Pathophysiology and Clinical Presentation
Chronic osteomyelitis remains one of the most formidable challenges in orthopedic surgery, notoriously difficult to eradicate completely. While systemic symptoms of acute infection may subside, one or more foci within the osseous architecture often harbor purulent material, infected granulation tissue, or a necrotic sequestrum. Patients frequently experience intermittent acute exacerbations spanning years or even decades, which may temporarily respond to rest and suppressive antibiotic therapy.
The pathophysiological hallmark of chronic osteomyelitis is the presence of infected, dead bone (sequestrum) encased within a severely compromised soft-tissue envelope. These infected osseous foci are typically surrounded by an involucrum of sclerotic, relatively avascular bone, which is further enveloped by thickened periosteum, scarred muscle, and fibrotic subcutaneous tissue.
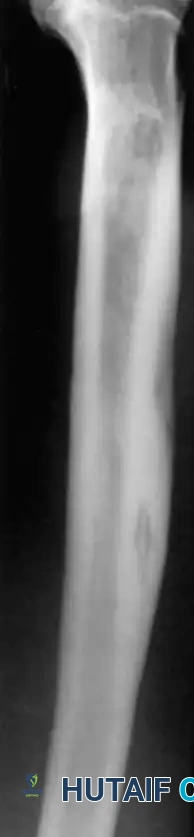
This avascular envelope of scar tissue creates a physiological barrier that renders systemic antibiotics essentially ineffective. Furthermore, bacteria in chronic osteomyelitis transition from a planktonic state to a sessile state, forming a complex glycocalyx biofilm. This slimy extracellular polymeric substance protects the bacteria from host phagocytic cells and prevents the penetration of antimicrobial agents. Secondary polymicrobial infections are common, and superficial sinus tract cultures notoriously fail to correlate with deep bone biopsy cultures. Eradication, therefore, mandates aggressive surgical excision combined with targeted, culture-specific antimicrobial therapy.
The Cierny-Mader Classification System
The management of chronic osteomyelitis requires a structured approach to determine whether treatment should be simple or complex, curative or palliative, and limb-sparing or ablative. The Cierny-Mader staging system remains the gold standard, combining anatomical extent with the physiological status of the host.
Anatomical Types
- Type I (Medullary): Endosteal disease, typically seen in hematogenous osteomyelitis or infected intramedullary nails.
- Type II (Superficial): Infection limited to the cortical surface, almost exclusively secondary to a soft-tissue coverage defect (e.g., exposed bone following trauma).
- Type III (Localized): A stable, well-demarcated lesion characterized by full-thickness cortical sequestration and cavitation. Complete debridement of this area does not compromise the mechanical stability of the bone.
- Type IV (Diffuse): A permeative osteomyelitic lesion that creates mechanical instability, either present at the time of diagnosis or occurring iatrogenically after appropriate radical debridement.
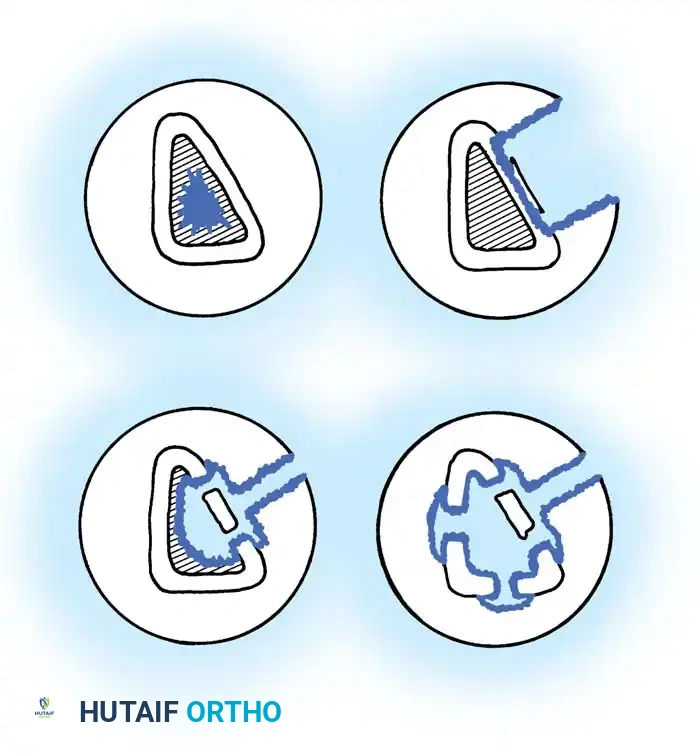
Physiological Host Classes
- Class A Host: Normal, immunocompetent patient with excellent local vascularity and physiological reserves.
- Class B Host: Compromised host. This is further subdivided into local compromise (B-L) such as venous stasis, arterial insufficiency, or extensive scarring, and systemic compromise (B-S) such as diabetes mellitus, malnutrition, immunosuppression, or tobacco abuse.
- Class C Host: Prohibitive host. The morbidity of the required surgical treatment outweighs the potential benefits, or the patient has a poor prognosis for cure. Palliative care or amputation is often indicated.
Clinical Pearl: A Type II lesion in a Class A host is designated as Stage IIA osteomyelitis. Always optimize Class B hosts (e.g., smoking cessation, glycemic control, nutritional supplementation) before undertaking complex reconstructive procedures.
Diagnostic Evaluation
The diagnosis of chronic osteomyelitis relies on a synthesis of clinical examination, laboratory parameters, and advanced imaging, culminating in the definitive histological and microbiological evaluation of infected bone.
Clinical and Laboratory Assessment
Physical examination must meticulously assess the integrity of the skin and soft-tissue envelope, map areas of tenderness, evaluate gross bone stability, and document the neurovascular status of the extremity. Laboratory studies are generally non-specific. While the Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) are elevated in the majority of patients, the white blood cell (WBC) count is elevated in only about 35% of cases.
Imaging Modalities
No single imaging technique can absolutely confirm or exclude osteomyelitis; however, a multimodal approach is essential for surgical planning.
- Plain Radiographs: The mandatory initial study. Signs of focal osteopenia, cortical destruction, periosteal reaction, and the presence of a sequestrum strongly suggest the diagnosis.
- Sinography: If an active sinus tract is present, the injection of a radiopaque contrast medium can delineate the tract's course and identify the deep focus of infection, serving as an invaluable adjunct for surgical planning.
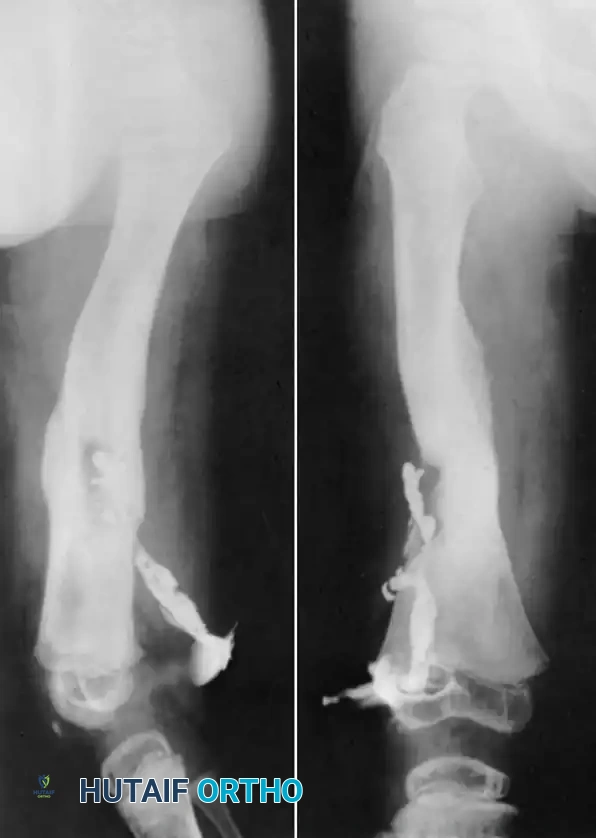
- Computed Tomography (CT): Provides unparalleled definition of cortical bone architecture. It is the superior modality for identifying small sequestra and planning the geometric extent of cortical resection.
- Magnetic Resonance Imaging (MRI): The modality of choice for evaluating the extent of marrow involvement and soft-tissue edema. In chronic osteomyelitis, MRI frequently reveals a well-defined rim of high signal intensity surrounding the active disease focus on T2-weighted images (the "rim sign"). However, MRI struggles to differentiate between active infection and fibrovascular scar tissue during the first postoperative year.
- Nuclear Medicine: Technetium-99m bone scans show increased osteoblastic activity but lack specificity. Gallium scans highlight leukocyte accumulation; a normal Gallium scan virtually excludes osteomyelitis. Indium-111–labeled leukocyte scans offer higher sensitivity and are particularly useful in differentiating chronic osteomyelitis from neuropathic (Charcot) arthropathy in the diabetic foot.
The Gold Standard: Deep Bone Biopsy
The absolute "gold standard" for diagnosis is an open bone biopsy sent for both histological analysis and aerobic/anaerobic/fungal/mycobacterial cultures. Swabs of superficial sinus tracts are highly misleading and should be avoided. Typically, Staphylococcus aureus is the predominant pathogen, though anaerobes, gram-negative bacilli, and increasingly, vancomycin-resistant enterococci (VRE) and methicillin-resistant S. aureus (MRSA) are isolated.
Principles of Surgical Treatment
Antibiotics alone cannot eradicate chronic osteomyelitis due to the avascular nature of the sequestrum and the protective bacterial biofilm. The cornerstone of treatment is radical surgical debridement, transforming a necrotic, infected environment into a viable, highly vascularized bed.
Surgical Warning: Inadequate debridement is the primary cause of recurrence. Simpson et al. demonstrated that wide resection margins (>5 mm of healthy bone) yielded a 0% recurrence rate, whereas marginal resections (<5 mm) resulted in a 28% recurrence rate, particularly in Class B hosts.
The surgical strategy must be multidisciplinary, involving an orthopedic surgeon, an infectious disease specialist, and often a plastic surgeon skilled in microvascular tissue transfer.
Surgical Technique: Sequestrectomy and Curettage
Sequestrectomy and radical curettage are technically demanding, time-consuming, and can result in significant blood loss. Meticulous preoperative planning is mandatory.
Preoperative Preparation and Positioning
- Sinus Tract Mapping: Inject the sinus tracts with methylene blue 24 hours prior to surgery. This stains the fibrotic, infected tract, making it easily identifiable for complete en bloc excision.
- Positioning: Position the patient on a radiolucent table to allow for intraoperative fluoroscopy.
- Tourniquet Use: A pneumatic tourniquet may be used for the initial exposure; however, it must be deflated prior to the final debridement to accurately assess bone viability (the "Paprika sign" – punctate bleeding from healthy Haversian canals).
Step-by-Step Surgical Approach
- Exposure and Tract Excision: Utilize an extensile approach. Excise all methylene blue-stained sinus tracts completely, down to the bone.
- Periosteal Elevation: Incise the indurated, thickened periosteum and elevate it 1.3 to 2.5 cm on each side of the lesion to expose the underlying involucrum.
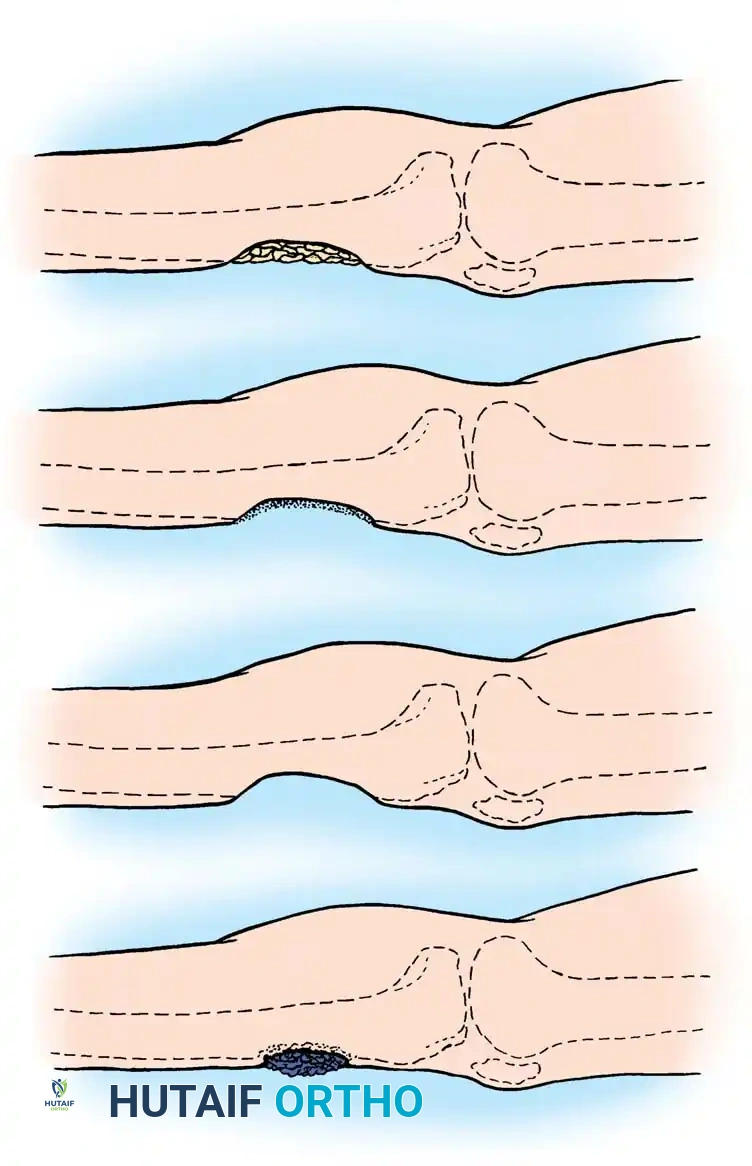
- Cortical Windowing: Use a high-speed burr or drill to outline a cortical window over the suspected abscess or sequestrum. Remove the cortical window with a sharp osteotome.
- Radical Debridement: Remove all sequestra, purulent exudate, and scarred, necrotic intramedullary tissue. If sclerotic bone seals off a medullary cavity, open the canal proximally and distally until healthy, bleeding marrow is encountered.
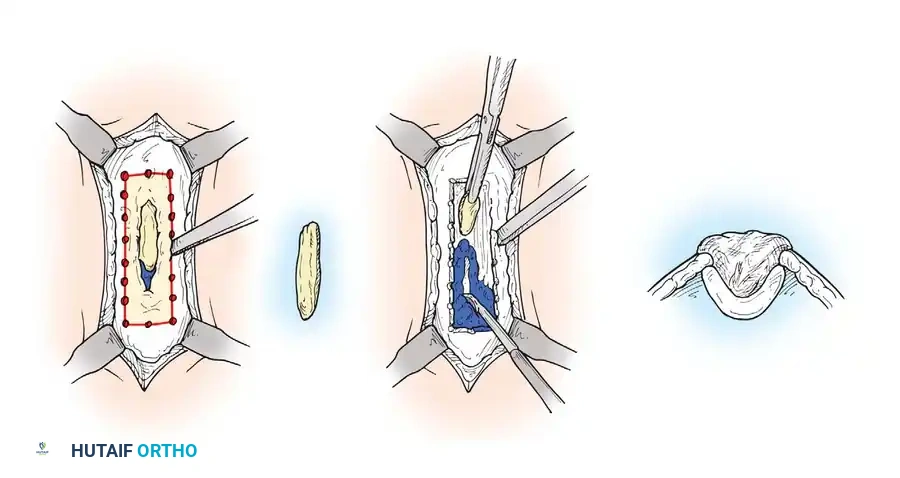
- Contouring the Defect: After removing all macroscopic infection, use a high-speed burr to saucerize the bone. Excise overhanging cortical edges to prevent the formation of a dead space where hematoma and subsequent infection can accumulate.
- Stabilization: If the debridement results in mechanical instability (converting to a Type IV lesion), the bone must be stabilized. An Ilizarov-type circular external fixator is preferred, as it avoids placing foreign hardware directly into the infected bed while providing rigid biomechanical stability.
- Wound Management: If the remaining soft-tissue envelope is healthy and tension-free, primary closure over suction drains may be attempted. If closure is impossible or the bed remains questionable, the wound must be packed open or managed with an antibiotic bead pouch for delayed closure.
Dead Space Management and Reconstruction
Adequate radical debridement inevitably leaves a large osseous dead space. If left unmanaged, this space will fill with hematoma, providing an ideal medium for recurrent infection. Several advanced techniques are employed to obliterate this dead space and reconstruct the limb.
1. Antibiotic Polymethyl Methacrylate (PMMA) Beads
PMMA beads impregnated with heat-stable antibiotics (e.g., Vancomycin, Tobramycin) serve a dual purpose: they physically obliterate the dead space and provide massive local concentrations of antibiotics (often 100 times the minimum inhibitory concentration) without systemic toxicity.
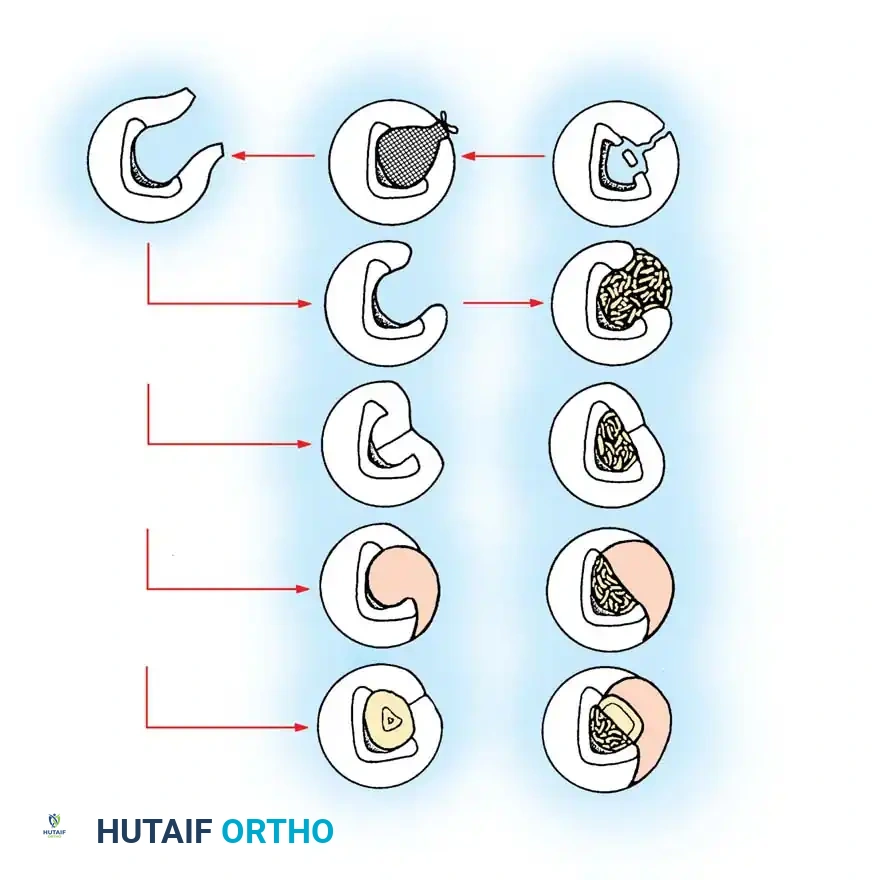
The beads are typically strung on a non-absorbable suture or surgical wire to facilitate easy removal. The wound is covered with a semi-permeable membrane (the "bead pouch" technique) to create a closed, high-concentration local environment. Beads are generally removed or exchanged at 3 to 6 weeks, followed by definitive bone grafting.
2. The Papineau Technique (Open Cancellous Grafting)
Described by Papineau, this technique relies on the principle that highly vascularized granulation tissue and autogenous cancellous bone are inherently resistant to infection.
* Phase 1: Radical excision and open wound management until a healthy bed of granulation tissue forms.
* Phase 2: The defect is densely packed with autogenous cancellous bone graft (typically harvested from the iliac crest).
* Phase 3: The graft is left open to the air, kept moist, and allowed to epithelialize secondarily, or it is covered with a split-thickness skin graft once the bone graft is fully covered by granulation tissue.
3. Local and Free Tissue Transfer
When the soft-tissue envelope is severely compromised, vascularized tissue must be imported to obliterate the dead space and improve local hemodynamics.
* Local Muscle Flaps: For proximal third tibial defects, a medial gastrocnemius rotational flap is ideal. For middle third defects, a soleus flap is utilized.
* Free Tissue Transfer: For distal third tibial defects, or massive defects elsewhere, microvascular free flaps (e.g., latissimus dorsi, rectus abdominis, or free fibula osteocutaneous flaps) are required. The imported muscle brings a robust blood supply that delivers systemic antibiotics, oxygen, and immune cells directly to the previously ischemic bed.
4. Distraction Osteogenesis (Ilizarov Technique)
For massive segmental bone defects (Type IV lesions), acute shortening followed by distraction osteogenesis, or bone transport using a circular external fixator, is the treatment of choice. This technique allows for the complete resection of all infected bone while simultaneously regenerating pristine, healthy bone through mechanical distraction, effectively addressing both the infection and the structural deficit.
Postoperative Protocol and Antimicrobial Therapy
The postoperative management of chronic osteomyelitis is as critical as the surgical intervention.
Antimicrobial Regimen
The duration and route of postoperative antibiotics remain a topic of academic debate. Traditionally, a 6-week course of culture-directed intravenous antibiotics is prescribed, managed by an infectious disease specialist. However, recent literature (e.g., Swiontkowski et al.) supports the use of highly bioavailable oral antibiotics (such as fluoroquinolones) following an initial short course of IV therapy, provided the organism is susceptible and the patient is compliant.
Rehabilitation and Monitoring
- Immobilization: The limb must be adequately splinted or protected in an external fixator to prevent pathological fractures through the stress risers created by cortical windowing and saucerization.
- Weight-Bearing: Strictly protected weight-bearing is enforced until radiographic evidence of bone graft incorporation or consolidation of the regenerate bone (in transport cases) is confirmed.
- Surveillance: Patients require long-term follow-up with serial inflammatory markers (ESR, CRP) and radiographs. Recurrence can occur years after apparent eradication, making lifelong vigilance a necessity for the orthopedic surgeon.
