Unlocking Faster Healing: Internal Fixation Devices Explained
Key Takeaway
For anyone wondering about Unlocking Faster Healing: Internal Fixation Devices Explained, Fracture healing rate is influenced by several factors. Cancellous bone heals faster (around 6 weeks) than cortical bone (9-18 weeks). Children's fractures unite rapidly with excellent remodeling, a process that slows with age. Maintaining stability, often through an internal fixation device or other methods, is crucial as excessive mobility at the fracture site impedes healing and callus formation.
Introduction and Epidemiology
Fracture management represents a cornerstone of orthopedic surgery, demanding a profound understanding of both the mechanical and biological environments required for optimal osteogenesis. The fundamental objective of internal fixation is to achieve a stable biomechanical construct that facilitates early, pain-free mobilization while establishing an environment conducive to rapid and predictable bone union. The epidemiology of fractures requiring internal fixation exhibits a bimodal distribution, characterized by high-energy trauma in the younger, predominantly male population, and low-energy fragility fractures in the increasingly prevalent geriatric demographic.
The selection of an internal fixation device—whether it be a dynamic compression plate, an intramedullary nail, or a specialized locking construct—profoundly alters the local mechanical environment. This mechanical modulation dictates whether the fracture will heal via primary (direct) bone healing or secondary (indirect) bone healing. Primary bone healing occurs without intermediate callus formation and requires absolute stability, typically achieved through rigid internal fixation and interfragmentary compression. Secondary bone healing involves endochondral ossification and external callus formation, requiring relative stability to stimulate the biological cascade. Understanding the myriad factors affecting the rate of healing, including the type of bone involved, the age of the patient, the degree of mobility at the fracture site, and the physical approximation of the bone ends, is critical for the orthopedic surgeon when designing a preoperative plan and selecting the appropriate internal fixation modality.
Surgical Anatomy and Biomechanics
The rate and mechanism of fracture healing are intrinsically linked to the microstructural anatomy of the involved bone and the biomechanical forces applied across the fracture gap. Internal fixation devices must be tailored to the specific anatomical and biomechanical demands of the fracture pattern.
Cancellous Versus Cortical Bone Healing
The architectural differences between cancellous (spongy) bone and cortical (compact) bone dictate distinct healing timelines and mechanical requirements.
Healing in cancellous bone, characterized by its porous, trabecular network and rich vascular supply, is generally well advanced six weeks from the time of the injury. The extensive surface area of the trabeculae allows for rapid revascularization and osteoblastic activity. Consequently, mechanical protection of the fracture can almost invariably be abandoned by the six-week mark. This principle applies to fractures of bones composed principally of cancellous tissue, as well as to fractures involving the metaphyseal and epiphyseal regions of long bones. Clinical applications of this anatomical rule include the following parameters
First, weight-bearing after a fracture of the calcaneus may typically be permitted after approximately six weeks. Second, a patient presenting with a traumatic wedge fracture of a vertebral body may safely commence full mobilization after six weeks. Third, plaster or rigid splint fixation may be discarded after five to six weeks following a Colles fracture of the distal radius. Fourth, weight may be allowed through the lower extremity six weeks following a surgically or conservatively managed fracture of the tibial plateau. Finally, bed rest or strict non-weight-bearing protocols for six weeks are usually advised for any fracture of the pelvis involving the posterior ring or those specific anatomical zones through which axial weight transmission is mediated.
Conversely, cortical bone healing relies heavily on the Haversian systems and requires a significantly longer duration to achieve structural integrity. Endosteal callus in diaphyseal cortical bone may take many months to become reasonably well established. Many uncomplicated long bone diaphyseal fractures may take 9 to 18 weeks to achieve radiographic and clinical union. In certain biomechanical environments, however, abundant external bridging callus—stimulated by relative stability—may allow an earlier return of function. For example, the average time to union of a fracture of the tibial shaft treated conservatively with a cast or functionally with relative stability is 16 weeks. Fractures of the humeral shaft, which tolerate functional bracing well due to the surrounding soft tissue envelope and gravity-assisted alignment, can often be left unsupported after 10 weeks. Interestingly, fractures of the metatarsals, metacarpals, and phalanges, where external bridging callus is usually substantial and the mechanical lever arms are shorter, are typically quite firm and clinically stable in 4 to 5 weeks.
The Role of Patient Age in Osteogenesis
Patient age is a paramount biological factor influencing the speed and quality of fracture union. In the pediatric population, the union of fractures is remarkably rapid due to a highly active periosteum, abundant osteoprogenitor cells, and robust vascularity. The speed of union inversely correlates with age, decreasing steadily until skeletal maturity is reached. Following epiphyseal closure, the baseline rate of osteogenesis plateaus, leaving little difference in the intrinsic healing rate between young adults and the elderly, provided there are no confounding metabolic bone diseases.
To illustrate the pediatric chronologic advantage, union may be expected in a fractured pediatric femur a little after the number of weeks equivalent to the child's numerical age have passed. A fractured femur in a child of three years is usually united and stable after four weeks; a fractured femur in a child of eight years is usually sound after nine weeks. In stark contrast, a fracture of the femoral shaft in a skeletally mature adult managed with intramedullary fixation may take three to six months to achieve complete radiographic union.
Beyond the sheer rapidity of union, pediatric patients possess remarkable powers of structural remodeling. This remodeling potential is dictated by the Heuter-Volkmann principle and Wolff's Law, driven by the proximity of the fracture to an active physis and the plane of the adjacent joint's motion. These powers are excellent for correcting translational displacement and are often highly effective for slight to moderate angulation in the plane of joint motion. However, remodeling is notoriously poor in the case of axial rotational deformities in both adults and children. The physiological power to remodel decreases precipitously once adolescence is reached and epiphyseal fusion becomes imminent, necessitating near-anatomic reduction in older adolescents mimicking adult standards.
Fracture Site Mobility and Strain Theory
The biomechanical concept of strain, elucidated by Perren, defines the deformation of granulation tissue within the fracture gap relative to the original gap width. Excessive mobility persisting at the fracture site—often due to poor fixation, inappropriate implant selection, or patient non-compliance—may severely interfere with the vascularization of the fracture hematoma. High strain environments lead to the disruption of early bridging callus, shear forces that destroy fragile capillary buds, and the ultimate prevention of endosteal new bone growth, culminating in a hypertrophic nonunion.
One of the primary objectives of all forms of internal and external splintage is to reduce mobility at the fracture site to a biologically tolerable level, thereby encouraging union. Absolute rigidity, achieved via lag screws and compression plates, reduces strain to less than two percent. While this facilitates direct osteonal remodeling (primary bone healing), it completely discourages external callus formation. In these constructs, the surgeon places total reliance on the internal fixation device—typically a plate—to withstand physiological loads until the long, slow process of endosteal union is complete. If the chosen splintage is biologically or mechanically inadequate, union will inevitably be delayed or permanently prevented.
Indications and Contraindications
The decision to proceed with operative internal fixation versus non-operative management requires a meticulous analysis of the fracture pattern, soft tissue envelope, patient comorbidities, and functional demands. Internal fixation is broadly indicated when conservative measures cannot achieve or maintain an acceptable reduction, or when prolonged immobilization would result in unacceptable systemic or local morbidity.
Summary of Operative Versus Non Operative Management
| Clinical Scenario | Operative Indication | Non Operative Indication | Rationale for Management Strategy |
|---|---|---|---|
| Intra-articular Fractures | Step-off > 2mm, articular incongruity, instability. | Non-displaced, stable patterns with intact capsular restraints. | Articular incongruity leads to post-traumatic osteoarthritis. Anatomic reduction and absolute stability are mandatory for joint preservation. |
| Diaphyseal Long Bone Fractures | Polytrauma, open fractures, failure of conservative management, segmental patterns. | Acceptable alignment parameters met (e.g., tibial shaft with <5 degrees varus/valgus). | IM nailing offers relative stability and early weight-bearing. Conservative bracing relies on soft tissue hinges and secondary healing. |
| Geriatric Hip Fractures | Nearly all cases (femoral neck, intertrochanteric). | Non-ambulatory patient with severe terminal illness (comfort care). | Early operative fixation reduces mortality, prevents decubitus ulcers, DVT/PE, and pulmonary complications associated with prolonged recumbency. |
| Open Fractures | Gustilo-Anderson Grades I-III requiring debridement and stabilization. | None. All open fractures require surgical evaluation and debridement. | Internal or external fixation stabilizes the soft tissue envelope, reducing the risk of deep infection and facilitating wound care. |
| Pediatric Physeal Fractures | Salter-Harris III/IV, or irreducible SH I/II. | Minimally displaced SH I/II. | Anatomic reduction of the physis is critical to prevent premature growth arrest and subsequent angular deformity. |
Contraindications to internal fixation include active, untreated systemic or local infections (which may necessitate external fixation as a temporizing measure), severe medical comorbidities precluding anesthesia, and critically compromised soft tissue envelopes that cannot tolerate surgical incisions without impending necrosis.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the bedrock of successful internal fixation. This involves a comprehensive radiographic evaluation, including orthogonal plain films and, frequently, computed tomography (CT) scans for complex articular or pelvic ring injuries. Digital templating is mandatory to determine the appropriate implant type, length, and screw trajectory, as well as to anticipate potential intraoperative anatomical challenges.
Patient positioning must facilitate optimal fluoroscopic access and allow for the planned surgical approach without compromising the sterile field. Depending on the fracture, this may involve a radiolucent flat table, a fracture table with skeletal traction capabilities, or specialized positioning devices such as a lateral decubitus beanbag.
Assessment of Bone End Separation
A critical component of preoperative and intraoperative assessment is evaluating the physical proximity of the fracture fragments. Union will be significantly delayed or entirely prevented if the bone ends are separated, as this physical gap interferes with the normal biological mechanisms of healing, preventing the bridging of the fracture hematoma and subsequent callus formation. The converse is also biomechanically true: compression of the fracture gap facilitates primary union by increasing friction between fragments, thereby increasing construct stability and decreasing strain.
Separation of bone ends may occur under several pathological or iatrogenic circumstances. Excessive skeletal traction, over-distraction during intramedullary nailing, or the improper application of a neutralization plate without prior interfragmentary compression can leave a persistent gap. Furthermore, soft tissue interposition is a classic and frequently encountered barrier to reduction and healing.
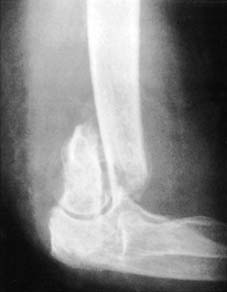
As demonstrated in the radiographic and anatomical assessments of supracondylar fractures of the humerus, soft tissue—such as the brachialis muscle, periosteal flaps, or neurovascular bundles—can become incarcerated between the bone ends. When soft tissue interposition is identified or suspected due to a rubbery, irreducible fracture gap, open reduction is mandated to extricate the entrapped tissue, protect neurovascular structures, and achieve intimate bony apposition prior to the application of internal fixation devices.
Detailed Surgical Approach and Technique
The surgical execution of internal fixation is guided by the AO principles: anatomic reduction of articular surfaces, stable fixation to fulfill local biomechanical demands, preservation of blood supply through meticulous handling of soft tissues, and early, active mobilization.
Principles of Internal Fixation Application
The approach to the fracture site must utilize established internervous and intermuscular planes to minimize denervation and devascularization of the surrounding musculature. For instance, the anterolateral approach to the humeral shaft utilizes the plane between the brachialis (musculocutaneous and radial nerves) and the brachioradialis (radial nerve), providing safe access to the diaphysis.
Once the fracture is exposed, the surgeon must decide between direct and indirect reduction techniques. Direct reduction, utilizing bone forceps and clamps, is appropriate for articular fractures and simple diaphyseal fractures where absolute stability is desired. This requires stripping of the periosteum adjacent to the fracture, which must be minimized to preserve the cortical blood supply.
For the application of a dynamic compression plate (DCP) or limited contact dynamic compression plate (LC-DCP), the plate is contoured to the tension band surface of the bone. An interfragmentary lag screw is often placed either outside or through the plate to compress the fracture plane. The plate is then secured using eccentric drilling techniques in the specialized screw holes, which translates the screw head down the slope of the hole, pulling the bone fragment longitudinally and generating axial compression across the fracture site.
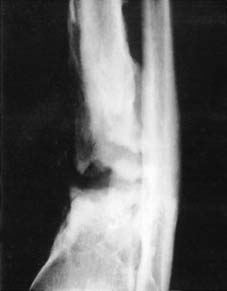
Conversely, in complex, multifragmentary diaphyseal or metaphyseal fractures, indirect reduction techniques are preferred. This involves restoring length, alignment, and rotation without exposing the fracture site directly, thereby preserving the vital fracture hematoma and periosteal blood supply. This is often achieved using closed traction, external fixators as reduction tools, or minimally invasive plate osteosynthesis (MIPO). In MIPO, a locking compression plate (LCP) is slid submuscularly across the fracture zone. Locking screws, which thread into the plate itself, create a fixed-angle construct that acts as an internal external-fixator. This provides excellent relative stability without relying on friction between the plate and the bone, preserving periosteal perfusion and promoting robust secondary callus formation.
Intramedullary nailing represents another powerful technique for relative stability, primarily indicated for diaphyseal fractures of the femur, tibia, and humerus. The technique involves establishing a starting point collinear with the medullary canal, passing a guide wire across the fracture, reaming the canal to generate autologous bone graft and increase the working diameter, and inserting a load-sharing titanium or steel nail. Interlocking screws are placed proximally and distally to control axial length and rotational alignment.
Complications and Management
Despite meticulous surgical technique and advanced internal fixation devices, complications can arise from mechanical failures, biological failures, or a combination of both. The orthopedic surgeon must be adept at identifying the etiology of the complication to implement the correct salvage strategy.
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology and Pathophysiology | Salvage Strategy and Management |
|---|---|---|---|
| Hypertrophic Nonunion | 5-10% of long bone fractures | Adequate biology (excellent blood supply) but inadequate mechanical stability (excessive strain). Characterized by "elephant foot" callus on radiographs. | Mechanical stabilization. Exchange nailing to a larger diameter nail, or application of a rigid compression plate. Bone grafting is rarely necessary. |
| Atrophic Nonunion | 2-5% of long bone fractures | Inadequate biology (poor blood supply, stripped periosteum, severe soft tissue injury). Little to no callus formation visible on radiographs. | Biological and mechanical augmentation. Requires debridement of necrotic bone ends, opening of the medullary canal, rigid internal fixation, and autologous bone grafting (e.g., iliac crest). |
| Hardware Failure (Breakage) | 1-3% | Fatigue failure of the implant due to delayed union or nonunion. The implant undergoes cyclical loading beyond its endurance limit before the bone heals. | Hardware removal, reassessment of the biological and mechanical environment, re-fixation with a more robust construct, and biological stimulation (bone graft). |
| Deep Surgical Site Infection | 1-2% (Closed), up to 30% (Severe Open) | Bacterial colonization of the hardware and necrotic tissue. Formation of a protective biofilm on the implant surface. | Aggressive surgical debridement (irrigation and debridement). If hardware is stable, suppressive antibiotics until union. If hardware is loose, removal, temporary external fixation or antibiotic spacer (Masquelet technique), and delayed reconstruction. |
| Malunion | Variable depending on bone | Inadequate intraoperative reduction, loss of fixation postoperatively, or asymmetrical growth arrest in pediatric patients. | Corrective osteotomy (opening wedge, closing wedge, or dome osteotomy) followed by rigid internal fixation to restore mechanical axis and joint orientation. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol is as critical as the surgical intervention itself. The protocol must be tailored to the specific internal fixation construct utilized, the anatomical location of the fracture, and the biological timeline of bone healing discussed previously.
For fractures involving cancellous bone that have been rigidly fixed (e.g., a tibial plateau fracture managed with a locking plate), the mechanical construct allows for immediate, active range of motion of the adjacent joints to prevent arthrofibrosis and promote cartilage nutrition. However, axial loading must be restricted. Based on the biological timeline of cancellous bone, protected weight-bearing may typically commence at six weeks postoperatively, progressing to full weight-bearing as radiographic consolidation becomes evident.
For diaphyseal fractures of cortical bone managed with intramedullary nails (e.g., a tibial shaft fracture), the load-sharing nature of the implant often permits immediate weight-bearing as tolerated. The micromotion generated by early weight-bearing across the interlocked nail stimulates secondary external callus formation via Wolff's Law. In contrast, cortical fractures managed with rigid absolute stability (e.g., a both-bone forearm fracture plated with LC-DCPs) require protection from heavy lifting and torsional forces until primary bone healing is radiographically confirmed, often taking 12 to 16 weeks, though early active range of motion is strongly encouraged to prevent tendon adhesions.
Summary of Key Literature and Guidelines
The evolution of internal fixation is deeply rooted in the foundational research and evolving guidelines of the Arbeitsgemeinschaft für Osteosynthesefragen (AO Foundation). The original AO principles, established in 1958, emphasized anatomic reduction and absolute stability. However, landmark biomechanical studies by Perren in the late 1970s regarding strain theory catalyzed a paradigm shift toward biological osteosynthesis. Perren's work demonstrated that while absolute stability is necessary for articular fractures, relative stability and the preservation of the soft tissue envelope yield superior results for diaphyseal and complex metaphyseal fractures by harnessing the body's natural callus-forming capabilities.
Subsequent literature has heavily focused on the biomechanics of locking plates. Studies have shown that the pull-out strength of fixed-angle locking screws is significantly higher in osteoporotic bone compared to conventional cortical screws, revolutionizing the management of geriatric fragility fractures. Furthermore, guidelines from orthopedic trauma societies consistently advocate for the prompt administration of prophylactic antibiotics and early surgical stabilization of open fractures to mitigate infection rates, alongside the judicious use of damage control orthopedics (DCO) in the hemodynamically unstable polytrauma patient, where definitive internal fixation is delayed in favor of rapid, temporary external fixation.
