Female Athlete Triad: Diagnosing Critical Sports Medicine Cases
Key Takeaway
Here are the crucial details you must know about Female Athlete Triad: Diagnosing Critical Sports Medicine Cases. ACL graft failure in younger individuals often relates to allograft use, inadequate fixation, or poor tunnel placement, frequently requiring revision. Staged revisions are indicated for wide or compromised tunnels. Graft impingement, causing limited extension, typically results from an anterior tibial tunnel. For female athletes, considering factors like the female athlete triad is crucial for comprehensive injury prevention and recovery.
Female Athlete Triad and Critical Sports Medicine Cases: The Complex Revision Anterior Cruciate Ligament Reconstruction
While the Female Athlete Triad—comprising low energy availability, menstrual dysfunction, and altered bone mineral density—remains a critical diagnostic pillar in sports medicine, our Grand Rounds today pivots to another exceedingly common, highly complex, and biologically demanding scenario in the young athletic population: the failed Anterior Cruciate Ligament (ACL) reconstruction. The intersection of biomechanics, graft biology, and surgical precision makes revision ACL reconstruction one of the most challenging procedures in orthopedic sports medicine.
Today, we will dissect a highly instructive case of a 24-year-old male ultimate frisbee player who presents with a reinjury to his previously reconstructed left knee. By analyzing the biological failures of allografts in young athletes, the biomechanical consequences of tunnel malposition, and the intricate decision-making algorithms for single-stage versus two-stage revisions, we will establish a comprehensive framework for managing critical sports medicine failures.
Detailed Patient Presentation and Mechanism of Injury
History of Present Illness and Index Procedure
The patient is a 24-year-old highly active male, participating competitively in ultimate frisbee, who presents to the orthopedic clinic with a chief complaint of left knee instability, massive effusion, and an inability to bear weight following an acute deceleration injury. He reports that four years prior, at the age of 20, he sustained a primary rupture of his left ACL while playing basketball. At that time, he underwent an arthroscopically assisted single-bundle ACL reconstruction utilizing a fresh-frozen bone-patellar tendon-bone (BPTB) allograft.
His postoperative recovery following the index procedure was reportedly uneventful. He achieved full range of motion, completed a standard rehabilitation protocol, and was cleared to return to competitive sports at eight months post-surgery. He functioned well until the current injury, which occurred when he planted his left foot to rapidly change direction during a frisbee match. He felt an audible and palpable "pop," followed by immediate hemarthrosis and a sensation that the knee had "given way."
Biological Considerations of Graft Choice
The selection of an allograft for a 20-year-old pivoting athlete represents a critical error in primary surgical decision-making and is the single greatest contributing factor to his current graft failure. The literature, particularly the Multicenter ACL Revision Study (MARS) cohort, has unequivocally demonstrated that in young, active patients (under the age of 25), allograft ACL reconstructions yield a significantly higher failure rate—often cited as three to four times higher—compared to autograft reconstructions.
This discrepancy is rooted in the biology of graft incorporation. Ligamentization of a graft undergoes four distinct phases: necrosis, revascularization, cellular proliferation, and remodeling. Allografts experience a markedly delayed and often incomplete creeping substitution compared to autografts. Furthermore, the sterilization processes required for allografts, particularly terminal gamma irradiation greater than 2.0 to 2.5 Mrad, severely compromise the collagen cross-linking and structural integrity of the graft. Consequently, the graft remains biologically immature and mechanically inferior for a much longer duration, rendering it highly susceptible to catastrophic failure under the extreme shear and rotational loads of cutting sports.
Comprehensive Clinical Examination Findings
Acute Assessment and Joint Effusion
Upon examination, the patient presents with a moderate antalgic gait, heavily favoring the contralateral limb, and demonstrates a classic quadriceps avoidance pattern. Inspection of the left knee reveals a massive, tense effusion, indicative of acute hemarthrosis. There are well-healed arthroscopy portal scars and a small proximal tibial incision from the previous allograft fixation. The skin is intact with no signs of erythema or localized infection.
Palpation reveals diffuse tenderness, most notably along the lateral joint line, raising a high clinical suspicion for a concomitant lateral meniscal tear—a frequent passenger in both primary and revision ACL injuries. The extensor mechanism is intact, with no palpable defects in the patellar or quadriceps tendons. The patient lacks 10 degrees of terminal extension and can flex to 110 degrees, limited primarily by the tense effusion and mechanical guarding.
Ligamentous and Rotatory Instability Testing
Ligamentous examination is paramount in defining the extent of the structural failure. The Lachman test is grossly positive, demonstrating a Grade III (>10mm) anterior translation with a soft, indistinct endpoint, confirming the catastrophic failure of the ACL graft. The anterior drawer test in 90 degrees of flexion is similarly positive.
Crucially, the pivot shift test—the most specific clinical maneuver for assessing functional rotatory instability—elicits a high-grade, explosive subluxation and reduction of the lateral tibial plateau (Grade III). This profound rotatory laxity not only confirms the graft rupture but also suggests that the index femoral tunnel may have been placed in a non-anatomic, highly vertical position, failing to control the pivot shift even prior to the acute rupture. Careful assessment of the secondary stabilizers reveals a negative dial test at both 30 and 90 degrees, ruling out posterolateral corner (PLC) involvement, and stable varus and valgus stress testing at 0 and 30 degrees, indicating intact collateral ligaments.
Advanced Imaging and Diagnostics
Radiographic Evaluation and Tunnel Analysis
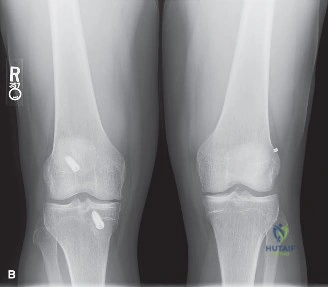
Standard weight-bearing radiographs, including anteroposterior (AP), lateral, Rosenberg (45-degree flexion posteroanterior), and Merchant views, are mandatory to assess bone stock, hardware placement, tunnel widening, and early degenerative changes.

The AP radiograph reveals metallic interference screws in both the femur and tibia. The tibial tunnel aperture appears significantly widened. Tunnel widening, or osteolysis, is a frequent complication in allograft reconstructions, driven by a combination of mechanical micromotion (the "bungee cord" and "windshield wiper" effects) and biological factors, including an exaggerated local cytokine response to the foreign allograft tissue and synovial fluid ingress into the bone tunnels.
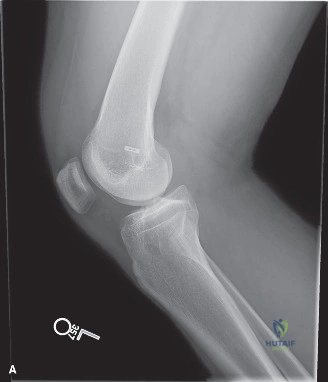
The lateral radiograph is critical for evaluating tunnel position. In this patient, the femoral tunnel is noted to be placed anteriorly and vertically, high within the intercondylar notch. This vertical placement is a classic hallmark of the traditional transtibial drilling technique, which often sacrifices anatomic femoral footprint placement to accommodate the rigid trajectory of the tibial tunnel. A vertical graft effectively resists anterior tibial translation (Lachman) but fails utterly to resist internal rotation, leaving the patient with persistent rotatory instability (positive pivot shift).
Advanced Cross-Sectional Imaging
Magnetic Resonance Imaging (MRI) is obtained to evaluate the soft tissues. The MRI confirms a complete mid-substance discontinuity of the ACL allograft. Furthermore, a complex tear of the posterior horn of the medial meniscus is identified. While meniscal pathology carries a worse prognosis for long-term joint preservation and accelerates osteoarthritic changes, it is not intrinsically associated with the biomechanical re-tear of the ACL graft itself.
A fine-cut Computed Tomography (CT) scan with 3D reconstructions is the gold standard for preoperative planning in the revision setting. The CT scan precisely quantifies the degree of tunnel widening and the exact spatial orientation of the existing tunnels. In this patient, the CT demonstrates a tibial tunnel that has expanded to 16 millimeters in diameter, with significant cystic changes extending into the metaphyseal bone.
Exhaustive Differential Diagnosis
When evaluating a failed ACL reconstruction, the differential diagnosis must categorize the etiology of failure to prevent repeating the same catastrophic outcome. The failure modes are broadly divided into biological failures, surgical technique errors, traumatic reinjuries, and missed concomitant instabilities.
| Etiology of Graft Failure | Clinical Presentation | Imaging Findings | Pathophysiology & Key Drivers |
|---|---|---|---|
| Biological Failure (Allograft) | Insidious onset of laxity or low-energy failure in a young athlete. | Graft attenuation or absence on MRI, frequently accompanied by severe tunnel widening. | Delayed incorporation, altered collagen cross-linking from irradiation, and immune-mediated osteolysis. |
| Surgical Error: Tunnel Malposition | Persistent rotatory instability (vertical tunnel) or loss of extension (anterior tibial tunnel). | Lateral X-ray shows femoral tunnel anterior to the anatomic footprint or tibial tunnel anterior to Blumensaat's line. | Non-anatomic biomechanics. Vertical grafts cannot resist the pivot shift. Anterior tibial tunnels cause roof impingement. |
| Traumatic Re-rupture | Acute high-energy pivoting event with a distinct "pop" and immediate hemarthrosis. | Acute mid-substance tear on MRI, often with new bone bruising patterns (e.g., lateral femoral condyle). | Force applied to the knee exceeded the ultimate tensile strength of a biologically incorporated and anatomically placed graft. |
| Missed Concomitant Instability | Chronic varus thrust, recurvatum, or persistent posterior sag despite ACL reconstruction. | MRI shows PLC injury, high-grade MCL tear, or unrecognized meniscal root avulsion. | Failure to address secondary stabilizers places excessive, unphysiologic strain on the ACL graft, leading to gradual elongation and rupture. |
Complex Surgical Decision Making and Classifications
Determining the Primary Cause of Failure
As established, the choice of allograft in a 24-year-old pivoting athlete made the greatest contribution to his graft failure. While inadequate graft fixation can result in residual laxity, and non-anatomic tunnel position fails to restore normal knee kinematics, the profound biological inferiority of the allograft in this specific demographic supersedes these factors. Meniscal pathology, while detrimental to the articular cartilage, does not cause graft rupture.
Single-Stage versus Two-Stage Revision
After careful consideration, the patient elects to proceed with a revision ACL reconstruction. The surgeon is now faced with a critical bifurcation in the treatment algorithm: perform a single-stage revision or a two-step, staged revision.
A single-stage revision involves hardware removal, tunnel debridement, and immediate placement of a new graft. This is viable when existing tunnels are anatomically placed and not significantly widened, or when they are entirely non-anatomic and can be completely bypassed by new, anatomically drilled tunnels.
A staged revision involves an initial surgery to remove hardware, debride the fibrous tissue within the tunnels, and pack the defects with bone graft (autologous iliac crest or allograft dowels). The patient then waits 4 to 6 months for bone consolidation before returning for the definitive ACL reconstruction.
The finding that most strongly favors a staged revision is tunnel widening. When existing tunnels expand beyond 14 millimeters, as seen in our patient's CT scan, achieving reliable, rigid aperture fixation of the new graft becomes biomechanically impossible. The lack of circumferential bone-to-graft contact severely impairs biological incorporation. Vertical femoral tunnel placement, retained hardware, prior autograft use, and meniscal tears do not inherently mandate a staged approach, as these can typically be addressed or bypassed in a single surgical setting.
Addressing Graft Impingement
A critical concept in both primary and revision ACL surgery is the avoidance of notch impingement. If, during the trial phase of the revision reconstruction, the surgeon notes that the graft impinges against the roof of the intercondylar notch as the knee is brought into terminal extension (leaving the knee 5 degrees short of straight), the primary culprit is an anteriorly placed tibial tunnel.
If the tibial tunnel is drilled too anteriorly, the graft will intersect Blumensaat's line before the knee reaches full extension. This "roof impingement" acts as a mechanical block to extension and causes repetitive abrasion of the graft, ultimately leading to graft attrition, localized synovitis, and the formation of a fibrotic "Cyclops lesion." To correct this, the surgeon must either dilate the posterior aspect of the tibial tunnel or perform a notchplasty to accommodate the graft, though anatomic tunnel placement remains the definitive solution.
Step-by-Step Surgical Technique and Intervention
Stage One: Hardware Removal and Bone Grafting
Given the severe 16-millimeter tunnel osteolysis, a two-stage approach is mandated. The patient is brought to the operating room, placed supine, and a tourniquet is applied to the proximal thigh. Standard anterolateral and anteromedial portals are established.
Diagnostic arthroscopy confirms the absence of a functional ACL and identifies the medial meniscal tear, which is addressed with an all-inside meniscal repair technique to preserve joint biomechanics. The remnants of the allograft are aggressively debrided using a motorized shaver and radiofrequency ablation.
Attention is then turned to hardware removal. A guide pin is placed through the existing tibial interference screw, and a cannulated screwdriver is used to extract it. If the screw is stripped or heavily integrated, a trephine reamer or osteotome may be required. The fibrous tissue lining the widened tibial and femoral tunnels is meticulously curetted to expose healthy, bleeding cancellous bone.
Cylindrical allograft bone dowels, matched to the diameter of the widened tunnels (e.g., 16mm for the tibia), are impacted into the defects. Platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) may be added to augment the biological healing response. The wounds are closed, and the patient is allowed to heal for 5 months.
Stage Two: Definitive Revision Reconstruction
Following CT confirmation of complete bone graft incorporation, the patient returns for the definitive reconstruction. The graft of choice for this demanding revision is a robust autograft. Given the virgin status of his extensor mechanism, a bone-patellar tendon-bone (BPTB) autograft or a thick quadriceps tendon autograft is selected.
The surgeon utilizes an independent drilling technique (anteromedial portal or outside-in technique) to ensure anatomic placement of the femoral tunnel, completely independent of the tibial tunnel trajectory. The anatomic femoral footprint is identified behind the resident's ridge (lateral intercondylar ridge).
The tibial tunnel is drilled meticulously, ensuring the intra-articular aperture is positioned posterior to the anterior horn of the lateral meniscus and safely posterior to Blumensaat's line in full extension to prevent the aforementioned roof impingement. The autograft is passed, and rigid fixation is achieved. Aperture fixation with titanium or bio-composite interference screws is utilized, ensuring maximal graft tensioning at 20 degrees of flexion.
Strict Post-Operative Protocol and Rehabilitation Stages
The rehabilitation following a revision ACL reconstruction is inherently more conservative than a primary reconstruction, acknowledging the compromised biological milieu and the necessity of protecting the newly integrating autograft.
Phase 1: Maximum Protection (Weeks 0-6)
The patient is placed in a hinged knee brace locked in full extension during ambulation. Weight-bearing is restricted to toe-touch (20% body weight) for the first 4 weeks to protect both the meniscal repair and the intra-articular graft fixation. Early emphasis is placed on achieving full, symmetric terminal extension to prevent arthrofibrosis and Cyclops lesion formation. Patellar mobilization is critical to prevent infrapatellar contracture syndrome.
Phase 2: Controlled Ambulation and Muscle Activation (Weeks 6-12)
The brace is unlocked as quadriceps control returns. The patient transitions to full weight-bearing. Closed kinetic chain exercises (e.g., mini-squats, leg presses) are initiated, keeping flexion angles between 0 and 60 degrees to minimize anterior shear forces on the tibia. Open kinetic chain hamstring exercises are permitted, but open chain quadriceps extensions are strictly avoided to prevent excessive graft strain.
Phase 3: Advanced Strengthening and Proprioception (Months 3-6)
Rehabilitation shifts toward functional strengthening, neuromuscular control, and proprioceptive training. Perturbation training and balance board exercises are integrated. Isokinetic testing is utilized to objectively measure quadriceps and hamstring strength deficits compared to the contralateral limb.
Phase 4: Return to Sport Progression (Months 6-12+)
Return to sport is not dictated by time, but by strict, objective functional criteria. The patient must demonstrate greater than 90% limb symmetry index (LSI) on isokinetic testing, pass a battery of functional hop tests (single hop, triple hop, crossover hop), and exhibit no effusion or clinical laxity. Psychological readiness is assessed using the ACL-RSI scale. Given the revision setting, return to competitive ultimate frisbee is typically delayed until 10 to 12 months postoperatively to ensure maximal biological ligamentization.
High-Yield Clinical Pearls and Pitfalls
- The Allograft Trap: Never utilize an allograft for primary ACL reconstruction in an athlete under the age of 25 participating in cutting/pivoting sports. The biological failure rate is unacceptably high. Autograft (BPTB, Hamstring, or Quadriceps) is the gold standard.
- Tunnel Widening Dictates Staging: Always obtain a preoperative CT scan in the revision setting. If tunnel osteolysis exceeds 14 millimeters, attempting a single-stage revision risks catastrophic loss of fixation. Respect the biology and perform a two-stage procedure with bone grafting.
- The Vertical Tunnel Fallacy: A vertical femoral tunnel (often resulting from transtibial drilling) may yield a negative Lachman test but will leave the patient with a persistent pivot shift. Anatomic, independent drilling is crucial for restoring rotatory stability.
- Beware Roof Impingement: If the knee lacks terminal extension intraoperatively after graft fixation, immediately suspect an anteriorly placed tibial tunnel. The graft is impinging on the intercondylar roof. This must be recognized and corrected before leaving the operating room to prevent the inevitable formation of a Cyclops lesion and chronic pain.
- Assess the Periphery: A failed ACL reconstruction is rarely an isolated event. Meticulously evaluate the posterolateral corner, the medial collateral ligament, and the meniscal roots. Unaddressed secondary restraints will rapidly doom the most perfectly placed revision ACL graft.
