INTRODUCTION TO NEUROMUSCULAR DIFFERENTIATION
In the realm of orthopaedic surgery, the clinical presentation of hypotonia, progressive weakness, and musculoskeletal deformity frequently necessitates a definitive differentiation between primary muscle disease (myopathy) and primary nerve disease (neuropathy). Establishing an accurate diagnosis is paramount, as the natural history, prognosis, and surgical management algorithms—such as tendon transfers, contracture releases, and spinal deformity corrections—differ drastically between the two etiologies.
While a meticulous history and physical examination form the foundation of clinical assessment, definitive differentiation relies on a sophisticated triad of diagnostic modalities: hematological and molecular studies, electromyography (EMG) with nerve conduction velocities (NCV), and histopathological analysis via muscle tissue biopsy.
HEMATOLOGICAL AND MOLECULAR DIAGNOSTICS
Serum Enzyme Assays
Serum enzyme assays, particularly the measurement of serum creatine kinase (CK), serve as the first-line biochemical indicators of striated muscle function and integrity. CK is a highly sensitive marker for sarcolemmal breakdown and muscle necrosis.
- Duchenne and Becker Muscular Dystrophy: In the early stages of Duchenne muscular dystrophy (DMD), CK levels are profoundly elevated, often reaching 20 to 200 times above normal physiological limits. This elevation directly parallels the volume of active muscle necrosis. As the disease progresses and functional muscle mass is progressively replaced by fibrofatty tissue, the absolute level of CK paradoxically declines. While CK levels are generally higher in DMD compared to Becker muscular dystrophy (BMD), significant overlap exists.
- Carrier Detection: CK assays are instrumental in identifying female carriers of DMD and BMD. A muscle provocation test—involving strenuous exercise prior to serum collection—can unmask the carrier state, as carrier females exhibit a significantly greater spike in CK levels post-exercise compared to non-carrier females.
- Other Enzymes: Aldolase follows a trajectory similar to CK and is reliably elevated in muscular dystrophies. Aspartate aminotransferase (AST) and lactate dehydrogenase (LDH) may also be elevated; however, their lack of specificity renders them less diagnostically valuable in isolation.
Creatine Metabolism
Urine creatine excretion is markedly excessive in dystrophic patients during active stages of muscle catabolism. However, this finding is non-specific, as any process inducing muscle breakdown—including excessive exercise, uncontrolled diabetes mellitus, starvation, and severe neuropathies—can precipitate creatinuria. Conversely, in myotonic dystrophy, blood creatine levels are characteristically decreased due to the liver's impaired ability to synthesize creatine phosphate.
Molecular and Genetic Profiling
Advancements in molecular biology have revolutionized the diagnostic algorithm. DNA mutation analysis, utilizing polymerase chain reaction (PCR) or Southern blot analysis, provides a definitive diagnosis for DMD and BMD by identifying deletions, duplications, or point mutations in the dystrophin gene located on the Xp21 chromosome. These minimally invasive tests, requiring only a small peripheral blood sample or amniotic fluid, are also the gold standard for carrier identification and prenatal diagnosis.
ELECTROPHYSIOLOGICAL EVALUATION (EMG AND NCV)
Electromyography (EMG) and nerve conduction velocity (NCV) studies are indispensable for localizing the lesion to the anterior horn cell, peripheral nerve, neuromuscular junction, or the muscle fiber itself.
💡 Clinical Pearl: The Electromyographic Triad
When evaluating EMG results, the orthopaedic surgeon must analyze three primary parameters of the motor unit action potential (MUAP): frequency, amplitude, and duration.
- Normal Muscle: At rest, normal skeletal muscle is electrically silent. Upon voluntary contraction, the EMG demonstrates MUAPs with characteristic, predictable frequency, duration, and amplitude.
- Myopathy: In primary muscle disease, the random destruction of muscle fibers within a motor unit leads to a classic EMG signature: increased frequency, decreased amplitude, and decreased duration of the motor action potentials. Because the nerve itself is intact, nerve conduction velocities (NCV) remain normal.
- Neuropathy: In primary nerve disease, denervation is followed by collateral sprouting and reinnervation of orphaned muscle fibers by surviving axons. This creates giant motor units. The resulting EMG shows decreased frequency, increased amplitude, and increased duration of the action potentials. Furthermore, NCVs are typically slowed due to demyelination or axonal loss.
- Myotonic Dystrophy: This condition presents a unique EMG phenomenon. Needle insertion provokes a sustained run of high-frequency, high-amplitude action potentials that gradually wax and wane. When amplified through an audio monitor, this creates the pathognomonic "dive-bomber" sound.
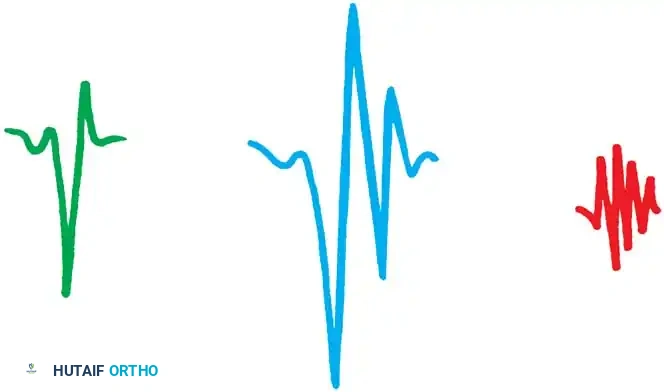
Fig. 32-1 Motor units seen in electromyography. A, Normal triphasic motor unit potential. B, Large polyphasic motor units as seen in neurogenic disorders, such as spinal muscular atrophy, in which they also are reduced in number. C, Small polyphasic motor units as seen in muscular dystrophy. These usually are of normal number. (Courtesy of Tulio E Bertorini, MD.)
MUSCLE TISSUE BIOPSY: PRINCIPLES AND HISTOPATHOLOGY
The interpretation of a meticulously harvested muscle biopsy remains the definitive method for differentiating myopathy from neuropathy, as well as sub-classifying various congenital dystrophies.
Histopathological Hallmarks
Standard hematoxylin and eosin (H&E) staining is supplemented by specialized techniques, including the Gomori modified trichrome stain, nicotinamide adenine dinucleotide-tetrazolium reductase (NADH-TR) stain, and alizarin red S stain. Electron microscopy is frequently employed for ultrastructural analysis.
- Normal Skeletal Muscle: Exhibits a mosaic pattern of polygonal myofibrils with a normal distribution of type I (slow-twitch, oxidative) and type II (fast-twitch, glycolytic) fibers. The normal physiological ratio of type I to type II fibers is approximately 1:2.
- Myopathic Muscle: Histopathology reveals profound architectural disruption. Key features include increased endomysial and perimysial fibrosis, necrosis of muscle fibers, and subsequent fibrofatty replacement. Fibers appear rounded rather than polygonal, with hyaline and granular degeneration. A hallmark of regeneration is the centralization of nuclei (internalized nuclei). Inflammatory cell infiltrates may be present, becoming markedly prominent in inflammatory myopathies like polymyositis. Histochemical stains often reveal a pathological preponderance of type I fibers. Fiber splitting and intracellular calcium accumulation are also frequently observed.
- Neuropathic Muscle: The microscopic landscape is starkly different. There is little to no increase in fibrous connective tissue. The defining feature is the presence of small, angular, atrophic fibers clustered between groups of normal-sized or hypertrophic muscle fibers (fiber type grouping). This grouping occurs due to the reinnervation of adjacent denervated fibers by a single surviving axon. Special stains typically demonstrate a massive preponderance (up to 80%) of type II fibers.

Fig. 32-2 A, Normal muscle biopsy specimen. Note polygonal shape of myofibrils, normal distribution of type I and type II fibers, and normal connective tissue. B, Muscular dystrophy. Fibers are more rounded, some have internalized nuclei, and others are atrophic. Connective tissue is increased. C, Chronic neurogenic atrophy. Notice grouping of fibers of the same type and some atrophic angular fibers. Fat is increased between muscle fascicles. (Courtesy of Tulio E Bertorini, MD.)
Preoperative Planning and Biopsy Site Selection
The diagnostic yield of a muscle biopsy is entirely dependent on selecting the appropriate muscle.
⚠️ Surgical Warning: Site Selection Pitfalls
Never biopsy a muscle that is profoundly weak or totally involved (end-stage). Such specimens yield only non-diagnostic fibrofatty tissue. Conversely, do not biopsy a completely normal muscle. Target a muscle with mild to moderate clinical involvement (Medical Research Council grade 4 or 3).
- Avoid the Gastrocnemius: In DMD, the gastrocnemius is involved very early and rapidly undergoes pseudohypertrophy and fibrofatty replacement, making it a notoriously poor biopsy site.
- Preferred Sites: The quadriceps (specifically the vastus lateralis at the midthigh) and the rectus abdominis are the most reliable sites. They typically show early pathological changes without total replacement of the muscle spindles.
- Avoid Artifacts: The selected muscle must not have been violated by an EMG needle within the past month, as needle tracts cause focal necrosis and inflammation that mimic myopathy. Furthermore, local anesthetic must never be infiltrated directly into the muscle belly being biopsied, as it induces severe histological artifacts.
SURGICAL TECHNIQUE: OPEN MUSCLE BIOPSY
Open muscle biopsy is the traditional gold standard, providing a robust tissue sample for comprehensive histochemical, immunohistochemical, and electron microscopic analysis.
Anesthesia and Positioning
While regional block anesthesia can be utilized, general anesthesia is often required, particularly in pediatric populations.
🚨 CRITICAL ANESTHESIA WARNING: Malignant Hyperthermia
Patients with underlying myopathies (especially central core disease and Duchenne muscular dystrophy) are at a significantly elevated risk for Malignant Hyperthermia (MH) and anesthesia-induced rhabdomyolysis. Depolarizing muscle relaxants (e.g., succinylcholine) and volatile inhalation anesthetics (e.g., halothane, isoflurane) are strictly contraindicated. Total Intravenous Anesthesia (TIVA) is the safest approach.
Step-by-Step Surgical Approach (Technique 32-1)
- Preparation and Incision: Administer a regional block or local infiltration using 1% lidocaine (strictly into the skin and subcutaneous tissue only, avoiding the fascia and muscle). Make a 1.5-cm longitudinal incision following Langer's lines over the selected muscle belly (e.g., vastus lateralis).
- Fascial Dissection: Dissect sharply through the subcutaneous fat. Carefully incise and split the enveloping deep fascia to clearly expose the underlying muscle fascicles.
- Securing the Specimen (Isometric Clamping): It is imperative to maintain the muscle fibers at their resting physiological length to prevent contraction artifact upon excision.
- Method A: Apply a specialized double-bladed muscle biopsy clamp (e.g., Rayport clamp) or two hemostats bound together across a bundle of muscle fibers.
- Method B: Alternatively, place two parallel silk sutures approximately 2 cm apart along the axis of the muscle fibers, tying them gently to secure the bundle.
- Excision: Using a sharp scalpel (No. 15 blade), section the muscle bundle around the outside of the clamp arms or outside the placed sutures. Do not use electrocautery near the specimen, as thermal injury destroys the tissue architecture.
- Hemostasis and Closure: Achieve meticulous hemostasis within the muscle bed using gentle pressure or pinpoint bipolar cautery. Close the fascia with absorbable sutures and the skin with subcuticular sutures or adhesive strips.
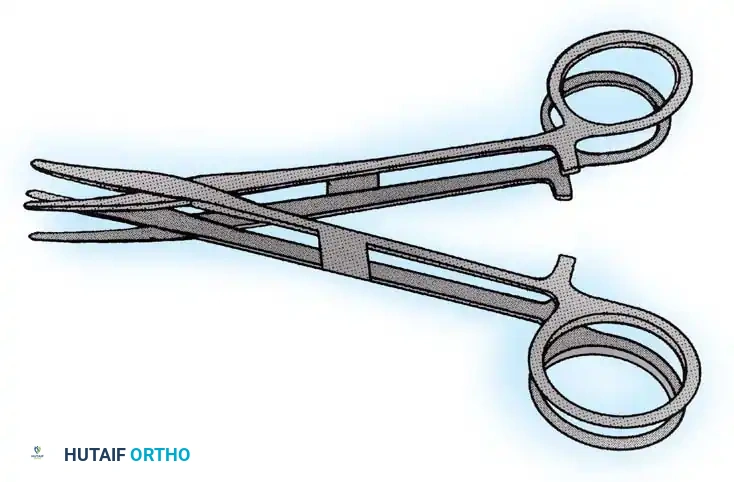
Fig. 32-3 Two hemostats bound together to preserve length when securing muscle biopsy.
Fig. 32-4 Muscle length maintained by muscle biopsy done on outer side of previously placed sutures. (Redrawn from Curtis B: Orthopaedic management of muscular dystrophy and related disorders, Instr Course Lect 19:78, 1970.)
Specimen Handling and Dystrophin Analysis
The excised specimen must be handled with extreme care. It should not be placed in standard formalin if histochemical or enzymatic studies are required. The pathologist must be notified in advance so that fresh tissue can be immediately snap-frozen in isopentane cooled by liquid nitrogen.
A secondary sample of muscle tissue must be harvested concurrently and sent specifically for dystrophin analysis via immunoblotting. Dystrophin, a critical structural protein linking the cytoskeleton to the extracellular matrix, is absent in Duchenne dystrophy and altered/decreased in Becker dystrophy. Quantification of this protein is a cornerstone of modern neuromuscular diagnosis.
SURGICAL TECHNIQUE: PERCUTANEOUS NEEDLE BIOPSY
Percutaneous muscle biopsy, popularized by Mubarak, Chambers, and Wenger, offers a minimally invasive alternative to open biopsy. It is particularly advantageous as it can be performed in an outpatient clinic setting under local anesthesia, minimizing anesthetic risks and reducing surgical morbidity.
Indications and Equipment
This technique is ideal for obtaining samples for standard histology, histochemistry, and biochemical assays. It utilizes a specialized Bergström biopsy needle, which consists of a hollow outer cannula with a cutting window and an inner sharp-edged trocar. Biopsy needles must have a minimal core diameter of 3 mm to ensure an adequate sample size.
Step-by-Step Surgical Approach (Technique 32-2)
- Preparation: Prepare the biopsy site (typically the midthigh over the rectus femoris or vastus lateralis) with an iodophor paint and apply a fenestrated adhesive drape.
- Local Anesthesia: Infiltrate the skin and subcutaneous tissue with 5 to 8 mL of 1% lidocaine without epinephrine. Crucially, when biopsying the quadriceps, ensure the fascia is adequately anesthetized, but avoid infiltrating the muscle belly itself.
- Equipment Check: Verify the Bergström biopsy needle to ensure smooth sliding of the inner cutter within the outer trocar. Cut a K-50 extension tube at an angle and attach it to the proximal end of the cutting needle; attach the other end to a 10-mL syringe to serve as a vacuum source.
- Incision: Use a No. 11 scalpel blade to make a small (3-5 mm) stab wound through the skin and fascia lata.
- Insertion: Insert the Bergström needle assembly into the muscle belly at an oblique angle to align with the muscle fibers.
- Tissue Aspiration: Withdraw the inner cutting needle approximately one-half of its length. Instruct an assistant to apply vigorous suction using the attached 10-mL syringe. This negative pressure draws the muscle tissue into the cutting chamber window of the outer cannula.
- Tissue Amputation: Swiftly compress the inner cutter down into the trocar, shearing off the aspirated muscle tissue.
- Specimen Retrieval: Withdraw the entire Bergström apparatus from the thigh. Carefully extract the muscle sample from the chamber using a fine needle or forceps, and place it immediately on saline-soaked gauze in a Petri dish.
- Multiple Passes: Through the exact same skin incision and fascial track, reinsert the needle. Rotate the needle window to a different quadrant and repeat the aspiration/cutting sequence until five or six adequate samples have been harvested.
- Closure: Apply direct pressure to achieve hemostasis. Close the small stab wound with 1/4-inch adhesive strips (Steri-Strips).
POSTOPERATIVE PROTOCOLS AND COMPLICATION MANAGEMENT
Postoperative care following both open and percutaneous muscle biopsies is generally straightforward but requires attention to prevent hematoma formation, which is the most common complication.
- Wound Care: Apply sterile dressing sponges over the incision, held firmly in place with foam tape to create a compressive—but strictly non-constricting—bandage. This compression should be maintained for 48 hours to mitigate intramuscular bleeding.
- Suture/Strip Management: Adhesive strips or subcuticular sutures are left in place for 10 to 14 days.
- Medications: Routine perioperative prophylactic antibiotics are not indicated unless the patient has specific comorbidities. Narcotic analgesics are rarely necessary; mild discomfort is typically well-managed with acetaminophen. NSAIDs should be used with caution in the immediate postoperative period to avoid exacerbating bleeding risks.
- Mobilization: Patients are encouraged to mobilize immediately as tolerated. Crutches are rarely required, though strenuous athletic activity should be avoided for 1 to 2 weeks to allow the fascial defect to heal and prevent muscle herniation.
