Mastering Necrotizing Soft Tissue Diagnosis with LRINEC Criteria
Introduction & Epidemiology
Necrotizing soft tissue infections (NSTIs) represent a heterogeneous group of rapidly progressive, life-threatening infections characterized by widespread necrosis of the fascia, subcutaneous tissue, and often muscle, with relative sparing of the skin in early stages. These infections are true surgical emergencies requiring prompt recognition, aggressive resuscitation, broad-spectrum antimicrobial therapy, and urgent, often serial, surgical debridement. Despite advancements in critical care and antimicrobial agents, mortality rates remain high, ranging from 15% to 35%, and can exceed 50% in cases of septic shock or delayed diagnosis.
The epidemiology of NSTIs reveals a global incidence of approximately 0.4 to 1 per 100,000 person-years, with a rising trend observed in recent decades, potentially due to an aging population, increasing prevalence of diabetes mellitus, obesity, and immunosuppression. Common risk factors include diabetes mellitus (present in 40-60% of cases), peripheral vascular disease, chronic kidney disease, malignancy, intravenous drug use, alcoholism, immunosuppression (e.g., organ transplantation, corticosteroid use, HIV), and prior trauma or surgery. While classic presentation often involves a clear portal of entry, up to 20% of cases can be spontaneous or cryptogenic.
Early and accurate diagnosis is paramount for improving patient outcomes. However, the initial clinical presentation of NSTI can be subtle and non-specific, often mimicking cellulitis or erysipelas, leading to delayed intervention. To address this diagnostic challenge, the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score was developed in 2004 by Wong et al. The LRINEC score is a validated tool designed to aid in differentiating NSTI from other severe soft tissue infections based on six routine laboratory parameters: C-reactive protein (CRP), total white blood cell count (WBC), hemoglobin (Hb), serum sodium (Na+), serum creatinine (Cr), and serum glucose (Glu).
A LRINEC score of ≥6 has been shown to have a positive predictive value of 92% and a negative predictive value of 96% for NSTI, suggesting that a high score warrants urgent surgical exploration, while a low score makes NSTI less likely but does not definitively rule it out. It is critical to understand that the LRINEC score is a diagnostic aid and should never replace clinical judgment, especially in patients with high clinical suspicion despite a low score. The score is particularly valuable in the initial assessment to expedite decision-making and mobilize resources for definitive management.
Surgical Anatomy & Biomechanics
Understanding the surgical anatomy of soft tissue layers and fascial planes is fundamental to comprehending the pathophysiology and guiding the surgical management of NSTIs. These infections typically spread along fascial planes, exploiting the relative avascularity of these layers compared to the richly vascularized muscle and skin.
The soft tissues of the musculoskeletal system consist of several distinct layers:
*
Skin:
Epidermis and dermis.
*
Subcutaneous tissue (superficial fascia):
Contains adipose tissue, superficial nerves, vessels, and lymphatics. This layer is often targeted in Type II NSTIs (monomicrobial, typically Group A Streptococcus).
*
Deep fascia:
A dense, fibrous connective tissue layer that envelops muscles and muscle compartments. This is the primary battleground for NSTIs, particularly Type I (polymicrobial). Infection here rapidly spreads, compromising blood supply to overlying skin and underlying muscle.
*
Muscle:
Enclosed within the deep fascia. Muscle necrosis is a hallmark of myonecrosis or clostridial gas gangrene.
*
Bone and periosteum:
Rarely directly involved initially, but can become secondarily infected in severe, extensive cases, leading to osteomyelitis.
The interconnectedness of fascial planes allows for rapid and extensive spread of infection. For instance, in the extremities, infection can track along the fascial septa dividing muscle compartments. In the trunk, the deep fascial planes of the anterior abdominal wall or paraspinal muscles can be involved. Knowledge of these planes is crucial for adequate surgical debridement, as incisions must extend to healthy, bleeding tissue beyond the involved compartments.
Biomechanical considerations in NSTI primarily relate to the consequences of tissue destruction and the physiological response. The intense inflammatory cascade leads to:
*
Microvascular thrombosis:
Compromises local blood supply, leading to ischemia and necrosis, particularly affecting the subcutaneous fat and deep fascia. This is a key distinguishing feature from cellulitis.
*
Tissue edema and compartment syndrome:
The exudate and inflammatory mediators cause significant edema, increasing interstitial pressure. In confined fascial compartments, this can lead to compartment syndrome, further exacerbating tissue ischemia and necrosis.
*
Bacterial toxins:
Exotoxins produced by bacteria (e.g., streptococcal pyrogenic exotoxins, clostridial alpha-toxin) cause direct cellular damage, systemic toxicity (toxic shock syndrome), and contribute to the rapid progression of tissue necrosis.
*
Loss of tissue integrity:
Necrotic fascia and muscle lose their structural integrity, contributing to wound dehiscence and functional impairment even after successful debridement.
From a biomechanical perspective, the loss of muscle and fascial integrity directly impacts musculoskeletal function. Extensive debridement can result in significant tissue deficits, leading to functional limitations requiring reconstructive efforts. Early recognition and debridement aim to minimize this tissue loss, preserving maximal function. The understanding of anatomical boundaries and potential pathways for spread is not merely academic but directly informs the scope and strategy of surgical intervention, dictating where incisions must be made and how far debridement must extend to truly reach healthy tissue.
Indications & Contraindications
The management of NSTI is primarily surgical. Indications for surgical exploration and debridement are driven by a high index of suspicion, often supported by clinical findings, imaging, and laboratory parameters, including the LRINEC score.
Indications for Operative Intervention (Surgical Exploration and Debridement):
1.
High Clinical Suspicion:
This is the paramount indication. Clinical signs highly suggestive of NSTI include:
* Pain out of proportion to physical findings.
* Rapid progression of erythema, edema, and tenderness.
* Crepitus (subcutaneous gas, palpable or on imaging).
* Skin changes: bullae (hemorrhagic or serous), dusky or violaceous discoloration, necrosis/gangrene, anesthesia of the skin (due to nerve damage).
* Systemic signs of sepsis or septic shock (fever, tachycardia, hypotension, altered mental status, multiorgan dysfunction).
* Failure to respond to appropriate antibiotic therapy for presumed cellulitis.
2.
LRINEC Score ≥ 6:
While not definitive, a high LRINEC score significantly increases the probability of NSTI and should prompt urgent surgical consultation and consideration for exploration.
3.
Imaging Findings:
Computed Tomography (CT) or Magnetic Resonance Imaging (MRI) demonstrating:
* Gas in the soft tissues (specific, but not sensitive).
* Thickening and edema of the deep fascia.
* Fluid collections tracking along fascial planes.
* Lack of clear tissue planes.
4.
Positive Finger Test:
A bedside diagnostic test where a small incision is made, and a blunt instrument (e.g., finger or clamp) is inserted into the wound. Ease of dissecting the fascia, absence of bleeding, and presence of "dishwater pus" are highly suggestive of NSTI.
5.
Clinical Deterioration:
Any patient with a severe soft tissue infection whose condition is rapidly worsening despite initial medical management should be considered for surgical exploration.
Contraindications for Operative Intervention:
True contraindications to surgical debridement for NSTI are exceedingly rare, as timely debridement is a life-saving measure. Most "contraindications" are relative and may necessitate a modified approach or careful risk-benefit analysis:
1.
Patient Instability:
While a severely unstable patient might require initial resuscitation, delaying debridement beyond a few hours significantly increases mortality. Often, bedside debridement or debridement in the intensive care unit may be necessary if transfer to the operating room is deemed too risky. The goal is to stabilize
for
surgery, not
instead of
surgery.
2.
Palliative Care:
In patients with known terminal illness, multiple comorbidities, or advanced age, where the overall prognosis is dismal and the burden of aggressive surgery outweighs potential benefits, a decision for palliative care may be made in conjunction with the patient, family, and medical team. This is a rare and highly individualized decision.
3.
Absence of Necrosis:
If surgical exploration unequivocally demonstrates no evidence of fascial or subcutaneous necrosis, the procedure can be terminated. However, this is usually determined intraoperatively after surgical access.
Operative vs. Non-Operative Indications in Necrotizing Soft Tissue Infections
| Feature / Indication | Operative Management (Surgical Debridement) | Non-Operative Management (Medical/Supportive) |
|---|---|---|
| LRINEC Score | ≥6 (high suspicion, warrants exploration) | <6 (reduces suspicion, but clinical judgment paramount) |
| Clinical Presentation | Pain out of proportion, skin anesthesia, bullae, dusky skin, crepitus, rapid progression, systemic toxicity (sepsis/shock), failure of conservative management. | Localized erythema/warmth, pain proportional to findings, no systemic toxicity, clear response to initial antibiotics (i.e., true cellulitis/erysipelas). |
| Diagnostic Imaging (CT/MRI) | Fascial thickening, gas in soft tissues, fluid collections tracking along planes, lack of clear tissue planes. | No evidence of deep fascial involvement or gas; findings consistent with superficial cellulitis. |
| Finger Test | Positive (easy dissection of fascia, dishwater pus, no bleeding). | Negative (firm fascial planes, normal bleeding). |
| Pathophysiology | Tissue necrosis (fascia, subcutaneous, muscle); microvascular thrombosis; widespread inflammation. | Localized inflammation; no tissue necrosis or vascular compromise. |
| Urgency | Extreme urgency (surgical emergency); often within 1-2 hours of diagnosis. | Urgent but not emergent; can be managed with antibiotics and supportive care. |
| Goals | Source control (remove necrotic tissue), prevent spread, reduce bacterial load, mitigate systemic toxicity. | Eradicate infection, reduce inflammation, prevent complications. |
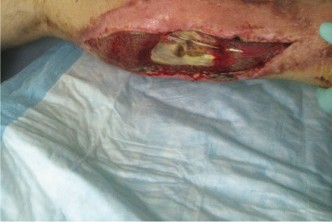
Pre-Operative Planning & Patient Positioning
Once the diagnosis of NSTI is suspected or confirmed, immediate and concurrent efforts must be made for patient resuscitation, pre-operative planning, and rapid transfer to the operating room. Time is critical, with every hour of delay in debridement increasing mortality.
1. Resuscitation and Stabilization:
*
Airway, Breathing, Circulation (ABC):
Standard trauma/critical care protocols. Many patients present in septic shock requiring aggressive fluid resuscitation and vasopressor support.
*
Intravenous Access:
Secure large-bore peripheral IVs or central venous access.
*
Antibiotics:
Initiate broad-spectrum intravenous antibiotics immediately after blood cultures are drawn. The regimen should cover Gram-positive (including MRSA), Gram-negative, and anaerobic organisms. Common initial regimens include a carbapenem (e.g., meropenem) or a combination of piperacillin-tazobactam, with vancomycin or linezolid for MRSA coverage, and clindamycin (due to its anti-toxin properties, especially for streptococcal or clostridial infections). Tailor based on local epidemiology and susceptibility patterns.
*
Labs:
Complete blood count, comprehensive metabolic panel, coagulation profile, lactate, CRP, LRINEC parameters, arterial blood gas. Re-evaluate frequently.
*
Imaging:
While surgery should not be delayed waiting for imaging, if there is diagnostic uncertainty, a rapid CT scan can help delineate the extent of infection, identify gas, and guide surgical planning. However, if clinical suspicion is high, proceed directly to the OR.
*
Consultations:
Involve critical care, infectious disease, and potentially plastic surgery early in the process.
2. Operating Room Preparation:
*
Anesthesia:
Alert anesthesia to the urgency and the potential for a critically ill patient requiring massive fluid shifts, blood product transfusions, and vasopressor support. An arterial line for continuous blood pressure monitoring and a central venous catheter are often required.
*
Equipment:
Prepare for extensive debridement. Have ample sterile drapes, large suction catheters, electrosurgical devices, copious irrigation fluids (saline, sterile water), various scalpel blades, and appropriate instruments for deep tissue dissection. Consider having a tissue biopsy kit available for sending samples for Gram stain, culture, and histopathology.
*
Blood Products:
Ensure cross-matched blood products (pRBCs, FFP, platelets) are readily available in the operating room.
3. Patient Positioning:
Patient positioning depends entirely on the anatomical location of the NSTI. The goal is to maximize exposure to allow for comprehensive debridement of all affected tissue.
*
Extremities:
Supine or lateral positions with the affected limb prepped circumferentially to allow for extensive incisions and potential rotation for access to all aspects of the limb. Use appropriate limb holders or supports.
*
Trunk/Abdomen:
Supine for anterior abdominal wall. Prone or lateral for back/flank infections.
*
Perineum/Genitalia (Fournier's Gangrene):
Lithotomy position for optimal exposure of the perineum, scrotum/labia, and buttock region.
*
Neck:
Supine with neck extension.
Always ensure pressure points are padded and that positioning does not compromise airway or vascular access. The surgical field must be prepped widely, often from the hairline to the toes, to allow for unpredictable extension of the debridement.
Detailed Surgical Approach / Technique
The surgical approach to NSTI is characterized by urgency, aggressiveness, and seriality. The primary goal is complete debridement of all necrotic tissue to achieve source control.
1. Initial Incision and Exploration:
*
Liberal Incision:
Make generous, longitudinal incisions that extend well beyond the visible skin erythema. Do not be constrained by cosmetic concerns. The incision should be deep enough to reach the deep fascia. For limb infections, multiple parallel incisions or a long curvilinear incision may be necessary to expose entire fascial compartments.
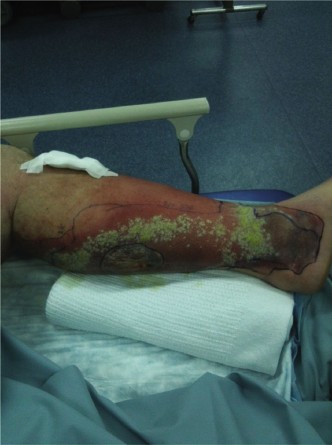
*
Assessment of Fascia:
Once the deep fascia is exposed, carefully inspect it. Healthy fascia is glistening, firm, and resists blunt dissection. Necrotic fascia appears dull, grayish, edematous, and friable. It often dissects easily from underlying muscle with a finger or blunt instrument ("finger test" performed intraoperatively). This is where the characteristic "dishwater pus" can be encountered.
*
Assessment of Subcutaneous Tissue:
Necrotic subcutaneous fat may appear greyish, liquefied, and avascular.
*
Assessment of Muscle:
Healthy muscle is beefy red, firm, and contracts when stimulated (e.g., with electrocautery). Necrotic muscle is dull, grayish, friable, and does not contract.
2. Debridement Strategy:
*
Radical Excision:
Systematically debride all non-viable tissue. This includes skin, subcutaneous fat, fascia, and muscle that are necrotic. The debridement must continue until only healthy, bleeding tissue remains.
*
Skin and Subcutaneous Tissue:
Excision of discolored, anesthetic, or bullae-ridden skin and liquefied, greyish subcutaneous fat.
*
Fascia:
Excision of all dull, non-bleeding, easily dissectable fascia. Use a scalpel or electrosurgery to cut back to healthy, firm fascia that bleeds when incised.
*
Muscle:
If muscle is involved (myonecrosis), resect all non-contractile, non-bleeding muscle. This can be extensive and may involve functional muscle groups.
*
Probing Fascial Planes:
Explore widely. Use fingers or blunt instruments to probe along fascial planes, ensuring no hidden pockets of infection or necrosis are missed. If any resistance is met, or if there is suspicion of tracking, extend incisions to explore further.
*
Biopsies:
Send tissue samples (from both affected and apparently healthy margins) for Gram stain, aerobic and anaerobic cultures, and histopathology. This guides subsequent antibiotic therapy and confirms the diagnosis.
*
Irrigation:
Copiously irrigate the wound with sterile saline.
*
Hemostasis:
Achieve meticulous hemostasis. Extensive debridement can lead to significant blood loss.
3. Wound Management Post-Debridement:
*
Packing:
After initial debridement, the wound is typically left open and packed loosely with wet-to-dry dressings (saline-soaked gauze) or iodine-soaked gauze. This allows for continued drainage, prevents premature wound closure over residual infection, and facilitates repeated examinations.
*
Negative Pressure Wound Therapy (NPWT):
NPWT can be applied after initial debridement to manage large, open wounds. It helps remove exudate, reduce edema, promote granulation tissue formation, and stabilize the wound prior to definitive closure or reconstruction.
*
Repeated Look-Backs (Serial Debridements):
This is a cornerstone of NSTI management. Patients are typically returned to the operating room every 24-48 hours (or sooner if clinical deterioration) for re-evaluation and further debridement until all necrotic tissue is removed and healthy, bleeding margins are definitively established. This may require multiple surgical procedures.
4. Internervous Planes (for specific anatomical sites):
While NSTI primarily affects fascial planes, an understanding of internervous planes is critical for orthopedic surgeons operating in specific areas to ensure thorough debridement while minimizing damage to vital structures. For example, in the upper extremity, knowing the internervous plane between the deltoid and pectoralis major for anterior shoulder access, or between the brachialis and brachioradialis for anterior humerus, allows for muscle-sparing access to deeper structures if they are involved. However, in the context of NSTI, the priority is aggressive debridement, and functional preservation is secondary to source control and life-saving measures. Nevertheless, careful dissection around neurovascular bundles is essential to prevent iatrogenic injury.
5. Reduction and Fixation (if bone/joint involved):
While less common as an initial presentation, if the infection has spread to involve bone (osteomyelitis) or joints (septic arthritis), these must be addressed directly.
*
Osteomyelitis:
Debridement of necrotic bone, potentially requiring sequestrectomy. Antibiotic beads or cement spacers may be used.
*
Septic Arthritis:
Arthrotomy, irrigation, debridement of synovium, and often placement of drains.
Fixation typically refers to fracture management. In the context of NSTI, if a fracture coexists, external fixation is generally preferred over internal fixation due to the high risk of implant contamination and infection in the presence of necrotizing soft tissues. Internal fixation in such circumstances is typically reserved for reconstructive phases after infection is fully controlled.

Complications & Management
Necrotizing soft tissue infections are associated with a high burden of complications, both acute and long-term, due to the aggressive nature of the infection and the extensive surgical interventions required.
Acute Complications:
*
Sepsis and Septic Shock:
The most immediate and life-threatening complication, leading to multi-organ dysfunction syndrome (MODS).
*
Incidence:
Near 100% in severe cases.
*
Salvage:
Aggressive resuscitation (fluids, vasopressors), broad-spectrum antibiotics, source control (debridement), monitoring in ICU, addressing organ dysfunction.
*
Acute Kidney Injury (AKI):
Common due to sepsis, hypovolemia, rhabdomyolysis (if muscle necrosis), and nephrotoxic antibiotics.
*
Incidence:
30-60%.
*
Salvage:
Maintain euvolemia, avoid nephrotoxic agents, consider renal replacement therapy (dialysis).
*
Acute Respiratory Distress Syndrome (ARDS):
Due to systemic inflammatory response, aspiration, or direct lung injury.
*
Incidence:
10-30%.
*
Salvage:
Lung-protective ventilation strategies, prone positioning, supportive care.
*
Disseminated Intravascular Coagulation (DIC):
A severe systemic complication causing widespread microvascular thrombosis and hemorrhagic diathesis.
*
Incidence:
10-20%.
*
Salvage:
Treat underlying sepsis, judicious blood product transfusion (platelets, FFP, cryoprecipitate).
*
Limb Loss/Amputation:
Required if infection is uncontrollable, threatens life, or results in irreversible tissue damage.
*
Incidence:
10-25%.
*
Salvage:
Aggressive early debridement, serial debridements, timely decision-making regarding amputation to save life.
*
Recurrent Infection:
Inadequate initial debridement or new inoculation.
*
Incidence:
Variable, especially if initial debridement is incomplete.
*
Salvage:
Re-exploration and debridement, review antibiotic regimen, wound care optimization.
Long-Term Complications:
*
Extensive Soft Tissue Defects:
Resulting from radical debridement, requiring complex reconstructive procedures (skin grafts, flaps).
*
Incidence:
High, depending on initial extent.
*
Salvage:
Serial debridement until a healthy wound bed, negative pressure wound therapy, early involvement of plastic surgery for reconstruction.
*
Functional Impairment:
Due to muscle loss, joint contractures, nerve damage, or extensive scarring.
*
Incidence:
Common, particularly in extremity or perineal infections.
*
Salvage:
Early and aggressive physical and occupational therapy, splinting, surgical releases, tendon transfers, or other reconstructive options.
*
Chronic Pain:
Neuropathic or nociceptive pain from tissue damage and scarring.
*
Incidence:
Variable, can be significant.
*
Salvage:
Multimodal pain management, physical therapy, psychological support, nerve blocks, or surgical interventions.
*
Psychological Distress:
PTSD, depression, anxiety due to critical illness, disfigurement, and prolonged recovery.
*
Incidence:
High.
*
Salvage:
Psychological counseling, support groups, early screening.
*
Malnutrition:
High metabolic demands during infection and healing.
*
Incidence:
High.
*
Salvage:
Early and aggressive nutritional support (enteral or parenteral).
Common Complications of Necrotizing Soft Tissue Infections & Management Strategies
| Complication | Incidence (%) | Salvage/Management Strategies |
|---|---|---|
| Sepsis/Septic Shock | 50-100 | Aggressive fluid resuscitation, vasopressors, broad-spectrum antibiotics, immediate source control (debridement), ventilatory support, renal replacement therapy. |
| Acute Kidney Injury | 30-60 | Maintain euvolemia, avoid nephrotoxic drugs, monitor renal function, consider renal replacement therapy (dialysis) if severe. |
| Multi-Organ Failure | 20-40 | ICU admission, organ-specific support (ventilator for ARDS, dialysis for AKI, vasopressors for shock), aggressive source control. |
| Limb Amputation | 10-25 | Timely and complete debridement, aggressive management of sepsis, adequate perfusion, judicious decision-making when limb salvage is no longer feasible or life-threatening. |
| Recurrent Infection | 5-15 (variable) | Re-exploration and further debridement, re-evaluation of antibiotic regimen based on culture results, ensuring complete source control. |
| Extensive Wound Defect | High (depending on extent) | Negative Pressure Wound Therapy (NPWT) to prepare wound bed, serial dressing changes, early plastic surgery consultation for skin grafting (split-thickness or full-thickness) or reconstructive flaps. |
| Functional Impairment | 20-50 | Early and aggressive physical/occupational therapy, range-of-motion exercises, splinting, scar management, reconstructive surgery (e.g., tendon transfers, contracture release). |
| Chronic Pain | 10-30 | Multimodal pain management (pharmacological, interventional, psychological), nerve blocks, neuropathic pain agents, physical therapy. |
| Psychological Distress | 20-40 | Early psychological assessment, counseling, support groups, pharmacotherapy for depression/anxiety, addressing body image concerns. |
| Malnutrition | 30-50 | Early nutritional assessment, aggressive enteral or parenteral nutrition, dietary counseling, vitamin supplementation. |
Post-Operative Rehabilitation Protocols
Post-operative management of NSTI survivors is a prolonged and multidisciplinary process focused on wound healing, infection control, functional recovery, and psychological support.
1. Wound Care and Infection Control:
*
Serial Debridements:
Continue with scheduled or on-demand serial debridements in the operating room until all necrotic tissue is removed and a healthy, granulating wound bed is achieved.
*
Dressing Changes:
Frequent (daily or twice daily) dressing changes are critical. Options include saline-soaked gauze, antimicrobial dressings, hydrogels, alginates, or negative pressure wound therapy (NPWT). NPWT is particularly useful for large wounds, promoting granulation and managing exudate, often acting as a bridge to definitive closure.
*
Antibiotic Management:
Tailor antibiotics based on intraoperative cultures and sensitivities. Continue for an appropriate duration (typically 10-14 days after the last debridement showing no evidence of infection, or longer for osteomyelitis/septic arthritis).
*
Wound Swabs/Biopsies:
Obtain regular wound cultures, especially if signs of local infection persist.
*
Definitive Wound Closure:
Once the wound bed is clean and granulating, and the infection is controlled, definitive closure can be planned. This may involve:
*
Primary Closure:
For small wounds with minimal tension.
*
Split-Thickness Skin Grafts (STSGs):
Common for large, superficial defects.
*
Full-Thickness Skin Grafts (FTSGs):
For smaller defects requiring better cosmetic or functional outcomes.
*
Local or Free Flaps:
For deeper, more complex defects involving exposed bone, tendon, or critical structures, providing vascularized tissue coverage.
*
Hyperbaric Oxygen Therapy (HBOT):
While controversial as a primary treatment, HBOT can be considered as an adjunctive therapy, particularly in clostridial myonecrosis or severe cases, to improve tissue oxygenation, enhance leukocyte function, and inhibit anaerobic bacterial growth.
2. Physical and Occupational Therapy:
*
Early Mobilization:
As soon as the patient is hemodynamically stable, initiate early mobilization to prevent joint stiffness, muscle atrophy, and deep vein thrombosis.
*
Range of Motion (ROM) Exercises:
Active and passive ROM exercises are crucial to prevent contractures, especially in joints adjacent to surgical sites.
*
Strengthening:
Progressive strengthening exercises for affected limbs or body parts.
*
Splinting and Orthotics:
May be necessary to maintain joint position, prevent contractures, or support weakened limbs.
*
Scar Management:
Early initiation of scar massage, silicone sheeting, and compression garments to minimize hypertrophic scarring and contractures.
*
Functional Retraining:
Retraining for activities of daily living (ADLs), gait training, and adaptive strategies.
3. Nutritional Support:
* NSTI patients are hypercatabolic. Aggressive nutritional support is vital for wound healing and immune function.
*
Enteral Nutrition:
Preferred route if the gastrointestinal tract is functional.
*
Parenteral Nutrition:
If enteral feeding is not tolerated or insufficient.
*
Dietary Counseling:
Provide high-protein, high-calorie diets with adequate micronutrients.
4. Psychological Support:
* Many patients experience post-traumatic stress disorder (PTSD), depression, anxiety, and body image issues.
*
Counseling:
Provide access to mental health professionals, support groups, and peer support.
*
Family Involvement:
Engage family in the recovery process.
5. Long-Term Follow-up:
* Regular follow-up with the orthopedic surgeon, plastic surgeon, physical therapist, and primary care physician is essential for monitoring wound healing, addressing functional deficits, and managing long-term complications.
* Reconstructive procedures may be staged over several months to years.
Summary of Key Literature / Guidelines
The management of NSTIs is guided by a body of literature emphasizing early recognition, aggressive surgical debridement, and comprehensive supportive care. Key guidelines and landmark studies inform current practice:
-
The LRINEC Score:
- Wong et al., 2004: The original study validating the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score, demonstrating its utility as a diagnostic adjunct. A score ≥ 6 had a high positive predictive value (92%) and negative predictive value (96%) for NSTI.
- Subsequent studies have confirmed its utility, but also highlighted its limitations. While a high score necessitates urgent action, a low score does not definitively rule out NSTI, especially in early stages or in specific patient populations (e.g., immunosuppressed). Clinical suspicion remains paramount.
- CRP: Among the LRINEC parameters, elevated C-reactive protein (CRP) is consistently a strong indicator of severe infection and tissue necrosis.
-
Importance of Early Surgical Debridement:
- Numerous retrospective and prospective studies consistently show that delayed surgical debridement (beyond 6-12 hours from diagnosis) is independently associated with increased mortality. For instance, studies by Sudarsky et al. and others have emphasized a "time-to-knife" imperative.
- Aggressiveness: The literature advocates for radical and serial debridement until all necrotic tissue is removed and healthy, bleeding margins are achieved. Incomplete debridement is a major predictor of poor outcomes.
-
Antimicrobial Therapy:
- Broad-Spectrum: Initial empiric therapy should be broad-spectrum, targeting Gram-positive (including MRSA, e.g., vancomycin, linezolid), Gram-negative (e.g., piperacillin-tazobactam, carbapenem), and anaerobic bacteria (e.g., clindamycin, metronidazole).
- Clindamycin: Recommended for Group A Streptococcus (GAS) and Clostridium infections due to its ability to inhibit bacterial toxin production, irrespective of bacterial load.
- Culture-Guided De-escalation: Antibiotics should be de-escalated based on culture and sensitivity results as soon as possible to minimize antimicrobial resistance and adverse effects.
- Duration: Typically 10-14 days, or until clinical resolution and no further need for debridement.
-
Adjunctive Therapies:
- Hyperbaric Oxygen Therapy (HBOT): While some studies suggest HBOT may improve outcomes, particularly in clostridial infections, its precise role in non-clostridial NSTI remains debated due to limited high-quality evidence. It should be considered an adjunct, not a replacement for surgery and antibiotics, and should not delay definitive debridement.
- Intravenous Immunoglobulin (IVIG): Considered for streptococcal toxic shock syndrome (STSS) as it may neutralize bacterial superantigens, but evidence for routine use in all NSTIs is not robust.
-
Critical Care Support:
- Guidelines from organizations like the Society of Critical Care Medicine (SCCM) and Infectious Diseases Society of America (IDSA) emphasize aggressive hemodynamic support, mechanical ventilation, and management of multi-organ dysfunction in the intensive care unit setting.
-
Reconstruction:
- The literature underscores the importance of a reconstructive ladder approach: primary closure, skin grafting, and local or free flaps. Early involvement of plastic surgery can optimize reconstructive outcomes and potentially reduce hospital stay.
- Negative pressure wound therapy (NPWT) is widely accepted for managing large, complex wounds following debridement, promoting granulation, and preparing the wound bed for definitive closure.
In summary, the consensus in the literature emphasizes a rapid, multidisciplinary approach to NSTIs, with the LRINEC score serving as a useful, though not exclusive, diagnostic tool to prompt urgent surgical intervention. The foundation of successful management remains aggressive, serial surgical debridement combined with appropriate broad-spectrum antibiotics and intensive supportive care.
Clinical & Radiographic Imaging