Introduction & Epidemiology
The successful management of soft tissue defects in the hand, particularly those involving exposure of critical underlying structures such as tendons, bones, and joints, represents a formidable challenge in reconstructive orthopedic surgery. The hand's unparalleled functional importance—encompassing intricate fine motor skills, robust grip strength, and essential sensory perception—demands reconstructive solutions that prioritize not only wound closure but also the restoration of mobility, durability, and protective sensation. Skin grafts, representing a fundamental rung on the reconstructive ladder, offer a valuable and often indispensable method for achieving coverage. However, the judicious selection of graft thickness is a nuanced clinical decision, directly influencing graft survival (take), post-operative contraction, long-term durability, sensory recovery, and ultimate aesthetic outcome.
Epidemiologically, hand injuries are exceedingly prevalent, constituting a substantial portion of presentations to emergency departments and specialized hand clinics. Trauma, including lacerations, avulsions, degloving injuries, crush injuries, and burns, frequently results in full-thickness skin loss that cannot be addressed by primary closure. The exposure of tendons, in particular, carries significant morbidity. Tendons, being relatively hypovascular structures, are highly susceptible to desiccation, infection, adhesion formation, and necrosis when left uncovered, leading to profound functional deficits. The precise anatomical variability within the hand—from the thin, pliable dorsal skin to the thick, keratinized palmar skin—further complicates reconstructive planning. This intricate interplay of anatomy, biomechanics, and functional imperative elevates the importance of a meticulously considered approach to soft tissue reconstruction.
This academic review, aimed at orthopedic surgeons, residents, and medical students, will systematically explore the principles governing the selection and application of skin grafts for exposed tendon and hand wounds. It will delve into relevant surgical anatomy and biomechanics, delineate contemporary indications and contraindications, detail pre-operative planning and surgical techniques for various graft thicknesses, analyze potential complications and their management, and provide an overview of current literature and rehabilitation protocols. The central query guiding this discussion is: "Which thickness skin graft is best for exposed tendon and hand wounds?"—a question whose answer is complex and multifactorial.
Surgical Anatomy & Biomechanics
A comprehensive understanding of the specialized anatomy and the unique biomechanical properties of the hand's soft tissues is foundational to successful skin grafting. The hand's structural and functional demands dictate specific requirements for its integumentary cover.
Skin of the Hand
The integument of the hand is regionally specialized:
- Dorsal Skin: Characterized by its relative thinness (typically 0.5-1.0 mm), pliability, and mobility. It is loosely attached to the underlying subcutaneous tissue, facilitating full flexion of the digits and wrist. This laxity, coupled with a rich underlying vascular supply from the dorsal fascial plexus, often provides a favorable recipient bed for skin grafts. However, its delicate nature also makes it highly susceptible to injury and desiccation. Hair follicles are present, which can be a consideration for graft aesthetics.
- Palmar (Volar) Skin: In stark contrast, the palmar skin is markedly thicker (1.5-3.0 mm), highly keratinized, and designed for optimal grip and tactile discrimination. It is firmly tethered to the underlying palmar fascia and bone by numerous vertical fibrous septae, creating a stable, non-mobile, and highly durable surface that resists shear forces. This robust attachment, while functionally advantageous, means that extensive palmar skin loss often results in significant contracture if not meticulously managed. The deep dermal layer contains a high density of specialized mechanoreceptors crucial for fine touch and proprioception. Palmar skin is devoid of hair follicles and sebaceous glands but is rich in eccrine sweat glands. The thickness and fixed nature of palmar skin, along with the deep creases, can influence both graft take and the achievement of functional pliability.
Tendons of the Hand
The specific anatomy of exposed tendons is a critical determinant in graft viability:
- Extensor Tendons: Located on the dorsum of the hand and digits, these tendons are typically enveloped by a delicate, vascularized connective tissue layer known as the paratenon . The paratenon is crucial as it facilitates tendon gliding and provides a vital vascular supply. If the paratenon remains intact and viable after injury and debridement, it presents an excellent vascularized bed capable of supporting a skin graft. Extensor tendons generally exhibit less gliding excursion compared to flexors, rendering them somewhat more tolerant of minor adhesions post-grafting, provided early motion is initiated.
- Flexor Tendons: Positioned volarly, these tendons course within synovial sheaths, particularly within the digital zones (e.g., "no man's land," Zone 2). Within these sheaths, flexor tendons derive their primary nutrition from the synovial fluid and from small vascular pedicles (vincula). Critically, flexor tendons within their synovial sheaths largely lack a true paratenon . Therefore, an exposed, bare flexor tendon, stripped of its synovial covering, is essentially avascular. An avascular structure cannot support a skin graft directly, making direct grafting onto bare flexor tendons an absolute contraindication. Such scenarios necessitate the interposition of a vascularized tissue layer (e.g., local flap, muscle flap, or paratenon graft from another site) before skin graft application.
- Peritenon/Epitenon: These terms describe the connective tissue layers immediately surrounding tendons. The epitenon is the fine, outermost connective tissue sheath directly investing the tendon fibers. The paratenon , a looser areolar connective tissue, surrounds the epitenon in many locations (e.g., dorsal hand), enabling gliding and contributing significantly to the tendon's vascularity. The key for graft success is the presence of a viable, vascularized peritenon/epitenon complex. If only the glistening, white tendon fascicles are exposed (i.e., the paratenon/epitenon has been lost), the bed is avascular and unsuitable for direct grafting.
Other Structures
- Joint Capsules and Ligaments: These structures have a comparatively limited vascular supply. While they can sometimes support a graft under ideal conditions, their poor intrinsic blood flow makes them less reliable as recipient beds than muscle or healthy subcutaneous tissue. Exposed joint surfaces, especially without surrounding viable tissue, are prone to desiccation and stiffening.
- Nerves and Vessels: These vital neurovascular bundles are highly sensitive to desiccation and direct pressure. Their exposure warrants immediate and robust coverage with well-vascularized tissue, often necessitating a flap rather than a graft, to ensure their long-term viability and function.
- Bone: Bare cortical bone, stripped of its periosteum, is an avascular structure and cannot provide the necessary nutrition for a skin graft to "take." If the periosteum is intact and viable, it can serve as a suitable recipient bed.
Biomechanics and Graft Physiology
The biological integration of a skin graft, commonly referred to as "graft take," involves a complex sequence of physiological events:
1.
Imbibition (0-48 hours):
The freshly applied graft survives by passively absorbing tissue fluid, including plasma and nutrients, from the underlying wound bed. The graft appears pale and slightly edematous.
2.
Inosculation (2-5 days):
Pre-existing capillaries within the graft begin to establish rudimentary connections with new capillaries budding from the recipient bed. This marks the onset of circulatory integration.
3.
Revascularization (5-7 days and beyond):
Host capillaries actively invade the graft, establishing a more robust and organized microcirculation. The graft typically appears pink and warm. Lymphatic drainage and, much later, nerve regeneration also commence, though sensory recovery is often incomplete.
Critical Biomechanical Implications of Graft Thickness:
- Take Rate: Split-thickness skin grafts (STSGs) inherently possess a higher take rate compared to full-thickness skin grafts (FTSGs). This is due to their thinner nature, requiring less oxygen and nutrients for survival and facilitating quicker revascularization. This characteristic makes STSGs advantageous for marginal or less-than-ideally vascularized recipient beds.
- Primary Contraction: This is the immediate, elastic recoil of the graft that occurs upon harvest, primarily due to the elastic fibers within the dermal component. FTSGs, containing the entire dermis, exhibit greater primary contraction than STSGs. This must be anticipated during graft harvest by cutting the graft slightly larger than the defect.
- Secondary Contraction: This is the post-take shrinkage of the graft and the underlying wound, driven by the activity of myofibroblasts. This process typically begins weeks to months after grafting. Thinner STSGs are highly susceptible to significant secondary contraction (up to 70-90%), which can lead to severe functional contractures, particularly in areas of high joint mobility in the hand. FTSGs, due to their thicker dermal component, demonstrate substantially less secondary contraction (typically 10-30%), making them functionally superior for joints and areas requiring pliability.
- Durability and Wear Resistance: FTSGs, by virtue of their complete dermal layer, are inherently more robust, durable, and resistant to chronic friction, shear forces, and trauma than STSGs. This enhanced resilience is critically important for the functional surfaces of the hand. STSGs can be fragile, prone to blistering, and susceptible to breakdown.
- Sensation: FTSGs generally offer superior sensory recovery compared to STSGs. This is attributed to the inclusion of a greater density of nerve endings within the thicker dermis and potentially better regenerative pathways. While complete restoration of normal sensation is rare, FTSGs provide better protective sensation and a more natural feel.
- Cosmesis: FTSGs typically achieve a better color match, texture, and overall aesthetic appearance, blending more naturally with the surrounding skin and avoiding the "cobblestone" or patchy appearance often seen with meshed STSGs. STSGs tend to appear paler or shinier.
In essence, the selection of skin graft thickness for exposed tendon and hand wounds is a carefully considered surgical judgment, balancing the priority of reliable graft take against the imperative of minimizing secondary contraction, maximizing durability, optimizing sensation, and achieving an acceptable aesthetic outcome. This decision is predominantly dictated by the vascularity of the recipient bed and the specific functional demands of the hand region.
Indications & Contraindications
The strategic application of a skin graft, and the selection of its specific thickness, for an exposed tendon or hand wound is predicated on a thorough evaluation of the wound characteristics, the status of underlying vital structures, the patient's physiological context, and the long-term functional requirements of the affected hand.
General Indications for Skin Grafts in Hand Wounds
Skin grafts are primarily indicated for clean, well-vascularized wounds with full-thickness skin loss where primary closure cannot be achieved without excessive tension, or where primary closure would compromise an underlying repair (e.g., tendon repair). The overarching objectives are to provide biological closure, protect exposed vital structures from desiccation and infection, and establish a suitable surface for rehabilitation and optimal function.
- Clean, Granulating Wound Beds: Skin grafts require a healthy, vascularized recipient bed. Wounds must undergo thorough debridement, be free of active infection, and demonstrate healthy granulation tissue, viable subcutaneous tissue, muscle, or periosteum.
- Coverage of Viable Peritenon: This is a crucial indication for exposed tendons. Dorsal hand extensor tendons, if they retain an intact and viable paratenon/epitenon layer, provide an excellent vascularized bed capable of supporting both STSG and FTSG.
- Small to Moderate Skin Defects: While meshed STSGs can cover large areas, grafts are generally preferred for defects where the goal is simply protective coverage without requiring significant bulk or specialized tissue properties (e.g., highly specialized tactile sensation).
- Dorsum of the Hand: The lax, thin skin of the dorsal hand, coupled with its generally good vascular supply, makes it an ideal location for skin grafting. FTSGs are often preferred here for their superior pliability and reduced contraction, crucial for full range of motion.
- Partial-Thickness Defects with Deep Dermal Loss: After debridement of non-viable tissue, if significant dermis is lost but a healthy subcutaneous layer remains, a graft can provide definitive coverage.
- Temporary Biological Dressing: In scenarios of heavily contaminated wounds, extensive tissue loss, or uncertain wound bed viability, a meshed STSG can serve as a temporary biological dressing after initial debridement. This allows for wound surveillance, infection control, and optimization of the wound bed for subsequent definitive reconstruction (either repeat grafting or flap coverage).
Graft Thickness Selection: Specific Indications
The choice between STSG and FTSG, and even between thin, intermediate, or thick STSG, is a critical determinant of outcome.
-
Split-Thickness Skin Graft (STSG) Indications:
- Marginal Recipient Beds: When the vascularity of the recipient bed is compromised but still deemed viable (e.g., extensive subcutaneous injury, areas with less robust granulation). The higher take rate of STSGs is advantageous here.
- Large Surface Area Defects: Meshed STSGs can be expanded to cover extensive wounds, reducing the donor site requirements.
- Contaminated Wounds (Post-Debridement): As a temporary cover after thorough debridement, especially if infection risk remains a concern, allowing for drainage and easier monitoring.
- When Secondary Contraction is Acceptable or Manageable: In areas less prone to critical functional contracture or where aggressive post-operative therapy can mitigate contraction (e.g., some dorsal hand wounds).
- Over Viable Peritenon: While FTSG is often preferred, STSG can achieve take over intact peritenon, particularly if the wound size dictates a larger graft or if donor sites for FTSG are limited.
-
Full-Thickness Skin Graft (FTSG) Indications:
- Well-Vascularized, Clean Recipient Beds: FTSGs demand an impeccably healthy, granulating bed, or direct placement onto viable muscle, fat, or especially the intact, vascularized peritenon of extensor tendons.
- Minimizing Secondary Contraction: Preferred for joints (e.g., dorsal MCP, IP joints) and other areas where contracture would severely impede function. Its inherent pliability is superior.
- Maximizing Durability and Wear Resistance: Ideal for areas subjected to friction or light pressure (e.g., dorsal hand).
- Optimal Sensory Recovery: FTSGs offer the best potential for sensory return among graft options.
- Improved Cosmesis: For visible areas of the hand where color and texture match are desirable.
- Smaller to Medium-Sized Defects: Donor site limitations typically restrict FTSG use to smaller defects.
Absolute Contraindications for Skin Grafts
When the wound bed cannot support a skin graft, or when specific tissue properties are required that grafts cannot provide, alternative reconstructive methods (e.g., local, regional, or free flaps) are indicated.
- Exposed Bare Cortical Bone (without periosteum): Avascular bone cannot provide nutrition for a graft. This invariably requires a vascularized flap.
- Exposed Bare Tendon (without viable peritenon/paratenon): This is a critical contraindication, particularly for flexor tendons denuded of their synovial sheaths. An avascular tendon will necrose without vascularized coverage. A flap (or an intervening vascularized tissue layer like a paratenon graft from another site or local muscle flap) is mandatory.
- Exposed Nerves or Vessels (without surrounding vascularized tissue): These delicate structures are highly susceptible to desiccation, direct pressure, and infection. They require robust, richly vascularized coverage provided by a flap to ensure survival and function.
- Exposed Joint Capsules or Ligaments with Compromised Vascularity: While sometimes possible, a poorly vascularized capsule is a marginal bed. A flap may be required for reliable, durable coverage to prevent desiccation and preserve joint integrity.
- Active Infection or Gross Contamination: An active infection in the recipient bed is an absolute contraindication, as bacteria will lyse the graft. Thorough debridement, aggressive wound care, and systemic antibiotics are necessary to achieve a clean wound before any definitive coverage.
- Wounds Requiring Significant Bulk or Specialized Sensation/Padding: Areas such as the fingertip pulp (requiring glabrous skin, specialized sensation, and padding), or other palmar weight-bearing surfaces, are poorly suited for grafts. Flaps are superior in providing these specialized characteristics.
| Factor | Operative (Graft/Flap) Indications | Non-Operative (Conservative) Indications |
|---|---|---|
| Wound Depth | Full-thickness skin loss, exposed vital structures (tendon, bone, nerve, joint capsule). | Partial-thickness dermal loss, superficial abrasions, minor lacerations that can heal by secondary intention or primary closure without tension. |
| Wound Bed Viability |
Clean, viable granulation tissue, exposed intact peritenon, muscle, healthy fat, or intact periosteum.
Graft choice dependent on bed quality: STSG for marginal/larger areas; FTSG for robust beds, functional needs. |
Intact dermis, no exposed vital structures, minimal tissue loss. Spontaneous re-epithelialization expected. |
| Size | Small to moderate defects where primary closure is not feasible. Larger defects may necessitate meshed STSG. | Very small defects amenable to secondary intention or primary linear closure without tension. |
| Contamination | Debrided, clean wounds after thorough irrigation. Temporary meshed STSG may be used for heavily contaminated wounds post-debridement to gain initial control and allow for later definitive coverage. | Minimal contamination, able to be thoroughly irrigated and dressed. No vital structure exposure. |
| Functional Need | Restoration of protective cover, prevention of desiccation of vital structures, enabling early mobilization and rehabilitation, preventing contracture. | Minor deficits, self-healing expected, minimal impact on hand function. |
| Location | Dorsum of hand (grafts often preferred, FTSG for pliability). Palmar non-weight-bearing areas. For critical palmar/fingerpad areas or exposed bare tendons/bone, vascularized flaps are often superior to grafts. | Anywhere if superficial, small, and not involving vital structures. |
| Tendon Involvement | Exposed tendons with intact, viable peritenon (e.g., dorsal extensor tendons). For exposed bare flexor tendons (without paratenon), a vascularized flap or intervening vascularized tissue (e.g., fascial or muscle flap) is required before any skin graft. | Tendons not exposed, or only minor superficial tendon exposure with viable surrounding soft tissue that can heal secondarily. |
| Underlying Bone/Joint | Exposed bone with intact, viable periosteum; joint capsule with good surrounding vascularity (FTSG generally preferred here for durability and pliability over mobile joints). | Bare cortical bone (no periosteum), exposed avascular joint capsule or ligaments. (Requires vascularized flap coverage). |
| Required Characteristics | Protective coverage, modest durability, some sensation (better with FTSG), controlled contraction (better with FTSG). | Simple re-epithelialization. |
Pre-Operative Planning & Patient Positioning
Comprehensive pre-operative planning and appropriate patient positioning are fundamental prerequisites for achieving optimal outcomes in skin grafting for hand wounds. This meticulous preparation ensures all necessary resources are marshaled, potential challenges are anticipated, and the surgical environment is optimized for precision and efficiency.
Pre-Operative Assessment
-
Patient-Centric Evaluation:
- Medical History: A thorough review of systemic comorbidities is essential. Conditions such as diabetes mellitus, peripheral vascular disease, chronic renal failure, smoking history, chronic steroid use, and nutritional deficiencies significantly impair wound healing and graft take by compromising microcirculation and immune response. Smoking cessation programs should be rigorously implemented pre-operatively. Document all known allergies.
- Medication Review: Identify medications that may affect coagulation (e.g., anticoagulants, antiplatelets) or wound healing (e.g., immunosuppressants, methotrexate). Management plans for these medications should be coordinated with the prescribing physicians.
- Psychosocial Factors: Assess patient expectations, compliance potential for post-operative therapy, and social support systems.
-
Detailed Wound Assessment:
-
Clinical Examination:
Perform a precise assessment of the wound's exact location, dimensions (length, width, depth), and geometry. Evaluate the quality and vascularity of the exposed underlying structures:
- Tendon: Is viable paratenon present? Is the tendon bare, dry, or desiccated? Are there signs of previous infection or inflammation?
- Bone: Is periosteum intact or are there areas of bare cortical bone?
- Joint: Is the joint capsule exposed? Is it viable?
- Neurovascular Structures: Are nerves or vessels exposed or injured?
- Contamination and Infection: Actively seek signs of local infection (erythema, warmth, tenderness, purulence, foul odor). Obtain wound cultures if infection is suspected. Active infection is a contraindication to immediate grafting. A period of serial debridement, antibiotic therapy, and open wound management (e.g., with negative pressure wound therapy or moist dressings) is often required until a clean, granulating wound bed is established.
- Vascularity: Assess local tissue perfusion and overall hand vascularity.
- Associated Injuries: Meticulously rule out concomitant injuries. Plain radiographs are mandatory to exclude fractures or foreign bodies. Advanced imaging (e.g., CT, MRI, CTA) may be indicated for complex injuries or vascular concerns.
-
Clinical Examination:
Perform a precise assessment of the wound's exact location, dimensions (length, width, depth), and geometry. Evaluate the quality and vascularity of the exposed underlying structures:
-
Graft Selection and Donor Site Planning:
- Recipient Bed Confirmation: Re-evaluate the quality of the recipient bed after debridement. This dictates the definitive graft choice (STSG vs. FTSG).
- Defect Mapping: Accurately measure the defect to determine the required graft size. Consider using a sterile template (e.g., from suture packet foil or sterile paper) to precisely mark the donor site.
-
Donor Site Selection Strategy:
- Full-Thickness Skin Grafts (FTSG): Donor sites must allow for primary closure and provide skin characteristics (thickness, color, hair-bearing status) that closely match the recipient site. Common FTSG donor sites include the groin crease, volar forearm, hypothenar eminence, supraclavicular fossa, and postauricular region. Prioritize concealment of the donor scar and minimize functional deficit.
- Split-Thickness Skin Grafts (STSG): Donor sites are typically larger and can heal by re-epithelialization. Common sites include the anterolateral thigh, buttocks, scalp, or upper arm. Considerations include ease of access, color match, hair growth (avoiding excessively hairy areas if possible), and patient comfort for post-operative healing. The inner aspect of the arm or the groin crease often offers a good color match for the hand.
- Meshing Decision: Determine if the STSG will be meshed. Meshing allows for expansion, drainage of exudate, and improved take on uneven surfaces but increases secondary contraction and creates a "cobblestone" appearance. For exposed tendons, meshing can be beneficial for fluid egress.
- Photography: Pre-operative photographs are indispensable for baseline documentation, surgical planning, and post-operative outcome assessment.
- Prophylactic Antibiotics: Administer intravenous broad-spectrum antibiotics pre-operatively, especially for traumatic wounds, open injuries, or if there is a concern for contamination.
- Pain Management Planning: Establish a comprehensive post-operative pain management strategy, including multimodal analgesia (e.g., regional blocks, NSAIDs, acetaminophen, opioids) to optimize patient comfort and facilitate early rehabilitation.
Patient Positioning and Surgical Setup
-
Anesthesia:
- Regional Anesthesia: Often the preferred choice for hand surgery (e.g., axillary, supraclavicular, or interscalene brachial plexus blocks). Provides excellent intraoperative analgesia, prolonged post-operative pain relief, and sympathectomy-induced vasodilatation, which theoretically can enhance graft take.
- General Anesthesia: May be necessary for anxious patients, very prolonged procedures, or if regional anesthesia is contraindicated or technically challenging.
-
Patient Positioning:
- Supine with Arm Board: The patient is positioned supine on the operating table. The affected upper extremity is abducted and placed on a well-padded hand table or arm board. Ensure that the entire arm, including the shoulder, is freely accessible for draping and manipulation.
- Donor Site Access: Position the patient to allow convenient, simultaneous access to both the recipient hand and the chosen donor site without requiring intraoperative repositioning. This may involve slight modification of standard supine positioning (e.g., slight hip abduction for thigh harvest).
-
Pneumatic Tourniquet Application:
- A pneumatic tourniquet is applied to the upper arm (typically ipsilateral to the recipient site). A bloodless field is critical for meticulous debridement, identification of viable tissue, precise hemostasis, and accurate graft placement. The tourniquet pressure is typically set to 250-300 mmHg or 100 mmHg above systolic blood pressure.
-
Sterile Preparation and Draping:
- Thorough antiseptic preparation of both the recipient hand/arm and the selected donor site (e.g., using chlorhexidine gluconate or povidone-iodine solution). Allow sufficient drying time for the antiseptic agent.
- Sterile draping should create a wide, expansive sterile field encompassing both the recipient and donor areas, allowing ample working space for the surgical team and equipment.
-
Specialized Instrumentation and Equipment:
- Standard Hand Surgery Set: Including fine dissecting forceps (e.g., Adson with teeth), fine skin hooks, iris scissors, and delicate needle holders.
- Magnification: Surgical loupes (2.5x to 4.5x magnification) are highly recommended to facilitate precise debridement, fine suturing, and graft inspection.
- Dermatome: A powered (e.g., electric, air-driven) or manual dermatome for STSG harvest. Ensure proper functioning and availability of various blade sizes.
- Sterile Lubricant: Mineral oil or similar agent for dermatome application.
- Skin Mesher: If an STSG is to be meshed, ensure the mesher and appropriate carrier plates are sterile and available.
- Suture Material: Fine, non-absorbable monofilament sutures (e.g., 5-0, 6-0 Nylon) for graft fixation. Absorbable sutures for donor site closure (e.g., 4-0 Vicryl). Surgical staples may also be used.
- Dressing Materials: Non-adherent dressings (e.g., Xeroform, Adaptic), sterile cotton, foam, or fluffed gauze for bolster dressings, and various conforming bandages and splinting materials (e.g., plaster, thermoplastic sheets).
Detailed Surgical Approach / Technique
The execution of skin grafting for exposed tendon and hand wounds demands an exacting surgical approach, where precision in every step, from wound bed preparation to dressing application, directly correlates with graft survival and functional outcome.
I. General Principles and Recipient Wound Preparation
- Tourniquet Inflation: After draping, the pneumatic tourniquet on the upper arm is inflated to the predetermined pressure to ensure a completely bloodless field. This critical step allows for meticulous assessment and preparation without confounding hemorrhage.
-
Thorough Debridement (The Paramount Step):
- Systematically excise all non-viable, necrotic, devitalized, or contaminated tissue from the wound bed. This includes foreign bodies, crushed tissue, and desiccated margins. The goal is to create a wound that is surgically clean and consists of only healthy, bleeding tissue.
-
Specific to Exposed Tendons:
Carefully assess the tendon's surface.
- If viable paratenon/epitenon is present (e.g., common on extensor tendons of the dorsal hand), it represents an excellent vascularized bed for a graft. Preserve this layer meticulously.
- If the tendon appears bare, glistening white, or desiccated (typical for flexor tendons without their synovial sheath), it indicates the absence of a viable vascular bed. Direct skin grafting onto such a surface will inevitably fail due to lack of nutrition. In these cases, the reconstructive plan must shift to provide a vascularized tissue layer (e.g., local fascial or muscle flap, regional flap, or free flap) over the bare tendon before any subsequent skin graft could be considered as a secondary coverage.
- Ensure all underlying bone, joint capsules, nerves, and vessels are free of non-viable tissue. Bare cortical bone (without periosteum) is an absolute contraindication for grafting and must be covered by a vascularized flap.
- Hemostasis: Achieve meticulous hemostasis of the entire recipient bed using fine bipolar cautery. Even minor oozing can lead to hematoma formation beneath the graft, which will mechanically lift the graft off its bed and prevent imbibition and revascularization, thus causing graft failure. Avoid excessive cautery directly on the graft bed to preserve its vascularity. Copious irrigation with warm saline is performed.
- Final Recipient Bed Assessment: Once debrided and hemostatic, the wound bed is re-assessed for its suitability. It should be flat, moist, and uniformly vascularized. This final assessment confirms the decision regarding the appropriate graft thickness.
II. Graft Harvest and Preparation
A. Full-Thickness Skin Graft (FTSG) Technique
FTSGs are generally preferred for exposed tendons of the dorsal hand (where viable paratenon is present), areas requiring optimal pliability, durability, sensation, and minimal secondary contraction.
- Creating the Template: A sterile template of the recipient defect (e.g., using sterile aluminum foil from a suture packet or sterile Xeroform gauze) is precisely created. It should match the exact dimensions and contours of the wound.
-
Donor Site Selection and Excision:
- The template is placed over the chosen donor site (e.g., groin crease, volar forearm, hypothenar eminence, supraclavicular fossa). The site is selected for its skin characteristics (thickness, color match, minimal hair) and laxity allowing for primary closure.
- The outline is marked with a sterile pen. The graft is typically harvested slightly larger than the defect (e.g., 5-10% larger) to account for primary contraction of the elastic dermal fibers.
- Using a #15 scalpel blade, the graft is carefully incised through the full thickness of the skin into the subcutaneous fat. Elevate the graft with fine skin hooks or Adson forceps, excising it with a scalpel, ensuring an even, full-thickness harvest.
-
Defatting the Graft (Crucial Step):
- Place the harvested FTSG, dermal side up, on a sterile firm surface (e.g., tongue depressor, gloved palm).
- Using fine, sharp, curved iris scissors or a small #15 scalpel, meticulously trim away all subcutaneous fat from the undersurface of the dermis. This is a critical step; residual fat is avascular and will act as a barrier to revascularization, leading to graft failure. The graft should appear thin and translucent, allowing visualization of dermal appendages.
-
Donor Site Closure:
- Achieve meticulous hemostasis at the FTSG donor site.
- The donor wound is closed primarily. This typically involves a layer of absorbable subcutaneous sutures (e.g., 4-0 Vicryl) to reduce tension, followed by non-absorbable skin sutures (e.g., 4-0 or 5-0 Nylon) or staples. A sterile dressing is applied.
B. Split-Thickness Skin Graft (STSG) Technique
STSGs are valuable for large defects, when donor sites for FTSG are limited, or when the recipient bed's vascularity is marginal but still viable (e.g., over compromised peritenon). Intermediate to thick STSGs are generally preferred for hand wounds to offer better durability and reduced contraction compared to very thin grafts.
-
Donor Site Selection and Preparation:
- A broad, flat donor site is selected (e.g., anterolateral thigh, buttocks, upper arm).
- The donor site is prepared by applying a thin, even layer of sterile mineral oil or other lubricant to facilitate smooth and consistent dermatome movement.
-
Graft Harvest with Dermatome:
- The dermatome (manual or powered) is set to the desired thickness (e.g., 0.012-0.016 inches or 0.3-0.4 mm for intermediate to thick STSG).
- With firm, even tension applied to the skin at the donor site, the dermatome is advanced in a smooth, continuous motion to harvest a uniform sheet of skin.
-
Meshing (Optional but Common for Hand Wounds):
- The harvested STSG may be passed through a skin mesher with an appropriate ratio (e.g., 1:1.5 or 1:3).
-
Advantages of Meshing:
- Allows the graft to expand, covering larger defects with less donor site harvest.
- Creates fenestrations that facilitate drainage of seroma or hematoma from beneath the graft, which is particularly beneficial when covering an exposed tendon where fluid accumulation can compromise take.
- Improves take on uneven or irregular recipient beds.
-
Disadvantages of Meshing:
- Increases secondary contraction significantly.
- Results in a characteristic "cobblestone" appearance, which can be cosmetically less desirable.
- Leaves small, unepithelialized interstices that must heal by secondary intention, potentially increasing overall healing time and risk of infection.
- The decision to mesh is a balance of these factors, often favoring meshing for drainage over exposed tendons or in larger, potentially exudative wounds.
III. Graft Application to Recipient Site
- Placement: The prepared graft (FTSG or STSG, meshed or unmeshed) is gently transferred and carefully laid onto the meticulously prepared, hemostatic recipient bed. Ensure the dermal side of the graft is in full, uniform contact with the underlying bed. Avoid stretching or undue tension on the graft.
-
Fitting and Securing:
- Trim any excess graft material precisely to match the wound defect, ensuring no overlapping edges that can lead to epithelial bridging or poor take.
- Secure the graft margins to the wound edges using interrupted fine non-absorbable monofilament sutures (e.g., 5-0 or 6-0 Nylon) or surgical staples. Ensure sutures provide even tension, good apposition, and eliminate any wrinkles or folds in the graft.
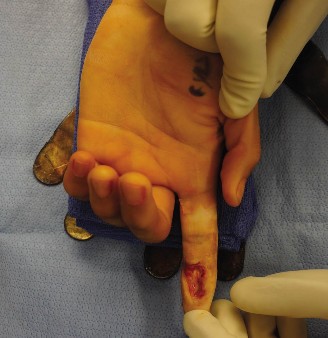
(The image above depicts the meticulous application of a skin graft to a hand wound, illustrating careful placement and secure suturing to ensure optimal contact with the vascularized recipient bed. Note the potential for fenestrations, if applicable, to prevent fluid accumulation.)
3. Fenestrations (for Non-Meshed Grafts): For unmeshed STSGs and FTSGs, several small, scattered stab incisions (fenestrations) are made into the graft using a #11 blade. These fenestrations serve as controlled exit points for any seroma or hematoma that might form beneath the graft, preventing its elevation and ensuring continuous contact with the wound bed. These are especially critical when grafting over structures like tendons, where fluid collection can impair function.
IV. Dressing and Immobilization (The "Art" of Grafting)
The immediate post-application dressing is paramount for graft survival, providing immobilization and gentle, continuous pressure.
- Non-Adherent Contact Layer: A single layer of non-adherent dressing (e.g., Xeroform gauze, Adaptic, petroleum gauze) is applied directly over the entire graft. This prevents adherence of the outer dressing materials to the delicate graft.
-
Bolster Dressing (Tie-Over or Stent Dressing):
This provides critical, continuous, gentle pressure to the graft and ensures its immobilization.
- Tie-Over Technique: The long ends of the sutures used to secure the graft periphery are left intact. A generous, soft, sterile bolster (e.g., cotton balls, foam, fluffed gauze) is meticulously molded and placed directly over the graft. The opposing long suture ends are then tied over the bolster, creating a gentle, conforming compressive force that maintains uniform graft contact with the recipient bed.
- Stent Dressing: Similar in principle, this may involve adhesive strips or additional sutures placed away from the wound edges to secure the bolster.
-
External Dressing and Hand Immobilization:
- A bulky, absorbent outer dressing (e.g., cast padding, soft gauze) is applied over the bolster.
- The hand and wrist must be completely immobilized in a functional position using a plaster or thermoplastic splint. This prevents any shear forces or movement that could disrupt the nascent revascularization of the graft.
- Functional Position: For dorsal hand grafts, the wrist is typically positioned in 20-30 degrees of extension, metacarpophalangeal (MCP) joints in 70-90 degrees of flexion, and interphalangeal (IP) joints in full extension ("intrinsic plus" or "safe" position). This position prevents collateral ligament shortening and maintains maximum functional length. For volar grafts, a position that minimizes tension on the graft while facilitating joint preservation is selected.
- Tourniquet Release: Carefully release the tourniquet. Meticulously inspect the donor site for hemostasis. Ensure there is no expanding hematoma under the recipient graft, which may necessitate immediate re-elevation and evacuation.
V. Post-Operative Care Initiation
- The extremity should be elevated continuously to minimize post-operative edema.
- The patient is monitored for signs of infection (fever, systemic inflammatory response) and hematoma formation.
- The first dressing change is typically performed on post-operative day 5-7. This is a critical juncture to assess initial graft take and address any issues.
Complications & Management
Despite meticulous surgical technique and vigilant post-operative care, skin grafting for hand wounds, particularly over exposed tendons, is not without potential complications. Early identification and prompt, appropriate management are paramount to salvaging the graft, preserving underlying structures, and optimizing long-term hand function.
Common Complications and Their Management Strategies
-
Partial or Total Graft Failure (Loss of Take): This is the most prevalent and functionally significant complication.
- Incidence: Highly variable depending on wound characteristics, patient comorbidities, and graft type. Reported rates for partial loss can range from 5-30%, with total graft loss occurring in 1-5% of cases.
- Description: Failure of the graft to revascularize, leading to necrosis and lysis. Clinically presents as a non-pink, non-blanching graft that may become dark, indurated, or soft and purulent.
-
Primary Causes:
- Hematoma or Seroma: The most common cause. Fluid accumulation beneath the graft lifts it from the recipient bed, disrupting contact and preventing imbibition and revascularization.
- Infection: Bacterial colonization, particularly by Streptococcus pyogenes or Pseudomonas aeruginosa , can rapidly cause proteolytic lysis of the graft.
- Shear Forces/Movement: Any motion or pressure that dislodges the graft or disrupts the delicate early capillary ingrowth.
- Inadequate Recipient Bed Vascularity: A poorly debrided, necrotic, or avascular bed (e.g., bare cortical bone, bare flexor tendon, irradiated tissue) cannot provide the necessary nutrients for graft survival.
- Poor Graft Handling: Trauma during harvest, defatting (especially FTSG), or placement.
-
Management Strategies:
- Hematoma/Seroma: Immediate identification (e.g., dark discoloration, tense swelling, palpable fluid under the graft) necessitates prompt intervention. Small collections may be aspirated with a fine needle. Larger or recalcitrant collections require surgical re-exploration, evacuation of the hematoma, meticulous hemostasis, and re-securing the graft with a new bolster.
- Infection: Early signs include fever, spreading erythema, purulence, foul odor. Remove the bolster, gently débride any lysed graft, obtain wound cultures, and initiate broad-spectrum intravenous antibiotics immediately. Daily dressing changes with topical antimicrobials (e.g., mafenide acetate, silver sulfadiazine, or dilute antiseptic solutions based on culture results) are crucial. Once the infection is controlled and the wound bed is healthy, re-grafting or alternative flap coverage can be considered.
- Shear: Ensure strict immobilization of the hand and wrist with a well-applied splint or cast. Reinforce or reapply the bolster dressing if movement is suspected.
- Failed Graft: If a significant portion of the graft has failed, débride the necrotic tissue, manage the wound with appropriate dressings (e.g., moist wound healing, NPWT) to promote granulation, and plan for re-grafting or, if indicated by the exposed underlying structures, local/regional/free flap coverage.
-
Infection (Recipient or Donor Site):
- Incidence: Ranges from 5-15%, with higher rates in contaminated traumatic wounds or immunocompromised patients.
- Description: Localized infection at either the graft site or the donor site. Can lead to graft lysis (recipient) or delayed healing/significant scarring (donor).
- Management Strategies: Systemic antibiotics guided by culture sensitivities. Local wound care, including debridement of purulent material. For donor sites, topical antibiotics and appropriate dressings.
-
Secondary Contraction and Contracture Formation:
- Incidence: High, particularly with thinner, meshed STSGs (can reach 70-90% contraction in highly mobile areas). FTSGs exhibit significantly less (10-30%).
- Description: Post-grafting shrinkage of the wound and graft, driven by myofibroblast activity in the wound bed. This can lead to severe restriction of joint range of motion, functional impairment, and cosmetic deformity, especially problematic over joint surfaces or dynamic areas of the hand.
-
Management Strategies:
- Prevention: The primary strategy is choosing the appropriate graft. FTSG or thicker STSG should be favored over thinner STSG in functionally critical areas. Early and aggressive post-operative hand therapy, including active and passive range of motion exercises, custom splinting (static progressive or dynamic), and scar massage, is crucial to counteract contraction forces. Silicone gel sheeting or pressure garments can also aid scar maturation and reduce contracture.
- Treatment: Established contractures often require surgical release, which may involve Z-plasties, local flap advancements, or repeat grafting (often with a FTSG or a more complex flap) to provide a larger, more pliable skin envelope.
-
Poor Sensation or Dysesthesia:
- Incidence: Common, especially with STSGs, which rarely achieve normal sensation. FTSGs generally offer better sensory potential but rarely restore completely normal sensation.
- Description: Grafted areas may exhibit diminished protective sensation (hypesthesia), altered sensation (dysesthesia), or uncomfortable hypersensitivity (hyperesthesia), impacting fine motor skills and safety awareness.
- Management Strategies: Sensory re-education therapy can improve functional interpretation of sensory input. If profound sensory loss in a critical area (e.g., pulp of a working digit) is functionally intolerable, revision with an innervated flap (e.g., neurovascular island flap) may be considered, though this is a complex decision.
-
Durability Issues / Graft Breakdown:
- Incidence: Higher with STSGs, particularly thin ones, and in areas subjected to repetitive friction, pressure, or minor trauma.
- Description: Thin STSGs can be fragile, prone to blistering, superficial abrasions, breakdown, and chronic ulceration.
- Management Strategies: Patient education on skin care, protective padding, regular moisturizing, and avoidance of trauma. For persistent or recurrent breakdown, surgical revision with a more durable FTSG or a vascularized flap may be necessary.
-
Cosmetic Dissatisfaction:
- Incidence: Subjective but frequent, especially with meshed STSGs.
- Description: Grafted areas may exhibit color mismatch (paler than surrounding skin, hyperpigmentation), a "cobblestone" texture (from meshed grafts), or hypertrophic scarring.
- Management Strategies: Sun protection (SPF 30+) for grafted skin, scar massage, silicone sheeting or gel, and corticosteroid injections for hypertrophic scars. Laser therapy can address dyspigmentation or textural irregularities. Surgical revision (e.g., re-grafting with an FTSG for better match, scar revision) may be an option for severe cases.
-
Donor Site Morbidity:
- Incidence: Virtually 100% of patients will experience some degree of donor site pain and scarring.
-
Description:
- STSG Donor Sites: Often painful (sometimes more so than the graft site), risk of infection, delayed healing, hypertrophic scarring, and permanent pigmentary changes (hypo- or hyperpigmentation).
- FTSG Donor Sites: Result in a linear scar due to primary closure, with potential for infection, dehiscence, or hypertrophic scarring.
- Management Strategies: Adequate analgesia, meticulous donor site care (e.g., occlusive dressings, hydrocolloids, or specialized polyurethane films for STSG donor sites), infection prevention and treatment, and scar management techniques (silicone, massage).
| Complication | Incidence (Approximate) | Description | Management Strategies |
|---|---|---|---|
| Graft Failure (Partial/Total) | 5-30% partial, 1-5% total | Lack of revascularization leading to necrosis and lysis of the graft. Often due to hematoma, infection, shear, or poor bed. | Hematoma/Seroma: Aspiration or surgical evacuation. Infection: Systemic antibiotics, topical antimicrobials, débridement of lysed graft, re-grafting once wound is clean. Shear: Strict immobilization, bolster reapplication. Poor Bed: Débridement, wound bed optimization, re-grafting or flap. |
| Infection (Recipient Site) | 5-15% | Bacterial colonization of the graft, leading to lysis and potential systemic sepsis. | Obtain cultures, systemic antibiotics, local wound care (débridement, topical agents), manage as open wound until clean for re-grafting. |
| Secondary Contraction | High (STSG > FTSG) | Post-take shrinkage of the graft and wound, causing joint contractures and functional limitation. Most severe with thin, meshed STSG. | Prevention: FTSG/thicker STSG when possible, early and aggressive hand therapy, continuous splinting (static progressive/dynamic), scar massage, silicone sheeting. Treatment: Surgical release (Z-plasty, flap), repeat grafting. |
| Poor Sensation / Dysesthesia | Common (STSG > FTSG) | Diminished, altered, or painful sensation in the grafted area. | Sensory re-education therapy. Consider innervated flap for critical functional areas if severe and intolerable. |
| Durability Issues | Higher with STSG | Fragility, blistering, breakdown, chronic ulceration, particularly in high-wear areas. | Protective padding, moisturizers. Surgical revision with more durable FTSG or flap for persistent issues. |
| Cosmetic Dissatisfaction | Subjective, but frequent | Color mismatch (pale, hyperpigmented), "cobblestone" appearance (meshed STSG), hypertrophic scarring. | Sun protection, scar massage, silicone sheeting, laser therapy. Surgical revision for severe cases. |
| Donor Site Morbidity | Nearly 100% | Pain, scarring, infection, delayed healing, dyspigmentation, hair growth changes. | Adequate analgesia, meticulous donor site care (e.g., occlusive dressings), infection management, scar management. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an indispensable component of successful skin graft management in the hand, directly influencing functional outcomes, minimizing complications like contracture, and optimizing patient reintegration. Protocols must be tailored to the specific graft thickness, location, underlying repairs (e.g., tendon repair vs. simple coverage), and individual patient factors.
Phase I: Immobilization and Graft Protection (Day 0 to Post-Op Day 5-7)
The primary objective during this initial phase is absolute immobilization of the graft to ensure successful take, preventing any shear forces or disruption of the delicate revascularization process.
-
Immobilization:
- Maintain the hand and wrist in the pre-determined functional position (e.g., intrinsic plus position for dorsal grafts, mild wrist extension for palmar grafts) via a well-padded plaster or thermoplastic splint. The splint must protect both the graft and its bolster.
- Patient education on avoiding any movement of the splinted area is crucial.
- Elevation: Continuous elevation of the entire extremity (above heart level) is mandatory to minimize edema, which can compromise graft vascularity and take. This often involves sling use during ambulation and elevation on pillows while resting/sleeping.
-
Wound and Donor Site Monitoring:
- Monitor the overall condition of the hand and extremity for signs of excessive swelling, neurovascular compromise (e.g., sensory changes, pallor, coldness), or systemic infection.
- Monitor the dressing for signs of exudate, bleeding, or dislodgement.
- Donor site care: For STSG donor sites, ensure the dressing remains intact and dry. For FTSG donor sites, monitor the incision for dehiscence or infection.
- Pain Management: Implement a robust multimodal pain management regimen to ensure patient comfort, which indirectly aids graft take by reducing patient-induced movement.
-
First Dressing Change (Post-Op Day 5-7):
This is a critical evaluation point.
- Performed by the surgeon or experienced hand therapist.
- Gently remove the outer dressing and bolster.
- Carefully inspect the graft for signs of take (pink color, blanching, adherence) versus failure (dark discoloration, non-adherence, purulence).
- Address any hematoma or seroma by gentle massage or aspiration if necessary.
- Cleanse the wound gently. Reapply non-adherent dressing, light gauze, and a new splint.
- If graft take is good, a transition to Phase II can begin. If significant graft loss, plan for re-grafting or alternative coverage.
Phase II: Early Mobilization and Scar Management (Post-Op Day 7 to Weeks 3-4)
Once graft take is confirmed, the focus shifts to carefully initiated mobilization to prevent joint stiffness and adhesion formation, coupled with early scar management.
-
Controlled Active Range of Motion (AROM):
- Begin gentle, controlled active range of motion exercises for non-grafted joints and for joints adjacent to the graft that are not directly under tension.
- For areas directly underlying the graft, passive range of motion (PROM) may be initiated carefully by the therapist, ensuring no excessive tension or shear on the graft. The specific timeline (e.g., 7-10 days for extensor grafts, 10-14 days for more fragile areas) is surgeon-dependent.
- Specific to Tendons: If an exposed extensor tendon with intact paratenon was grafted, early, protected active motion can commence as soon as graft take is secure (often around day 7-10) to minimize adhesions. The range and force of motion must be strictly controlled by the hand therapist.
- Edema Control: Continue elevation, consider gentle compression gloves (after suture removal) if swelling persists, and initiate gentle retrograde massage.
-
Scar Management:
- Initiate gentle scar massage once the wound is completely dry and stable (typically after 2-3 weeks).
- Introduce silicone gel sheeting or topical silicone products to aid scar maturation and reduce hypertrophy.
- Patient education on proper skin care, moisturizing, and sun protection (grafted skin is sensitive to UV).
-
Splinting:
- Static splinting continues to be used at rest or overnight to maintain gains in ROM and prevent contracture.
- Consider dynamic or static progressive splinting if early signs of joint stiffness or contracture are noted.
- Progressive Strengthening: Light grip strengthening and fine motor activities can be introduced carefully, ensuring no undue stress on the graft.
Phase III: Progressive Strengthening and Functional Reintegration (Weeks 4-6 onwards)
This phase aims for increasing functional demands, achieving full range of motion, and maximizing strength and dexterity.
- Progressive Range of Motion: Continue and advance AROM and PROM exercises, gradually increasing the intensity and complexity. The goal is to achieve maximal functional ROM.
- Strengthening: Gradually introduce progressive resistance exercises, focusing on grip strength, pinch strength, and endurance.
- Sensory Re-education: For FTSGs, which have better sensory potential, sensory re-education programs (e.g., desensitization, texture discrimination) can be initiated to improve tactile perception.
- Work/Activity Simulation: Tailored activities mimicking daily living, work, or sport-specific demands are introduced to facilitate full functional reintegration.
- Splinting: May be continued as needed to address residual contracture or maintain achieved ROM. Custom fabrication may be required.
- Scar Management: Continues with emphasis on softening, flattening, and desensitizing the scar. Pressure garments may be necessary for severe scarring.
Important Considerations Across Phases
- Hand Therapist Collaboration: Close collaboration between the surgeon and a certified hand therapist (CHT) is paramount. The CHT is uniquely qualified to assess progress, modify protocols, and address emerging issues.
- Patient Compliance: Emphasize the critical role of patient adherence to home exercise programs and splinting regimens for optimal outcomes.
- Long-Term Follow-Up: Grafted skin, especially STSG, requires long-term monitoring for changes in texture, pigmentation, durability, and sensation. Secondary contractures can develop over months.
- Psychological Support: Hand injuries can have significant psychological impact. Providing support and realistic expectations is crucial.
Summary of Key Literature / Guidelines
The body of literature on skin grafting for hand wounds is extensive, reflecting decades of clinical experience and research. While there are no universal, single-source guidelines, a consensus of best practices has emerged, particularly concerning graft thickness over exposed tendons.
-
"Golden Rule" for Tendon Coverage: The most fundamental principle in hand reconstruction is that bare tendon (without viable paratenon/epitenon) , especially flexor tendon, will not support a skin graft. This is consistently emphasized across surgical texts and academic reviews. The reason is the avascular nature of exposed tendon fascicles, which are prone to desiccation and necrosis without a vascularized covering. When bare tendon is exposed, a vascularized flap (local, regional, or free) is mandatory before any form of skin grafting can be contemplated. If only peritenon is exposed, then a graft can be considered.
-
Full-Thickness Skin Grafts (FTSG) for Functional Areas:
- Numerous studies and expert opinions advocate for the use of FTSGs in functionally critical areas of the hand, particularly over the dorsum of joints (e.g., MCP, IP joints) and where viable extensor tendon peritenon is present.
- The primary rationale, as highlighted by authors like Brand and Hollister (1999) and Grabb's Encyclopedia of Flaps (2009) , is the superior resistance to secondary contraction, enhanced durability, better pliability, improved cosmetic match (color and texture), and greater potential for sensory recovery offered by FTSGs. This allows for earlier and more aggressive rehabilitation, critical for preserving joint motion.
- Donor sites for FTSGs are limited but well-described, typically from the groin, volar forearm, or hypothenar eminence, chosen for their laxity and skin characteristics.
-
Split-Thickness Skin Grafts (STSG) for Large or Compromised Wounds:
- STSGs remain invaluable, particularly for covering large surface area defects, especially when FTSG donor sites are insufficient or when the recipient bed is considered marginal (but still viable).
- Thinner STSGs (0.008-0.012 inches) offer the highest take rate but suffer from maximal secondary contraction, fragility, and poor long-term durability and sensation. They are rarely the first choice for definitive coverage over mobile parts of the hand or exposed tendons.
- Intermediate to Thick STSGs (0.012-0.018 inches) offer a compromise, providing a reasonable take rate with somewhat improved durability and reduced contraction compared to very thin grafts. They are often utilized over viable peritenon on the dorsum of the hand when a large area needs to be covered.
- The use of meshed STSGs , while allowing for expansion and drainage, is known to increase secondary contraction and result in a less aesthetic "cobblestone" appearance. This trade-off is accepted in cases of very large defects or potentially exudative beds.
-
The Role of Wound Bed Preparation:
- The literature consistently emphasizes that meticulous wound debridement and absolute hemostasis are the most critical factors influencing graft take, regardless of thickness. Studies by authors such as Knighton (1969) and contemporary hand surgery texts underscore that even the best graft will fail on a non-viable or hemorrhagic bed.
- The concept of a "clean, granulating wound" or a bed with viable fat, muscle, or peritenon is a recurring prerequisite.
-
Rehabilitation and Long-Term Outcomes:
- Modern hand surgery protocols stress the importance of early, protected motion initiated by certified hand therapists, particularly for grafts overlying dynamic structures. Prolonged immobilization, while initially necessary, contributes to stiffness and adhesion formation.
- Long-term follow-up studies, such as those reviewed by Mackinnon and Dellon (1985) regarding sensation, indicate that while some sensory return is possible, complete restoration of normal discriminative sensation is rare, especially with STSGs.
- The need for continuous scar management (massage, silicone, splinting) for months to years post-grafting is also well-documented to mitigate contracture and improve pliability.
In summary, the academic literature and established clinical guidelines strongly support a differentiated approach: FTSGs are generally preferred for exposed, viable peritenon-covered extensor tendons and other critical functional areas of the hand due to their superior biomechanical properties and functional outcomes. STSGs, particularly intermediate to thick varieties, are reserved for larger defects or less ideal recipient beds where maximizing take rate is paramount, acknowledging their limitations regarding contraction and durability. Crucially, direct grafting onto bare, avascular tendon or bone is contraindicated, necessitating a vascularized flap.
