INTRODUCTION TO FRACTURE HEALING AND BONE REGENERATION
Bone is a highly specialized connective tissue unique in its ability to undergo true regeneration rather than mere repair. Unlike other tissues that heal via fibrous scar formation, bone restores its original histological structure and mechanical properties—a process termed restitutio ad integrum. The multitude of factors governing fracture healing have been the subject of exhaustive clinical, biomechanical, and laboratory investigations. While our understanding of the cellular and molecular pathways has expanded exponentially, the orchestration of these events remains one of the most complex phenomena in orthopaedic surgery.
Fracture healing must be conceptualized through multiple lenses: biological, biochemical, mechanical, and clinical. It requires the precise spatial and temporal recruitment of specific cell lineages (fibroblasts, macrophages, chondroblasts, osteoblasts, and osteoclasts) and the subsequent expression of genes controlling matrix production, growth factors, and transcription factors.
For the practicing orthopaedic surgeon, mastering these principles is not merely an academic exercise; it is the foundation of surgical decision-making. Whether selecting between absolute stability (compression plating) or relative stability (intramedullary nailing), the surgeon is actively manipulating the biological environment to dictate the pathway of bone regeneration.
Clinical Pearl: Clinical union precedes radiographic union. Clinical union is achieved when the progressively increasing stiffness and strength of the mineralizing callus make the fracture site stable and pain-free upon physiological loading. Radiographic union is confirmed when plain radiographs demonstrate mature bone trabeculae or cortical bone bridging the fracture site across at least three out of four cortices.
THE TRIPHASIC BIOLOGICAL CASCADE OF FRACTURE HEALING
A fracture initiates a continuous, overlapping sequence of inflammation, repair, and remodeling. If this interdependent cascade proceeds undisturbed, the injured bone is restored to its pre-injury state.
1. The Inflammatory Phase (Days 0 to 7)
Immediately following the mechanical disruption of bone, the rupture of endosteal, periosteal, and surrounding soft-tissue vessels results in hematoma formation. This fracture hematoma is not merely a passive blood clot; it is a highly active biological crucible.
Inflammatory cells (neutrophils, macrophages) invade the hematoma, initiating the lysosomal degradation of necrotic tissue. As Bolander and others have demonstrated, the degranulating platelets and macrophages within the hematoma release a potent cocktail of signaling molecules, most notably Transforming Growth Factor-beta (TGF-β) and Platelet-Derived Growth Factor (PDGF). These cytokines initiate the chemotaxis and proliferation of mesenchymal stem cells, setting the stage for the reparative phase.
2. The Reparative Phase (Days 5 to Months)
Usually beginning 4 to 5 days post-injury, the reparative phase is characterized by the invasion of pluripotential mesenchymal cells. Depending on the mechanical environment and oxygen tension, these cells differentiate into fibroblasts, chondroblasts, or osteoblasts, forming a soft fracture callus.
A critical prerequisite for this phase is angiogenesis. The proliferation of new blood vessels within the periosteal tissues and marrow space routes appropriate progenitor cells to the fracture site, forming a bed of granulation tissue. As chondrocytes undergo hypertrophy and apoptosis, the cartilaginous matrix calcifies. Osteoblasts then lay down woven bone upon this calcified cartilage scaffold via endochondral ossification, transitioning the soft callus into a hard callus.
3. The Remodeling Phase (Months to Years)
The transition of the fracture callus to woven bone and its subsequent mineralization signals the onset of the remodeling phase. This phase is governed by Wolff’s Law, which dictates that bone adapts to the mechanical loads placed upon it.
During remodeling, the disorganized, mechanically inferior woven bone is systematically replaced by highly organized lamellar bone through the action of osteoclast cutting cones and trailing osteoblasts (creeping substitution). The medullary canal is restored, and the bone's morphology and mechanical strength return to normal. Radioisotope studies demonstrate increased metabolic activity at fracture sites long after painless function is restored, indicating that remodeling continues for years.
ANATOMICAL ZONES OF FRACTURE REPAIR
Einhorn elegantly described four distinct healing responses based on anatomical location: the bone marrow, cortex, periosteum, and external soft tissues.
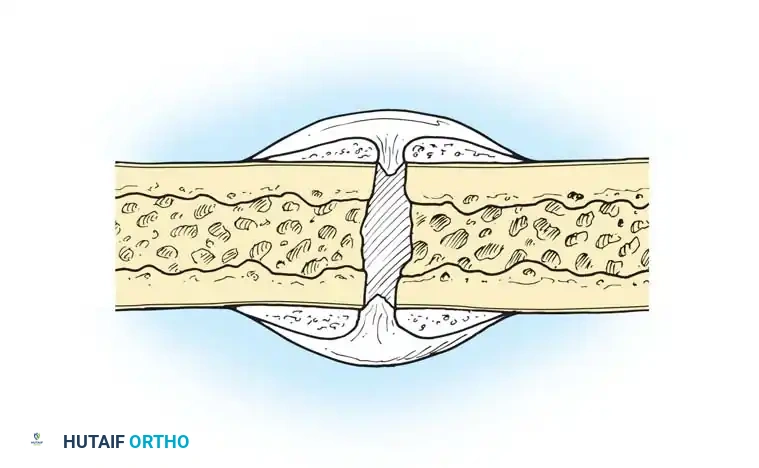
The four anatomical compartments contributing to fracture healing: External soft tissue, Periosteum, Cortex, and Bone marrow.
Of these, the periosteal response is arguably the most critical for secondary fracture healing. The periosteum contains an inner cambium layer rich in committed osteoprogenitor cells and an outer fibrous layer containing uncommitted mesenchymal cells. Following injury, these cells recapitulate embryonic intramembranous ossification (forming bone directly without a cartilage intermediate) adjacent to the fracture ends, and endochondral bone formation (via a cartilage intermediate) in the central fracture gap.
Surgical Warning: The periosteal response is highly sensitive to surgical technique. Aggressive periosteal stripping during open reduction internal fixation (ORIF) devascularizes the cortex and destroys the primary engine of callus formation. Modern minimally invasive plate osteosynthesis (MIPO) techniques are specifically designed to preserve this vital envelope.
The external soft-tissue response is equally dependent on mechanical factors and provides the rapid cellular activity necessary for early bridging callus. This response can be severely depressed by rigid immobilization or extensive surgical dissection.
MECHANOBIOLOGY: THE INFLUENCE OF STRAIN ON OSTEOGENESIS
The type, amount, and location of bone formed during fracture repair are heavily influenced by the mechanical environment—specifically, the degree of interfragmentary strain.
Bassett and Herrmann classically demonstrated that mesenchymal cells subjected to compression and low oxygen tension differentiate into chondroblasts (forming cartilage), whereas cells under tension and high oxygen tension differentiate into fibroblasts (forming fibrous tissue).
Carter et al. further expanded on this, proposing that the specific type of stress applied to immature tissue dictates the pathway of ossification.
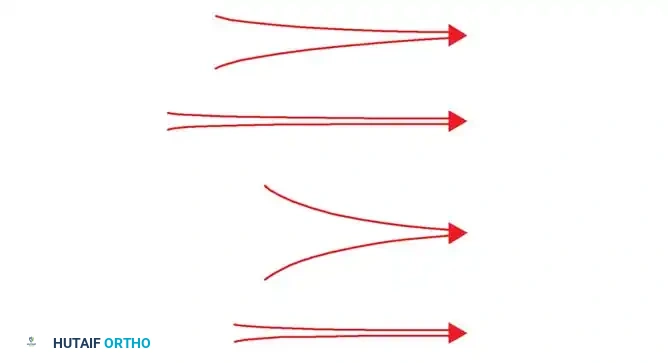
Hypothetical bone formation mechanism under different types of mechanical stress, illustrating how intermittent compressive/shear stresses promote endochondral ossification, while high shear or tensile stresses lead to fibrous tissue formation.
Clinical Application of Mechanobiology
- Absolute Stability (Strain < 2%): Achieved via lag screws and compression plates. This eliminates micro-motion, bypassing callus formation entirely. Healing occurs via primary osteonal reconstruction (intramembranous ossification and creeping substitution).
- Relative Stability (Strain 2% - 10%): Achieved via intramedullary nails, bridge plates, or external fixators. This permits controlled micro-motion, stimulating robust secondary healing via endochondral ossification (callus formation).
- Instability (Strain > 10%): Excessive motion tears the delicate granulation tissue and newly forming capillaries, leading to hypertrophic nonunion or fibrous tissue formation.
SYSTEMIC AND LOCAL FACTORS INFLUENCING FRACTURE HEALING
Fracture healing does not occur in a vacuum; it is profoundly affected by the patient's physiological state and the local wound environment. Uhthoff classified these factors into systemic and local categories.
Systemic Factors
- Endocrine and Metabolic: Diabetes mellitus significantly impairs microvascular perfusion and cellular proliferation. Growth hormone and thyroid hormones enhance healing, while systemic corticosteroids induce microvascular osteonecrosis and inhibit osteoblastogenesis.
- Pharmacological Agents: Nonsteroidal anti-inflammatory drugs (NSAIDs) inhibit cyclooxygenase (COX) enzymes, thereby suppressing prostaglandin E2 (PGE2) synthesis—a critical mediator of the early inflammatory phase. Prolonged NSAID use is a known risk factor for delayed union.
- Substance Abuse: Nicotine is a potent vasoconstrictor that diminishes peripheral tissue oxygenation and inhibits osteoblast function. Smoking cessation is mandatory for optimal osteogenesis.
Local Factors
- Degree of Trauma: High-energy injuries (e.g., severe comminution, open fractures) strip the bone of its soft-tissue envelope, destroying the local blood supply.
- Surgical Factors: Implant-induced altered blood flow, the degree of fixation rigidity, and the extent of surgical trauma (e.g., thermal necrosis from drilling) directly impact the biological viability of the fracture ends.
HOST CLASSIFICATION AND RISK STRATIFICATION
Predicting complications in fracture healing requires evaluating the whole patient. Karladani et al. demonstrated that soft-tissue condition and trauma energy are primary predictors of complications. Furthermore, Bowen et al. found that the number of compromising comorbidities directly correlates with infection rates in long bone fractures.
To systematize this, a comprehensive Host Classification system is utilized, serving as a critical adjunct to the Gustilo-Anderson classification for open fractures:
- Type A Host (Normal): No systemic illness, no immunosuppression, no substance abuse, stable socioeconomic status. (Infection risk: ~11%)
- Type B Host (Compromised but Controlled): One controlled systemic illness (e.g., controlled diabetes), isolated tobacco use, impaired socioeconomic status. (Infection risk: ~17%)
- Type C Host (Severely Compromised): Uncontrolled or multiple systemic illnesses, active immunosuppression, polysubstance abuse, severe malnutrition. (Infection risk: ~32%)
Clinical Pearl: Morbid obesity (BMI ≥ 40) is an independent predictor of complications. Karunakar et al. noted that morbidly obese patients are five times more likely to develop wound infections following operative fixation of pelvic and acetabular fractures. Host classification should dictate the aggressiveness of debridement and the timing of definitive wound closure.
SURGICAL AUGMENTATION: BONE GRAFTING AND ORTHOBIOLOGICS
When the biological capacity for fracture healing is overwhelmed—due to massive bone loss, atrophic nonunion, or severe host compromise—surgical intervention with bone grafting is indicated. A successful bone graft must possess one or more of the following properties:
1. Osteoconduction: Providing a physical, three-dimensional scaffold for cellular ingrowth and vascularization.
2. Osteoinduction: Providing biochemical signals (e.g., Bone Morphogenetic Proteins - BMPs) that recruit and induce mesenchymal stem cells to differentiate into osteoblasts.
3. Osteogenesis: Providing living, viable osteoprogenitor cells capable of directly forming new bone.
Autogenous Bone Graft (The Gold Standard)
Autograft remains the gold standard because it is the only material that possesses all three properties: osteoconductive matrix, osteoinductive proteins, and osteogenic cells.
* Harvesting Techniques: The anterior or posterior iliac crest is the traditional donor site. More recently, the Reamer-Irrigator-Aspirator (RIA) system has been utilized to harvest large volumes of highly osteogenic intramedullary graft from the femur or tibia.
* Complications: Autograft harvesting is not benign. It increases operative time, blood loss, and carries significant donor-site morbidity, including chronic pain, hematoma, infection, and iatrogenic fractures.
Allogenic Bone Grafts
Allografts provide an osteoconductive scaffold and are available in massive structural forms or cancellous chips.
* Processing: To eliminate the risk of viral transmission and immune rejection, allografts undergo rigorous processing (freezing, irradiation, or freeze-drying). Unfortunately, this sterilization destroys all osteogenic cells and significantly degrades the osteoinductive proteins.
* Incorporation: Allografts incorporate slowly via creeping substitution. The host bone must resorb the dead graft tissue before laying down new bone, making structural allografts susceptible to late fatigue fractures.
Bone Graft Substitutes and Tissue Engineering
To circumvent the limitations of autograft and allograft, a vast array of bone graft substitutes has been developed.
Table: Categories of Bone Graft Substitutes
* Ceramic-based: Calcium phosphate, calcium sulfate, and hydroxyapatite. These are purely osteoconductive and rely on the host's biology to populate the scaffold.
* Factor-based: Recombinant human Bone Morphogenetic Proteins (rhBMP-2 and rhBMP-7). These are powerfully osteoinductive and are used in recalcitrant nonunions or complex spinal fusions.
* Cell-based: Bone Marrow Aspirate Concentrate (BMAC). As pioneered by Connolly and Shindell, percutaneous injection of autogenous bone marrow delivers a high concentration of mesenchymal stem cells directly to the fracture site. When combined with an osteoconductive ceramic scaffold, BMAC mimics the properties of an autograft without the associated donor-site morbidity.
SURGICAL APPROACHES AND POSTOPERATIVE PROTOCOLS
Step-by-Step Biological Osteosynthesis
- Preoperative Planning: Assess the host classification (Type A, B, or C) and the mechanical demands of the fracture. Select implants that provide the appropriate mechanical environment (absolute vs. relative stability).
- Positioning and Approach: Utilize radiolucent tables to facilitate intraoperative fluoroscopy. Employ indirect reduction techniques and MIPO approaches to preserve the fracture hematoma and the periosteal blood supply.
- Handling of the Fracture Site: Avoid aggressive stripping of soft tissues. If bone grafting is required, prepare the recipient bed by decorticating sclerotic bone until punctate bleeding (the "paprika sign") is observed, ensuring a vascularized bed for the graft.
- Fixation: Apply the chosen implant. If using a bridge plate for a comminuted diaphyseal fracture, ensure a long working length to distribute strain and promote secondary callus formation.
Postoperative Rehabilitation
The postoperative protocol must balance the mechanical protection of the implant with the biological stimulation of the bone.
* Early Phase (0-2 weeks): Focus on soft-tissue healing, edema control, and early range of motion of adjacent joints to prevent stiffness and promote venous return.
* Intermediate Phase (2-6 weeks): Introduce controlled, progressive weight-bearing. According to mechanobiology principles, cyclical axial loading stimulates endochondral ossification. The progression of weight-bearing should be guided by the radiographic appearance of bridging callus.
* Late Phase (6+ weeks): Full weight-bearing and functional rehabilitation. Monitor for signs of delayed union (lack of progressive callus at 3 months) or nonunion (no healing at 6-9 months), which may necessitate biological augmentation (e.g., BMAC injection) or mechanical revision.
CONCLUSION
Fracture healing is a marvel of biological engineering. As orthopaedic surgeons, our role is not to "heal" the bone, but to meticulously construct the optimal mechanical and biological environment that allows the body's innate regenerative cascade to proceed unhindered. By deeply understanding the cellular phases of osteogenesis, the mechanobiology of strain, the systemic risks of the host, and the strategic application of orthobiologics, surgeons can reliably guide even the most complex fractures to successful clinical union.
📚 Medical References
- fracture healing: an overview for clinicians, I, Clin Orthop Relat Res 248:283, 1989.
- Frost HM: The biology of fracture healing: an overview for clinicians, II, Clin Orthop Relat Res 248:294, 1989.
- Girdlestone GR: The treatment of fractures in the light of their ischaemic complications, J Bone Joint Surg 14:755, 1932.
- Gustilo RB: The fracture classifi cation manual, St Louis, 1991, Mosby. Hallab N, Merritt K, Jacobs JJ: Current concepts review: metal sensitivity in patients with orthopaedic implants, J Bone Joint Surg 83A:428, 2001.
- Heckman MM, Whitesides TE, Grewe SR, et al: Histologic determination of the ischemic threshold of muscle in the canine
