Flexor Hallucis Longus Transfer for Chronic Achilles Tendinosis
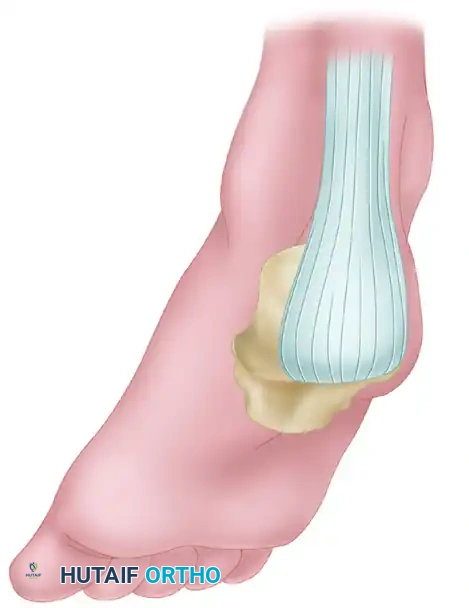
Key Takeaway
The flexor hallucis longus (FHL) transfer is a robust surgical intervention for chronic noninsertional Achilles tendinosis when more than 50% of the tendon is compromised. This procedure restores plantarflexion strength and provides vascularized tissue to the degenerative Achilles. Key steps include aggressive debridement, FHL harvest via a medial or posterior approach, and secure fixation into the calcaneus to optimize postoperative biomechanics and functional recovery.
Comprehensive Introduction and Patho-Epidemiology
Chronic noninsertional Achilles tendinosis represents a profound and debilitating degenerative cascade within the largest and strongest tendon in the human body. Unlike acute inflammatory tendinitis, chronic tendinosis is characterized histologically by angiofibroblastic hyperplasia, mucoid degeneration, and a highly disorganized type III collagen matrix that replaces the normal, robust type I collagen architecture. This degenerative process typically localizes 2 to 6 centimeters proximal to the calcaneal insertion, an anatomical region universally recognized as the vascular watershed area of the Achilles tendon. Within this zone, the intrinsic blood supply derived from the musculotendinous junction and the osseous insertion is inherently tenuous, rendering the tissue exceptionally vulnerable to repetitive microtrauma, hypoxia, and subsequent failed healing responses.
The epidemiological profile of chronic Achilles tendinosis spans a broad spectrum of patients, affecting both high-level athletes and sedentary individuals. While historically associated with middle-aged recreational athletes—often colloquially termed "weekend warriors"—the incidence has steadily risen across diverse demographics. Systemic risk factors play a critical role in the pathogenesis and exacerbation of this condition. Prolonged exposure to systemic or local corticosteroids, the administration of fluoroquinolone antibiotics, hyperlipidemia, inflammatory arthropathies, and poorly controlled diabetes mellitus dramatically increase the susceptibility to tendon degeneration and eventual rupture. These metabolic and pharmacological factors disrupt tenocyte metabolism and collagen cross-linking, further compromising the structural integrity of the watershed zone.
When non-operative modalities—encompassing eccentric loading protocols (such as the Alfredson protocol), extracorporeal shockwave therapy (ESWT), platelet-rich plasma (PRP) injections, and prolonged immobilization—fail to yield symptomatic relief after a period of six months, surgical intervention becomes the definitive standard of care. For mild to moderate localized degeneration, simple longitudinal tenotomy, aggressive debridement of the tendinotic core, and tubularization of the remaining healthy tendon may suffice. However, a critical biomechanical threshold is breached when the necessary debridement results in a defect comprising more than 50% of the tendon's cross-sectional area. At this juncture, the residual tendon is biomechanically insufficient to withstand the immense forces of normal ambulation, which can exceed three to four times body weight during the push-off phase of gait.
In these advanced, severe cases of tendinosis, or in the setting of chronic neglected ruptures with substantial gap formation, augmentation with a tendon transfer is strictly indicated to restore the plantarflexion moment arm. The Flexor Hallucis Longus (FHL) has emerged as the unequivocal gold standard for Achilles augmentation. It is overwhelmingly preferred over alternative transfers, such as the flexor digitorum longus (FDL) or the peroneus brevis, due to a convergence of highly favorable anatomical and biomechanical factors. The FHL boasts a superior cross-sectional area, an in-phase firing pattern during the gait cycle that mimics the triceps surae, and, crucially, a highly vascularized, low-lying muscle belly. This muscle belly acts as a vascularized pedicle graft when transposed into the Achilles defect, providing an essential neo-angiogenic blood supply to the avascular, degenerative watershed zone, thereby facilitating robust biological healing.
Detailed Surgical Anatomy and Biomechanics
A profound, three-dimensional understanding of the posterior ankle, deep posterior compartment of the leg, and plantar midfoot anatomy is absolutely essential for the safe harvest, routing, and transfer of the Flexor Hallucis Longus. The FHL is a bipennate muscle that originates extensively from the inferior two-thirds of the posterior surface of the fibula, the adjacent interosseous membrane, and the transverse intermuscular septum. Uniquely among the deep posterior compartment musculature, the FHL muscle belly extends far distally, often reaching the level of the tibiotalar joint line and occasionally extending into the fibro-osseous tunnel posterior to the medial malleolus. This low-lying muscle belly is the cornerstone of its utility as a transfer, providing a rich, intrinsic vascular bed that can be sutured directly into the debrided Achilles defect.
As the FHL tendon courses distally, it traverses the posterior aspect of the ankle joint, lying in a dedicated sulcus between the medial and lateral tubercles of the posterior talar process. It then passes beneath the sustentaculum tali of the calcaneus before entering the plantar aspect of the foot. In the plantar midfoot, the anatomical relationship between the FHL and the FDL becomes surgically critical at the Master Knot of Henry. Located deep to the abductor hallucis muscle and the plantar fascia, the Master Knot of Henry is the anatomical decussation where the FHL tendon crosses dorsal (deep) to the FDL tendon. At this juncture, fibrinous slips typically connect the two tendons, which must be meticulously released during a distal harvest to allow proximal excursion of the FHL graft.
The neurovascular topography surrounding the FHL dictates the surgical approach and the boundaries of safe dissection. Proximally, within the deep posterior compartment, the posterior tibial artery and tibial nerve lie medial to the FHL muscle belly. During posterior dissection, straying medially risks catastrophic injury to this primary neurovascular bundle. Distally, at the level of the Master Knot of Henry, the medial plantar nerve and artery are situated immediately deep and lateral to the FHL tendon. The margin of error in this region is measured in millimeters; iatrogenic retraction or sharp dissection injury here can lead to devastating, permanent plantar numbness, debilitating neuromas, and localized vascular compromise to the medial plantar aspect of the foot.
Biomechanically, the FHL is uniquely suited to augment or replace the Achilles tendon. It is the second strongest plantarflexor of the ankle, subordinate only to the triceps surae complex. Isokinetic studies demonstrate that the FHL possesses approximately 30% of the cross-sectional area and work capacity of the native Achilles tendon, making it significantly stronger than the FDL or the peroneus brevis. Furthermore, the FHL is an in-phase synergist; it naturally fires during the stance and push-off phases of the gait cycle. This innate motor programming eliminates the need for extensive postoperative cortical re-education, allowing for a seamless and intuitive integration of the transferred muscle into the patient's normal biomechanical kinetic chain. Its axis of pull closely parallels that of the Achilles, minimizing sheer stresses and optimizing the mechanical advantage for ankle plantarflexion.
Exhaustive Indications and Contraindications
The decision to proceed with a Flexor Hallucis Longus transfer must be predicated on a rigorous evaluation of the patient's pathology, functional demands, and systemic health. The primary indication for this procedure is chronic noninsertional Achilles tendinosis where intraoperative debridement is anticipated to, or inherently does, resect greater than 50% of the tendon's cross-sectional area. In such scenarios, primary tubularization is doomed to mechanical failure, and vascular augmentation is required to heal the remaining compromised tissue. Additionally, the FHL transfer is the procedure of choice for chronic, neglected Achilles tendon ruptures presenting with a defect gap greater than 3 centimeters that cannot be opposed with maximal ankle equinus.
Revision Achilles tendon surgery constitutes another major indication. Patients who have failed previous primary repairs, minimally invasive interventions, or V-Y fascial advancements often present with severely compromised local tissue envelopes, extensive scarring, and devascularized tendon stumps. The introduction of the FHL provides not only structural bridging but also introduces virgin, highly vascularized tissue into a hostile, scarred bed. Furthermore, in older, lower-demand patients presenting with acute ruptures in the setting of profound pre-existing tendinosis, primary FHL augmentation may be considered to mitigate the high risk of re-rupture and to stimulate healing in an otherwise biologically inert environment.
Conversely, absolute contraindications must be strictly respected to avoid catastrophic postoperative complications. Active localized or systemic infection precludes any tendon transfer or implantation of hardware. Severe peripheral vascular disease, characterized by absent pedal pulses or an ankle-brachial index (ABI) less than 0.5, represents an absolute contraindication, as the surgical incisions—particularly the posterior longitudinal approach—are highly prone to necrosis and dehiscence in the ischemic limb. Charcot neuroarthropathy of the hindfoot or midfoot is similarly a contraindication, as the altered biomechanics and loss of protective sensation will inevitably lead to transfer failure or progressive midfoot collapse.
Relative contraindications require nuanced clinical judgment. Patients with a profound inability or unwillingness to comply with a strict, months-long postoperative rehabilitation protocol are poor candidates, as premature weight-bearing will rupture the transfer, while prolonged immobilization without therapy will result in irreversible arthrofibrosis. Pre-existing hallux rigidus or severe first metatarsophalangeal joint arthritis should be carefully evaluated; while FHL transfer removes the dynamic deforming force on the hallux, the loss of active hallux plantarflexion might exacerbate gait abnormalities in patients already compensating for first ray pathology.
| Category | Indications | Contraindications |
|---|---|---|
| Absolute | - Chronic tendinosis requiring >50% debridement - Chronic rupture with >3cm gap unable to be opposed - Revision Achilles reconstruction with devascularized bed |
- Active local or systemic infection - Severe peripheral vascular disease (ABI < 0.5) - Active Charcot neuroarthropathy |
| Relative | - Acute rupture in elderly patient with severe pre-existing tendinosis - Insertional tendinosis requiring complete detachment of Achilles |
- Inability to comply with strict postoperative rehabilitation - Severe pre-existing hallux rigidus/arthritis - Morbid obesity compromising posterior wound healing |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous pre-operative planning begins with a comprehensive clinical examination. The orthopedic surgeon must assess the resting tension of the Achilles complex, noting any palpable gaps or severe nodular thickening. The Thompson test should be performed, though it may be falsely negative in chronic cases where thick, non-functional scar tissue bridges the rupture gap. Crucially, the surgeon must evaluate and document the baseline strength of hallux plantarflexion. Informing the patient pre-operatively about the anticipated, albeit usually well-tolerated, loss of great toe push-off strength is a mandatory component of the informed consent process.
Advanced imaging is non-negotiable in the workup of chronic Achilles pathology. Weight-bearing radiographs of the foot and ankle are obtained to rule out concomitant osseous pathology, such as a Haglund's deformity, insertional calcific tendinosis, or underlying pes planovalgus that might alter the biomechanical axis of the hindfoot. Magnetic Resonance Imaging (MRI) without contrast is the gold standard for soft tissue templating. Axial and sagittal T1 and T2-weighted sequences are utilized to precisely quantify the proximal-to-distal extent of the tendinosis, measure the exact gap in chronic ruptures, and assess the degree of peritendinous inflammation. Furthermore, the MRI must be scrutinized to evaluate the quality of the FHL muscle belly itself; severe fatty infiltration or atrophy of the FHL precludes its use as a functional transfer.
In the operating theater, the administration of a satisfactory general anesthetic or a regional neuraxial block (spinal or epidural) is standard, often supplemented with a continuous popliteal sciatic nerve block for postoperative analgesia. The patient is carefully transitioned into the prone position on a radiolucent operating table. Meticulous attention must be paid to padding all bony prominences, particularly the face, ulnar nerves at the elbows, and the contralateral knee and toes, to prevent pressure necrosis and neuropraxias during the potentially lengthy procedure. A high thigh pneumatic tourniquet is applied to provide a pristine, bloodless surgical field, which is critical for identifying the delicate neurovascular structures during the deep dissection.
The operative limb is prepped and draped in a sterile fashion from the tips of the toes to the proximal thigh. This extensive draping is a critical technical point; it allows the surgeon to freely manipulate the knee and ankle joints throughout the procedure. The ability to flex the knee to 90 degrees and assess the resting equinus tension of the foot—comparing it intraoperatively to the contralateral limb if necessary—is paramount for setting the correct tension on the FHL graft during final fixation.
Step-by-Step Surgical Approach and Fixation Technique
Incision and Achilles Exposure
The procedure commences with a longitudinal incision placed just medial to the medial border of the Achilles tendon, typically measuring 10 to 12 centimeters in length, centered directly over the nodular, diseased section of the tendon. A medial approach is deliberately chosen to avoid the sural nerve and the lesser saphenous vein, which course along the posterolateral aspect of the ankle. Dissection is carried sharply through the subcutaneous tissues down to the paratenon.
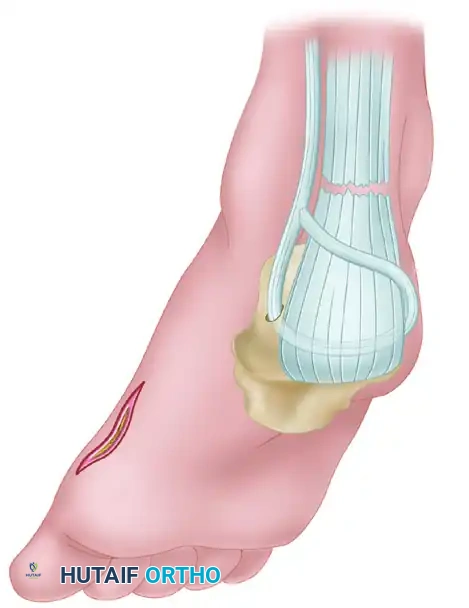
The paratenon is a highly vascularized, delicate fascial layer that must be incised longitudinally and preserved with the utmost care. It is gently elevated off the underlying diseased Achilles tendon and retracted medially and laterally. Preservation of this layer is non-negotiable, as it will be meticulously closed over the final repair to provide a gliding surface and prevent the overlying skin from tethering directly to the tendon transfer. Any hypertrophic, inflammatory peritendinitis or dense adhesions are carefully removed using a rongeur or sharp dissection. A double skin hook retractor is utilized to retract the medial border of the Achilles posteriorly, affording excellent visualization of the deep, anterior surface of the tendon where the degeneration is often most profound.
Achilles Debridement
The core of the tendinotic Achilles is systematically and sharply debrided using a scalpel. The surgeon must resect all pathological tissue until healthy, normal-appearing tendon is encountered proximally and distally. Healthy Achilles tendon is readily identifiable by its pearly white appearance, firm texture, and tightly packed, parallel type I collagen fibers. In stark contrast, the tendinotic tissue presents as dull, yellowish, friable, and grossly disorganized, often containing areas of mucoid degeneration or focal necrosis. The "50% Rule" is strictly applied here: if the thorough debridement leaves a defect encompassing less than 50% of the tendon's cross-sectional area, the remaining healthy tendon is simply tubularized with interrupted 2-0 braided, nonabsorbable sutures. However, if the resection exceeds this critical 50% threshold, the structural integrity is compromised, and the surgeon must immediately proceed with the FHL harvest.
FHL Identification and Midfoot Harvest
To access the FHL proximally, a longitudinal incision is made through the deep fascia of the posterior compartment, directly anterior to the debrided Achilles tendon. The numerous small veins accompanying the muscle in this region must be meticulously cauterized. The interval between the FHL medially and the peroneal tendons laterally is developed. During this deep dissection, the surgeon must maintain a strict lateral bias relative to the FHL tendon to avoid any iatrogenic injury to the posterior tibial neurovascular bundle, which lies dangerously close on the medial aspect.
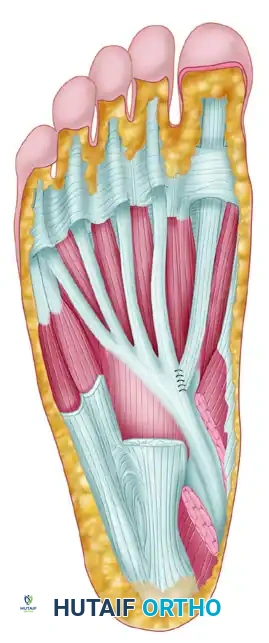
Once the FHL is isolated posteriorly, attention shifts to the medial midfoot to secure maximum graft length. A separate 4 to 5-centimeter longitudinal incision is made over the medial aspect of the foot, following the dorsal arch of the abductor hallucis muscle. The deep fascia is incised, and the abductor hallucis muscle is retracted plantarly to expose the deep plantar structures. The Master Knot of Henry is identified proximally. At this critical juncture, meticulous blunt dissection is mandatory. The medial plantar nerve and artery are identified just deep and lateral to the FHL tendon and gently protected with a vessel loop or blunt retractor.
Tenodesis of FHL to FDL
Before the FHL is transected, its distal stump must be secured to prevent the development of a postoperative hallux malleus (claw toe) and to mitigate the loss of great toe push-off strength. With the interphalangeal and metatarsophalangeal joints of the hallux held in neutral to slight plantarflexion, the FHL tendon is sutured side-to-side to the adjacent FDL tendon.

This tenodesis is performed using multiple interrupted 2-0 Vicryl (polyglactin 910) or equivalent absorbable sutures. This vital step ensures that the flexor digitorum longus will dynamically drive hallux plantarflexion during the terminal stance phase of gait.
Tendon Delivery and Transfer
Following secure tenodesis, the FHL tendon is sharply transected just proximal to the suture line. The surgeon must then systematically release all fibrinous connections, fascial slips, and anomalous interconnections between the FHL and FDL within the midfoot to allow unhindered proximal excursion. The free end of the FHL tendon is then grasped with a hemostat and delivered proximally into the primary posterior calf incision. For noninsertional Achilles tendinosis with an intact calcaneal insertion, the FHL muscle belly and tendon are laid directly into the trough created by the Achilles debridement. The low-lying muscle belly is sutured into the proximal Achilles stump, providing a rich, vascularized pedicle to drive neo-angiogenesis, while the tendon is woven through the distal Achilles stump to act as a biomechanical bridge.
Calcaneal Fixation
In cases of severe insertional tendinosis requiring complete detachment, or in chronic ruptures where the distal stump is entirely absent, the FHL must be anchored directly into the calcaneus. The dorsal surface of the calcaneal tuberosity is exposed and decorticated to a bleeding bone bed. A transosseous tunnel is created using successively larger drill bits, typically aiming for a 3/8-inch (approx. 7-8mm) transverse or dorsal-to-plantar tunnel to accommodate the FHL graft.
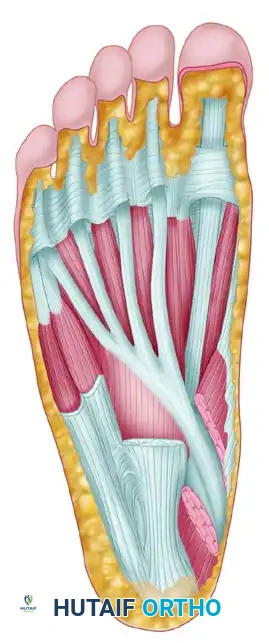
The FHL tendon is woven through the remaining proximal Achilles tendon stump using a Pulvertaft weave technique, maximizing the surface area of tendon-to-tendon contact for optimal biological healing. The distal end of the FHL is then passed through the calcaneal bone tunnel. Tensioning the graft is the most critical biomechanical step of the procedure. The knee is flexed to 90 degrees, and the ankle is positioned in moderate equinus (approximately 15 to 20 degrees of plantarflexion). The graft is pulled taut to replicate the resting tension of the native Achilles. It is imperative to ensure that the ankle can be passively brought to neutral after fixation, but no further, to prevent over-lengthening. The tendon is sutured onto itself using interrupted No. 2 Ethibond or equivalent heavy, nonabsorbable braided sutures. Alternatively, a highly secure fixation can be achieved using an interference-type absorbable biocomposite screw placed directly into a dorsal-to-plantar calcaneal tunnel, yielding superior biomechanical pull-out strength.
Closure
Following meticulous hemostasis and copious irrigation with sterile saline, the closure begins. The preserved paratenon is drawn over the FHL transfer and repaired with interrupted 2-0 absorbable sutures. This layer is vital for preventing cutaneous tethering. The subcutaneous tissue is closed with 3-0 absorbable sutures, and the skin is reapproximated using a 3-0 or 4-0 nonabsorbable nylon or prolene suture in a vertical mattress fashion to minimize tension on the fragile posterior skin edges. A sterile, non-adherent dressing is applied, followed by a well-padded short leg splint with the ankle locked in 15 to 20 degrees of equinus.
Complications, Incidence Rates, and Salvage Management
Despite the high success rate of the FHL transfer, the procedure is technically demanding and carries specific, potentially severe risks. The orthopedic surgeon must be acutely aware of these complications, employing rigorous preventative strategies and possessing a clear algorithm for salvage management.
Neurologic injuries are among the most feared complications. Sural nerve injury can occur during the primary posterior approach if the incision or deep dissection drifts laterally. This results in numbness along the lateral border of the foot and the potential for a highly symptomatic neuroma. Medial plantar nerve injury is an even more devastating complication, carrying a higher risk of occurrence during the midfoot harvest at the Master Knot of Henry. Injury here leads to profound numbness of the plantar medial foot, intractable pain, and gait alterations.
Wound complications are notoriously common in posterior ankle surgery due to the tenuous, watershed vascularity of the skin overlying the Achilles tendon. Wound dehiscence, marginal skin necrosis, and deep surgical site infections can rapidly compromise the tendon transfer. Over-lengthening of the Achilles-FHL complex is a catastrophic biomechanical failure. If the graft is secured with the ankle in excessive dorsiflexion, or if the patient bears weight prematurely, the complex will heal in an elongated position. This results in a permanent calcaneus gait, profound push-off weakness, and an inability to perform a single-leg heel raise.
| Complication | Estimated Incidence | Prevention Strategy | Salvage Management |
|---|---|---|---|
| Sural Nerve Injury | 2 - 5% | Maintain strictly medial paratendinous incision; avoid lateral retraction. | Neuroma excision and proximal burying of the nerve stump into deep muscle. |
| Medial Plantar Nerve Injury | 1 - 3% | Meticulous blunt dissection at the Master Knot of Henry; direct visualization. | Neurolysis; gabapentinoids; specialized orthotics to offload insensate areas. |
| Wound Dehiscence / Necrosis | 5 - 10% | Preserve paratenon; layered, tension-free closure; strict postoperative elevation. | Vacuum-assisted closure (VAC); local rotational flaps; free tissue transfer (e.g., anterolateral thigh flap) for massive defects. |
| Hallux Malleus | 5 - 8% | Secure tenodesis of distal FHL stump to FDL in neutral/slight plantarflexion. | Flexor-to-extensor transfer (Girdlestone-Taylor procedure) or interphalangeal joint arthrodesis. |
| Over-lengthening / Weakness | 3 - 6% | Tension graft in 15-20° equinus; strict adherence to NWB protocols. | Revision surgery with FHL advancement or alternative augmentation (e.g., peroneus brevis); custom AFO bracing. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation protocol following an FHL transfer is a delicate, meticulously timed balance between protecting the mechanical integrity of the tendon transfer and preventing irreversible arthrofibrosis, muscle atrophy, and deep vein thrombosis. The protocol is strictly phased, guided by the biological phases of tendon healing.
Phase 1: Maximum Protection (Weeks 0 to 4)
The primary objective during the first four weeks is the absolute protection of the surgical repair and the optimization of wound healing. Immediately postoperatively, the patient is placed in a rigid, non-weight-bearing (NWB) short leg cast or a heavy posterior splint with the ankle immobilized in 15 to 20 degrees of equinus. This position maximally unloads the Achilles-FHL complex. Strict elevation of the operative limb above the level of the heart is mandatory for the first 7 to 10 days to mitigate dependent edema, which is the primary driver of wound dehiscence. At the 2-week mark, the cast is bi-valved, the wound is meticulously inspected, and sutures are removed. Assuming normal wound healing, the patient is immediately placed back into a NWB cast or a locked rigid boot in the same degree of equinus.
Phase 2: Progressive Weight-Bearing (Weeks 4 to 8)
At the 4-week juncture, the biological bridging of the tendon transfer is sufficient to begin controlled, progressive loading, which stimulates proper collagen alignment. The patient is transitioned to a prefabricated controlled ankle motion (CAM) walking brace. To maintain the protective equinus position, multiple rigid heel lifts (totaling approximately 2.5 to 3 inches in height) are inserted into the boot. The patient initiates progressive partial weight-bearing with the assistance of crutches. The protocol dictates the removal of one wedge of the heel lift every 5 to 7 days. As the wedges are removed, the ankle gradually stretches out of equinus. By the end of week 8, all wedges should be removed, the ankle should rest in a neutral position within the CAM boot, and the patient should be fully weight-bearing.
Phase 3: Range of Motion and Strengthening (Weeks 8 to 14)
Formal, supervised physical therapy commences at week 8. The CAM boot is transitioned to a supportive, rigid-soled athletic shoe fitted with a small (1/4 inch) silicone heel cup to reduce impact stress. Therapy focuses initially on restoring active and active-assisted range of motion. Gentle, progressive calf stretching is initiated, utilizing towel stretches and slant boards, but aggressive, forceful passive dorsiflexion is strictly avoided to prevent stretching the transfer. Strengthening begins with isometric plantarflexion exercises against resistance bands. As tolerance improves, the patient progresses to concentric bilateral heel raises, eventually advancing to unilateral concentric and, finally, controlled eccentric loading protocols.
Phase 4: Return to Activity (3.5 Months and Beyond)
The final phase focuses on functional restoration and return to pre-injury activity levels. Gradual return to low-impact, linear activities such as stationary cycling, elliptical training, and swimming is permitted around the 3.5 to 4-month mark. High-impact activities, distance running, and sports involving explosive push-off, jumping, or sudden changes of direction (e.g., basketball, tennis) are strictly delayed until 5 to 6 months postoperatively. Clearance for unrestricted sports is contingent upon rigorous objective criteria: the complete resolution of pain, full functional range of motion, and the recovery of at least 80% to 85% of the contralateral limb's plantarflexion strength, ideally measured via isokinetic dynamometry.
Summary of Landmark Literature and Clinical Guidelines
The evolution of the Flexor Hallucis Longus transfer for Achilles tendinosis is deeply rooted in robust clinical and biomechanical literature. The procedure was popularized and standardized by Wapner and colleagues in the early 1990s, who first detailed the elegant surgical technique of utilizing the FHL muscle belly as a vascularized pedicle to address the avascular nature of chronic Achilles ruptures. Their landmark studies demonstrated excellent clinical outcomes and established the FHL as the superior transfer option over the peroneus brevis, which alters hindfoot eversion mechanics, and the F
