below 6 mg/dL, or below 5 mg/dL in severe tophaceous gout). High SUA levels increase the risk of post-operative flares and continued crystal deposition.
- Active Infection (Uncontrolled): Systemic or widespread local infection must be managed appropriately before definitive surgery on tophi.
- Significant Medical Comorbidities: Elderly patients often have multiple comorbidities (cardiac disease, renal failure, diabetes, coagulopathies) that significantly increase surgical and anesthetic risks. A thorough pre-operative medical workup and risk-benefit analysis are essential.
- Coagulopathy: Uncorrected bleeding disorders increase the risk of hematoma formation, particularly after extensive tophus debridement.
Operative vs. Non-Operative Indications
| Indication Category | Operative Management | Non-Operative Management |
|---|---|---|
| Diagnosis | Open biopsy for inconclusive aspiration/imaging | Synovial fluid aspiration (polarized microscopy) |
| Imaging (X-ray, US, CT, MRI) | ||
| Acute Flare Management | Urgent debridement for infected tophus/septic joint | NSAIDs, Colchicine, Corticosteroids |
| Chronic Disease Mgmt. | Tophus excision/debridement (large, symptomatic tophi) | Urate-lowering therapy (ULT - allopurinol, febuxostat, probenecid) |
| Nerve decompression (e.g., carpal tunnel release) | Lifestyle modifications (diet, weight loss) | |
| Tendon repair/reconstruction (for rupture) | Acute flare prophylaxis (low-dose colchicine, NSAIDs) | |
| Arthroplasty/Arthrodesis (for severe joint destruction) | Pain management (analgesics) | |
| Complications | Surgical debridement for ulcerated/infected tophi | Antibiotics for cellulitis/local infection (without deep involvement) |
| Amputation (rare, for severe, uncontrolled infection/gangrene) | Physical therapy for functional limitations |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential for any surgical intervention related to tophaceous gout, whether for diagnostic purposes or for managing complications. This is particularly true in the elderly population with their inherent medical complexities.
Pre-operative Evaluation
-
Detailed History and Physical Examination:
- History: Focus on the chronicity and frequency of gout flares, duration of hyperuricemia, current and past urate-lowering therapy adherence, specific hand pain characteristics (acute vs. chronic, localization, radiation), associated symptoms (numbness, tingling, weakness indicative of nerve compression), functional limitations, and prior treatments. A comprehensive medical history is crucial, noting comorbidities (renal impairment, cardiovascular disease, diabetes), allergies, and a complete medication list (including diuretics, aspirin, ciclosporin, which can affect urate levels).
- Physical Examination: Assess the hand and wrist for warmth, erythema, swelling, tenderness, joint effusions, palpable tophi (location, size, consistency), skin integrity over tophi, neurological status (motor and sensory deficits, Phalen's, Tinel's signs), range of motion (active and passive), and grip strength. Compare findings to the contralateral hand.
-
Laboratory Studies:
- Serum Uric Acid (SUA): While often normal or even low during an acute flare, baseline SUA is important for long-term management.
- Complete Blood Count (CBC): To assess for anemia, leukocytosis (may indicate infection or inflammation).
- Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP): Non-specific inflammatory markers that are often elevated during acute flares or infection.
- Renal Function Panel (BUN, Creatinine, eGFR): Essential due to the high prevalence of renal impairment in gout patients and its impact on medication choices and anesthetic risk.
- Coagulation Profile (PT/INR, PTT): Important, especially if extensive dissection or debridement is anticipated, given potential for hematoma.
-
Synovial Fluid Analysis:
This is the gold standard for definitive diagnosis. Aspiration from a joint effusion or suspected tophus should be performed whenever feasible. Analysis includes cell count with differential (often >50,000 WBCs/mm³ with neutrophil predominance during acute flares, though can be lower in elderly/corticosteroid-treated patients), Gram stain, culture, and, most critically, polarized light microscopy for identification of negatively birefringent needle-shaped MSU crystals.
-
- Figure: Classic negatively birefringent, needle-shaped monosodium urate crystals observed under polarized light microscopy, a definitive diagnostic finding in acute gout.
-
-
Imaging Studies:
- Plain Radiographs: Initial imaging of choice. Look for soft tissue swelling, characteristic "punched-out" erosions with sclerotic margins and overhanging edges (Martel sign), preservation of joint space until late stages, and calcifications (though less common than CPPD). Radiographic changes are often absent in early disease.
- Ultrasound (US): Highly sensitive for detecting early changes. Key findings include the "double contour sign" (hyperechoic line on the surface of articular cartilage, representing MSU crystal deposition), identification of non-palpable tophi, synovial proliferation, and increased vascularity indicative of inflammation. It is also useful for guiding aspiration of effusions or small tophi.
- Computed Tomography (CT): Provides detailed assessment of bone erosions, intraosseous tophi, and periarticular soft tissue tophi. Dual-energy CT (DECT) can specifically identify and quantify MSU crystal deposits based on their unique attenuation properties, proving highly valuable for diagnostic confirmation, mapping crystal burden, and monitoring treatment response, especially in atypical presentations or when aspiration is difficult.
- Magnetic Resonance Imaging (MRI): Useful for assessing soft tissue involvement (tenosynovitis, bursitis), nerve compression, synovial thickening, and differentiating gouty inflammation from infection or other pathologies. Tophi typically appear as intermediate signal intensity on T1-weighted images and variable signal on T2-weighted images, often with rim enhancement after gadolinium administration.
Patient Positioning
For surgical procedures on the hand and wrist related to tophaceous gout, the standard positioning is:
- Supine Position: Patient supine on the operating table.
- Arm on Hand Table: The affected arm is placed on a radiolucent hand table, ensuring proper ergonomics for the surgical team and accessibility for fluoroscopy if needed.
- Tourniquet: A pneumatic tourniquet is applied to the proximal arm or forearm to provide a bloodless field, crucial for meticulous dissection and identification of vital structures amidst inflammatory tissue and tophaceous deposits.
- Sterile Preparation and Draping: Standard sterile preparation of the limb from the elbow to the fingertips, followed by sterile draping, isolating the operative field.
- Anesthesia: Regional anesthesia (e.g., axillary block or supraclavicular block) is often preferred, especially in elderly patients, to minimize systemic side effects of general anesthesia. Local infiltration with epinephrine may be used in conjunction or for minor procedures.
Detailed Surgical Approach / Technique
Surgical intervention for tophaceous gout in the hand is not a primary treatment but rather an approach to confirm diagnosis or manage complications. The specific technique depends on the indication (diagnostic aspiration/biopsy, tophus excision, nerve decompression, joint reconstruction).
1. Arthrocentesis / Synovial Fluid Aspiration (Diagnostic Gold Standard)
This procedure is typically performed in a clinic or emergency setting but is fundamental for diagnosis.
*
Site Selection:
Identify the maximally swollen or painful joint or the most prominent soft tissue swelling suggestive of a tophus. Avoid areas with overlying skin infection or prominent neurovascular structures. For an acute flare, aim for the joint effusion. For a suspected tophus, aim for the soft, fluctuant part.
*
Sterile Technique:
Meticulous sterile technique is paramount to prevent iatrogenic infection. Skin preparation with chlorhexidine or povidone-iodine. Local anesthetic (e.g., 1% lidocaine) infiltration.
*
Aspiration:
1. Use a 25-gauge or 22-gauge needle for joint aspirations in small joints, and larger bore (18-gauge) for thick synovial fluid or tophi if needed.
2. Insert the needle into the joint space or suspected tophus, applying gentle negative pressure with a syringe.
3. Aspirate sufficient fluid (at least 0.5-1 mL) for analysis. If no fluid is obtained, inject a small amount of sterile saline (0.5 mL) and re-aspirate (saline lavage technique).
4. Transfer fluid immediately to appropriate containers: plain tube for crystal analysis, sterile tube for culture, EDTA tube for cell count.
*
Post-Aspiration:
Apply pressure, sterile dressing. Educate patient on potential flare and ice application.
*
Fluid Analysis:
Rapid transport to the lab for:
*
Cell count and differential:
Differentiates inflammatory from non-inflammatory.
*
Gram stain and culture:
Rule out septic arthritis.
*
Polarized light microscopy:
Crucial for identifying negatively birefringent, needle-shaped MSU crystals.
This is the definitive diagnostic test.
2. Open Biopsy / Tophus Excision (Debridement)
This is indicated for diagnostic uncertainty, nerve compression, impending skin breakdown, or functional impairment from large tophi.
-
Incision Planning:
- Longitudinal Incisions: Generally preferred in the hand and wrist, placed strategically to avoid neurovascular bundles, crossing joint creases transversely, and on the non-functional (dorsal or less loaded) side if possible.
- Elliptical Incision: May be necessary if the overlying skin is severely compromised or ulcerated, to excise non-viable skin along with the tophus.
- Blood Supply: Consider local skin flap viability, especially in areas with poor perfusion or previous surgery.
-
Dissection:
- Identification of Anatomical Structures: Meticulous dissection under tourniquet control is critical. Tophi can encapsulate nerves, vessels, and tendons. Use magnification (loupes) for precise identification and protection of vital structures.
- Tophi Characteristics: Tophi often appear as chalky white, yellowish, or grayish granular material, sometimes encased in a fibrous capsule or surrounded by a pseudobursa. They can be firm, hard, or paste-like.
-
Excision Principles:
- Subcapsular Excision: For smaller, well-demarcated tophi, careful dissection along their fibrous capsule allows for complete removal while preserving surrounding healthy tissue.
- Debulking: For very large, diffuse tophi, complete removal may not be feasible or safe. In such cases, debulking to reduce mass effect and alleviate symptoms is the goal. Care must be taken to avoid leaving large exposed surfaces of tophaceous material, which can delay wound healing or predispose to infection.
- Pseudobursae Management: Chronic tophi often form within or around bursal sacs. These inflamed bursal walls should be excised along with the tophus.
- Nerve Decompression: If nerve compression (e.g., median nerve in carpal tunnel) is identified pre-operatively or intra-operatively, standard nerve decompression techniques (e.g., carpal tunnel release, ulnar nerve release) are performed following tophus removal in the vicinity of the nerve. Ensure complete release of all compressive elements.
- Tendon Debridement/Repair: If tophi are invading tendon substance or causing tenosynovitis, careful debridement of tophaceous material from the tendon can be performed. If tendon rupture has occurred, primary repair or reconstruction may be necessary, often using local tendon grafts or transfers depending on the specific tendon and defect.
-
Wound Management:
- Copious Irrigation: After tophus removal, thorough irrigation of the wound with saline helps to remove residual particulate matter and reduce the inflammatory burden.
- Hemostasis: Meticulous hemostasis to prevent post-operative hematoma formation, which can increase the risk of infection and delayed wound healing.
-
Closure:
- Layered Closure: Close deep fascial layers and subcutaneous tissue if possible, to eliminate dead space.
- Skin Closure: Close skin with fine non-absorbable sutures. Avoid excessive tension. In cases of significant skin loss, local flaps or skin grafting may be required.
- Specimen Handling: All excised tissue must be sent for histopathological examination. Histology will reveal a characteristic foreign body granulomatous reaction with birefringent urate crystals. Culture should also be performed if infection is suspected.
Complications & Management
Complications following surgical intervention for tophaceous gout in the hand can range from common surgical issues to those specific to the underlying disease process. Meticulous pre-operative planning and surgical technique are paramount to minimize these risks.
Common Complications and Management Strategies
| Complication | Incidence (Approximate) | Salvage Strategies / Management |
|---|---|---|
| Acute Post-operative Flare | 5-20% |
Prevention is Key:
Prophylaxis with low-dose colchicine (0.5-0.6 mg BID) or NSAIDs initiated pre-operatively and continued for several weeks post-operatively.
Treatment: Oral corticosteroids (e.g., prednisone 0.5 mg/kg/day for 5-10 days, then taper) are often preferred over NSAIDs due to potential renal issues in elderly. Colchicine can be restarted. Intra-articular corticosteroid injection may be considered for localized flare. Ensure patient is on adequate urate-lowering therapy. |
| Wound Healing Complications | 10-30% |
Risk Factors:
Poor skin integrity over chronic tophi, steroid use, diabetes, poor nutrition, extensive debridement.
Management: * Hematoma: Evacuation, meticulous hemostasis. * Dehiscence/Necrosis: Local wound care, serial debridement, negative pressure wound therapy (NPWT), secondary closure, or skin grafting/local flap coverage if severe. Optimize nutritional status. Manage underlying comorbidities. |
| Infection (Superficial/Deep) | 2-10% |
Risk Factors:
Open wounds, skin ulceration, compromised skin, hematoma.
Management: * Superficial: Oral antibiotics targeting common skin flora, local wound care. * Deep/Septic Arthritis: Urgent surgical irrigation and debridement, broad-spectrum IV antibiotics tailored to culture results. Repeated debridements may be necessary. Consider empiric treatment for Gram-positive and Gram-negative organisms. |
| Nerve Injury (Iatrogenic) | <1-5% |
Prevention:
Meticulous dissection, use of loupes, careful identification and protection of neurovascular structures.
Management: * Neuropraxia: Observation, hand therapy, electrodiagnostic studies if persistent. * Axonotmesis/Neurotmesis: Surgical exploration, neurolysis, or nerve repair/grafting depending on the extent of injury and functional deficit. |
| Persistent Pain / Recurrence | Variable (Long-term) |
Management:
* Inadequate ULT: Re-evaluate and optimize urate-lowering therapy (SUA target <6 mg/dL, or <5 mg/dL in severe tophaceous gout). Ensure patient adherence. * Residual Tophi: If symptomatic and causing functional limitation, revision surgery may be considered after a period of stable SUA control. * Secondary OA: Symptomatic management, potential arthroplasty/arthrodesis. * Complex Regional Pain Syndrome (CRPS): Early diagnosis, multidisciplinary pain management. |
| Joint Stiffness / Reduced ROM | 10-25% |
Risk Factors:
Prolonged immobilization, severe pre-operative joint destruction, extensive surgical dissection.
Management: Early and aggressive hand therapy (mobilization exercises, scar massage, splinting). Manipulation under anesthesia may be considered for recalcitrant stiffness after adequate wound healing. Tenolysis may be indicated for severe tendon adhesions. |
| Tendon Rupture | <1% (Post-op), Higher (Spontaneous) |
Prevention:
Careful dissection around tendons, avoiding excessive debridement of tendon substance.
Management: Surgical repair or reconstruction (tendon graft/transfer) depending on the affected tendon, defect size, and patient factors. |
| Urate Nephropathy/Renal Failure | Systemic Complication |
Prevention:
Maintain adequate hydration, monitor renal function closely. Avoid nephrotoxic drugs.
Management: Nephrology consultation, dialysis if acute severe kidney injury. This is a general complication of gout, not usually post-surgical, but highlights systemic risk. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial following surgical intervention for tophaceous gout in the hand to restore function, prevent stiffness, and facilitate long-term recovery. The protocol must be tailored to the specific surgical procedure performed and the individual patient's baseline function and comorbidities. Close collaboration between the surgeon, hand therapist, and rheumatologist is essential.
Phase I: Early Post-operative (Days 0-14)
- Goals: Protect surgical site, control pain and edema, maintain mobility of uninvolved joints.
-
Protection:
- Immobilization: A static volar or dorsal splint is applied immediately post-operatively to protect the surgical site, maintain anatomical alignment, and prevent undue stress on repairs. Duration depends on the procedure (e.g., 1-2 weeks for diagnostic biopsy/minor debulking, up to 4-6 weeks for tendon repair or complex joint reconstruction).
- Wound Care: Regular wound checks, dressing changes as per surgeon's protocol. Monitor for signs of infection (erythema, warmth, purulence). Suture removal typically at 10-14 days.
-
Edema Control:
- Elevation: Keep hand elevated above heart level, especially during the first few days.
- Gentle Compression: Soft compression dressing once the initial bulky dressing is removed.
- Ice Packs: Apply ice to the surgical area for 15-20 minutes every 2-3 hours to reduce swelling and pain.
-
Pain Management:
- Prescribed oral analgesics. Avoid NSAIDs immediately post-op if there is a risk of bleeding or renal impairment, but they may be initiated for flare prophylaxis after a few days.
-
Mobility (Uninvolved Joints):
- Gentle active range of motion (AROM) exercises for shoulder, elbow, and uninvolved fingers to prevent stiffness.
- Patient Education: Instruct on signs of complications, medication adherence, and the importance of hand hygiene.
Phase II: Intermediate (Weeks 2-6)
- Goals: Gradually increase AROM and passive range of motion (PROM) of involved joints, initiate light strengthening, manage scar tissue.
-
Range of Motion:
- Splint Weaning: Gradual weaning from the protective splint, often transitioning to a removable custom splint for night wear or specific activities.
- AROM: Start gentle active range of motion exercises for the surgical digits/wrist, guided by the hand therapist. Progress gradually as pain allows.
- PROM: Passive range of motion exercises, initiated cautiously, respecting pain limits and surgical precautions (e.g., no aggressive PROM if tendon repair or joint capsule repair).
- Blocking Exercises: For specific joint or tendon glide improvement.
-
Scar Management:
- Initiate scar massage once the wound is healed to prevent adhesions and reduce sensitivity.
- Use silicone gel sheets or topical creams.
-
Strengthening:
- Begin with very light isometric exercises for intrinsic and extrinsic muscles. Progress to isotonic exercises with minimal resistance (e.g., putty, resistance bands).
- Edema Management: Continue elevation, retrograde massage, and compression as needed.
Phase III: Advanced (Weeks 6+)
- Goals: Progressive strengthening, endurance, functional activity retraining, return to full activity.
-
Strengthening:
- Advance strengthening exercises with increasing resistance, incorporating grip and pinch strength activities.
- Focus on functional movements relevant to the patient's daily activities, hobbies, and work.
-
Endurance:
- Repetitive fine motor tasks to improve endurance and dexterity.
-
Functional Training:
- Simulation of work-specific or sport-specific activities.
- Neuromuscular re-education.
- Splinting: Discontinue splinting as functional goals are met and joint stability is confirmed, though some patients may benefit from occasional protective splinting during strenuous activities.
- Medical Management Reinforcement: Crucial to emphasize continued adherence to urate-lowering therapy and regular follow-up with the rheumatologist to prevent future flares and crystal deposition. This is paramount to surgical success and long-term joint health.
- Return to Activity: Gradual return to full activities, guided by functional assessment and patient comfort.
Summary of Key Literature / Guidelines
The management of gout, including acute tophaceous gout in the hand, is continuously evolving, guided by robust clinical research and consensus guidelines from major professional bodies. For orthopedic surgeons, understanding these overarching principles is critical, particularly regarding the interface between medical and surgical management.
-
American College of Rheumatology (ACR) Guidelines for the Management of Gout: These comprehensive guidelines are the cornerstone for medical management.
- Diagnosis: ACR emphasizes that definitive diagnosis relies on the identification of negatively birefringent MSU crystals in synovial fluid. While clinical presentation and imaging can be highly suggestive, crystal identification remains the gold standard. In cases where aspiration is not possible or inconclusive, advanced imaging (DECT, US) or surgical biopsy are considered.
- Urate-Lowering Therapy (ULT): The primary goal of long-term gout management is to achieve and maintain target serum uric acid (SUA) levels, typically below 6 mg/dL, and for patients with tophaceous gout, below 5 mg/dL to promote tophus dissolution. Xanthine oxidase inhibitors (allopurinol, febuxostat) are first-line ULT.
- Acute Flare Management: Guidelines recommend NSAIDs, colchicine, or corticosteroids as first-line agents. Combination therapy may be used for severe flares. Prophylaxis for flares is recommended during the initiation and titration of ULT.
- Comorbidities: ACR guidelines highlight the importance of managing comorbidities common in gout patients, particularly renal impairment, cardiovascular disease, and hypertension, as these impact treatment choices.
-
Imaging in Gout:
- Ultrasound: Literature increasingly supports the use of musculoskeletal ultrasound as a sensitive tool for early detection of MSU deposits (double contour sign, tophi) and for guiding aspirations. Studies by Filippucci et al. (2007) and others have validated its utility.
- Dual-Energy CT (DECT): Recognized as a non-invasive method for specific identification and quantification of MSU crystal deposits, especially in complex cases or when aspiration is difficult. Its role in assessing total body urate burden and monitoring treatment response is emerging (Molloy et al., 2017; Dalbeth et al., 2016).
- Radiographs: While often normal in early disease, characteristic erosions (Martel sign) are crucial for chronic tophaceous gout, as described by Martel et al. (1968).
-
Surgical Management of Tophaceous Gout:
- Surgical intervention, while not a first-line treatment, plays a critical role in addressing complications. Literature reviews (e.g., Roddy & Doherty, 2010; Khanna et al., 2012) emphasize that surgery is indicated for nerve compression, severe functional impairment due to large tophi, skin ulceration, infection, and severe joint destruction.
- Studies on surgical outcomes for tophaceous gout in the hand and wrist suggest favorable results for nerve decompression and tophus excision in selected patients, particularly with stable long-term urate control. However, recurrence of tophi or post-operative flares remain concerns if ULT is suboptimal.
- The importance of timing surgery (avoiding acute flares) and initiating prophylactic anti-inflammatory agents perioperatively is consistently highlighted in surgical literature (e.g., Yu et al., 2007).
-
Gout in the Elderly: Specific challenges in diagnosing and managing gout in the elderly are increasingly recognized. Atypical presentations, polyarticular involvement, hand localization, and the presence of comorbidities and polypharmacy necessitate a high index of suspicion and a multi-disciplinary approach involving rheumatology, orthopedics, and geriatrics (Pascual, 2006; Perez-Ruiz et al., 2015).
In conclusion, accurate diagnosis of acute tophaceous gout in the elderly hand requires a high index of suspicion, a thorough clinical evaluation, and judicious use of diagnostic modalities, with crystal identification remaining the definitive criterion. Orthopedic surgeons must be adept at recognizing the varied presentations, understanding the indications for surgical intervention in complicated cases, and coordinating care with rheumatologists to ensure optimal long-term outcomes and prevent recurrent flares and progressive joint destruction.
