Introduction to Benign Bone Tumors
Benign bone tumors and nonneoplastic conditions simulating bone tumors represent a diverse spectrum of osseous lesions frequently encountered in orthopedic oncology and general practice. By definition, these bony lesions do not behave aggressively on a local level and lack the biological capacity to metastasize. The majority of these lesions remain asymptomatic or minimally symptomatic, often discovered incidentally during radiographic evaluation for unrelated trauma or joint pain.
However, clinical intervention becomes paramount when these lesions precipitate complications, most notably pathological fractures, severe pain, or mechanical joint block. The diagnostic algorithm relies heavily on a synthesis of patient demographics, precise anatomical location, radiographic morphology, and, when necessary, histopathological confirmation. This masterclass focuses on the two most prominent benign bone-forming tumors: the highly symptomatic Osteoid Osteoma and the typically quiescent Bone Island (Enostosis).
Bone-Forming Tumors: Osteoid Osteoma
Osteoid osteoma is a benign, bone-forming neoplasm characterized by a small, highly vascularized radiolucent nidus surrounded by a dense zone of reactive cortical sclerosis. It is a hallmark lesion in orthopedic oncology, demanding a nuanced understanding of its biochemical behavior and surgical management.
Demographics and Clinical Presentation
Osteoid osteomas exhibit a strong predilection for young men, with the vast majority of cases diagnosed in the second or third decades of life. While an occasional lesion is reported in older demographics, the classic patient is a young adult male presenting with a highly specific pain profile.
Almost any bone in the axial or appendicular skeleton can be involved. However, there is a distinct predilection for the lower extremities, with approximately 50% of all cases localized to the femur or tibia. The tumor may arise within cortical bone (most common), cancellous bone, or subperiosteally. Multicentric lesions have been reported but remain exceedingly rare. Crucially, no malignant transformation of an osteoid osteoma has ever been documented in the literature.
The clinical hallmark of an osteoid osteoma is intense, unremitting pain that is characteristically worse at night. This pain is biochemically mediated by high levels of Prostaglandin E2 (PGE2) produced by the lesional nidus. Consequently, the pain is classically and dramatically relieved by aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) within 20 to 30 minutes of administration.
CLINICAL PEARL: The "Aspirin Test" is highly sensitive for osteoid osteoma. If a young patient presents with nocturnal bone pain that completely resolves with NSAIDs, osteoid osteoma must be the primary differential diagnosis until proven otherwise.
Secondary clinical signs depend heavily on the anatomical location of the lesion:
* Juxta-articular Lesions: When located near a joint (e.g., the femoral neck), the intense inflammatory response can cause localized swelling, joint stiffness, synovial hypertrophy, and eventual flexion contractures.
* Spinal Lesions: Osteoid osteomas of the spine typically involve the posterior elements (pedicle, lamina, pars interarticularis). The localized muscle spasm induced by the inflammatory nidus frequently results in a painful, rigid scoliosis. The lesion is almost universally located on the concave side of the scoliotic curve.
Diagnostic Imaging
Imaging studies are usually definitive, and a biopsy is rarely required to confirm the diagnosis prior to definitive treatment. The diagnostic criteria hinge on identifying the central nidus, which by definition is less than 1.5 cm in diameter. Lesions identical in histology but larger than 1.5 cm are classified as osteoblastomas.
Plain Radiographs:
Standard orthogonal radiographs are often sufficient to establish the diagnosis, particularly in diaphyseal cortical lesions. The classic appearance is a small, radiolucent central nidus surrounded by a massive, thick rim of reactive sclerotic bone.
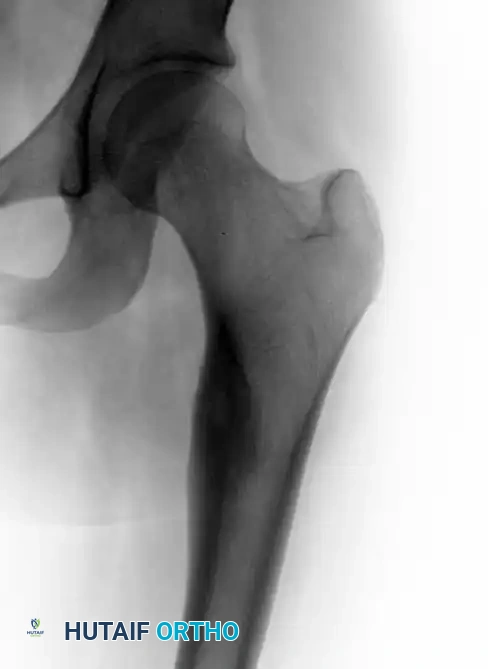
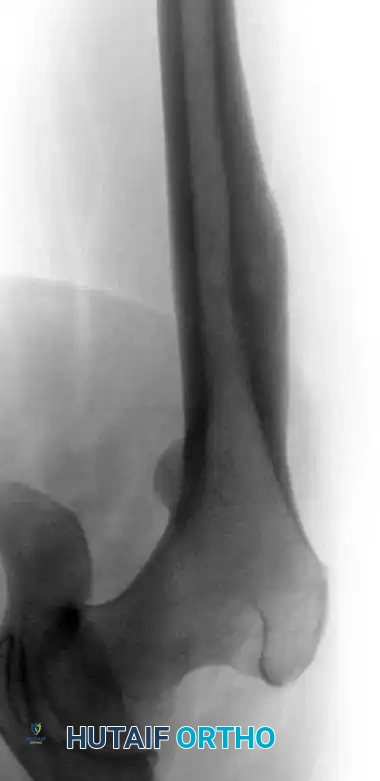
Anteroposterior view of the left hip in a 17-year-old girl complaining of left thigh pain, demonstrating a small radiolucent lesion with a thick sclerotic rim of reactive bone.
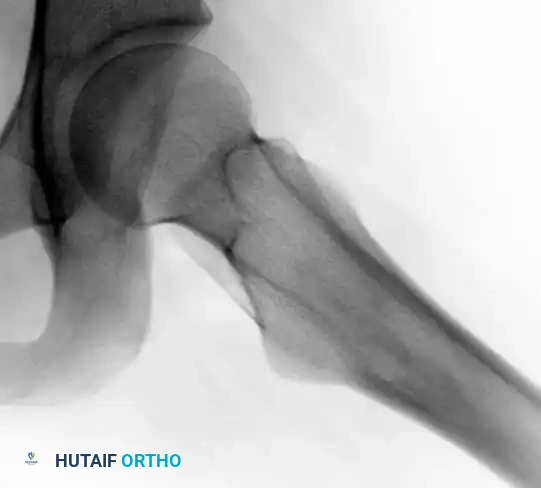
Lateral view of the same left hip, further delineating the reactive cortical thickening characteristic of an osteoid osteoma.
Lateral radiograph demonstrating an osteoid osteoma in the lateral cortex of the femoral diaphysis of a 16-year-old boy.
Computed Tomography (CT):
CT is the gold standard imaging modality for identifying the nidus and confirming the diagnosis. It is particularly invaluable for lesions in complex anatomy, such as the spine, pelvis, or femoral neck, where the nidus may be obscured by overlapping sclerotic bone on plain films.
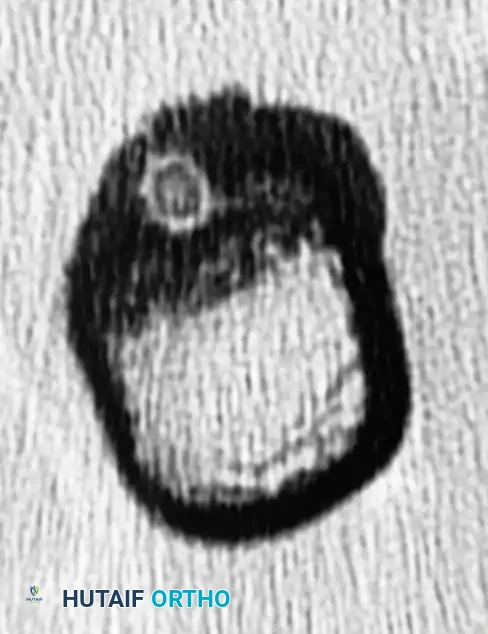
Axial CT scan clearly showing the radiolucent nidus with central calcification, confirming the diagnosis of osteoid osteoma.
Technetium-99m Bone Scintigraphy:
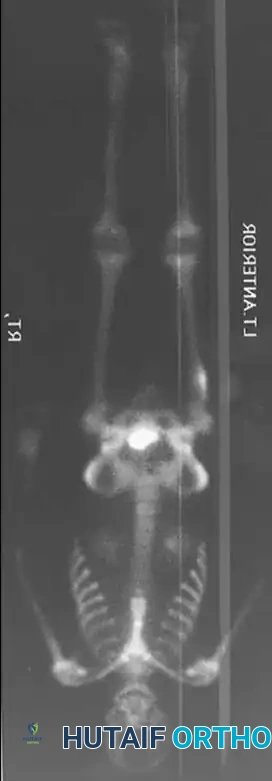
Bone scans are highly sensitive and demonstrate a classic "double-density" sign: a focal, intense area of increased uptake (the hyperemic nidus) superimposed on a broader area of moderately increased uptake (the reactive sclerotic bone).
Technetium bone scan showing intense, focal increased uptake in the area of the diaphyseal lesion.
Magnetic Resonance Imaging (MRI):
While MRI is frequently ordered for undifferentiated bone pain, it can be notoriously misleading in the context of osteoid osteoma. MRI typically shows extensive surrounding bone marrow and soft tissue edema.
SURGICAL WARNING: The massive edema seen on MRI can easily obscure the small nidus, leading the unwary clinician to misdiagnose the lesion as a highly aggressive malignancy (e.g., Ewing sarcoma) or acute osteomyelitis. Always correlate MRI findings with a high-resolution CT scan.
Histopathology
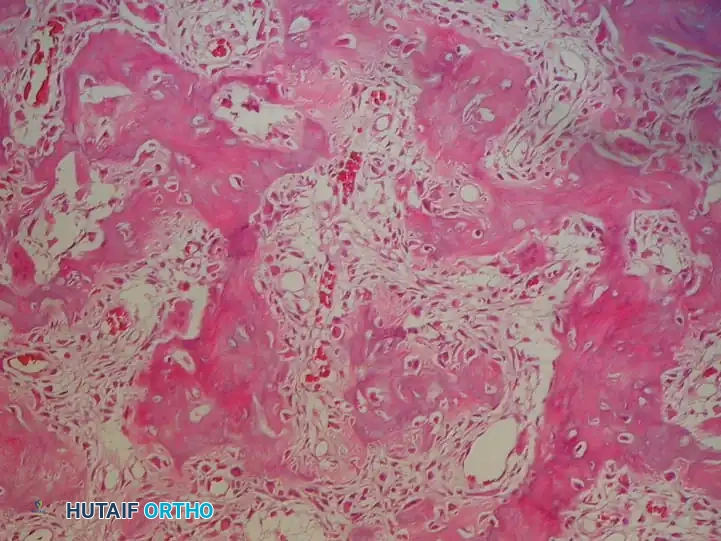
The microscopic appearance of an osteoid osteoma is highly characteristic and identical to that of an osteoblastoma, differentiated only by size. The lesion consists of a highly vascularized, fibrovascular stroma interspersed with interconnecting, immature bony trabeculae (woven bone). These trabeculae are prominently rimmed by active, plump osteoblasts.
Typical microscopic appearance of an osteoid osteoma, demonstrating fibrovascular tissue with immature bony trabeculae rimmed by prominent osteoblasts.
Crucially, there is absolutely no nuclear atypia, pleomorphism, or abnormal mitotic figures. Osteoclasts and occasional multinucleated giant cells can be observed within the stroma. The lesion is sharply demarcated from the surrounding reactive, mature sclerotic lamellar bone.
Management Strategies
Multiple treatment modalities are available, ranging from conservative medical management to minimally invasive ablation and open surgical resection. The choice of treatment depends on the lesion's location, the severity of symptoms, and patient preference.
Medical Management
If the patient's symptoms are adequately controlled with NSAIDs and they are amenable to long-term medical therapy, pharmacological management can be utilized as the definitive treatment. The natural history of osteoid osteoma is one of spontaneous "burnout" and healing, typically occurring within 3 to 4 years. However, the gastrointestinal and renal risks of chronic NSAID use must be carefully weighed against the benefits of avoiding intervention.
Percutaneous Radiofrequency Ablation (RFA)
For most patients with lesions of the pelvis or long bones of the extremities, percutaneous CT-guided radiofrequency ablation (RFA) has supplanted open surgery as the treatment of choice.
Technique:
1. Positioning and Anesthesia: The patient is positioned in the CT scanner based on the lesion's location. General anesthesia or deep sedation is required, as the heating process is intensely painful.
2. Localization: A high-resolution CT scan localizes the nidus.
3. Access: A bone biopsy needle (typically 11- or 14-gauge) is advanced through the cortex directly into the center of the nidus under CT guidance. A core biopsy is often sent for histopathological confirmation.
4. Ablation: A radiofrequency electrode is inserted through the cannula of the biopsy needle. The generator is activated, and the temperature at the uninsulated tip is increased to 90ºC for exactly 6 minutes. This induces coagulative necrosis of the entire nidus.
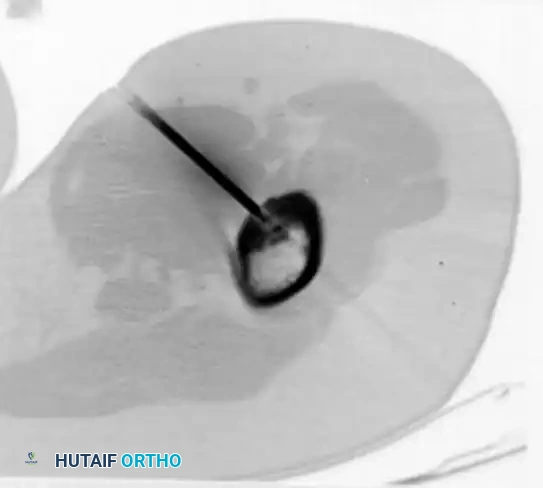
A radiofrequency ablation probe placed precisely into the nidus under CT guidance.
Outcomes: RFA is typically performed as an outpatient procedure. Patients experience immediate pain relief and can return to full weight-bearing and activity almost immediately. The recurrence rate is less than 10%.
PITFALL: RFA is generally contraindicated for spinal lesions located within 1 cm of the spinal cord or exiting nerve roots due to the risk of thermal neural injury. It is also relatively contraindicated in the small bones of the hands and feet, where thermal necrosis of adjacent skin or cartilage may occur.
Open Surgical Management: The Burr-Down Technique
When RFA is contraindicated, unavailable, or has failed, open surgical management is required. The surgical objective is the complete removal of the entire nidus. Historically, en bloc resection was performed; while associated with a near-zero recurrence rate, it removes a massive amount of structural bone, creating a significant stress riser and a high risk of postoperative pathological fracture.
Today, the "burr-down" technique is the gold standard for open resection, minimizing the removal of healthy reactive bone while ensuring complete nidal eradication.
Step-by-Step Surgical Approach:
1. Preoperative Planning: Precise localization using preoperative CT is mandatory.
2. Positioning: The patient is positioned on a radiolucent table to allow for unhindered intraoperative fluoroscopy.
3. Surgical Approach: A direct approach to the affected bone is utilized. Muscle-splitting techniques are preferred over muscle detachment to accelerate rehabilitation.
4. Intraoperative Localization: The exact location of the nidus is identified using multi-planar fluoroscopy. In rare cases where the nidus is entirely intramedullary and invisible on fluoroscopy, intraoperative gamma probe localization (following preoperative injection of technetium-99m methylene diphosphonate) can be utilized.
5. Cortical Unroofing: A high-speed power burr is used to meticulously shave down the reactive sclerotic bone directly overlying the nidus. Continuous cold saline irrigation is essential to prevent thermal necrosis of the surrounding healthy bone.
6. Nidus Excision: As the burr reaches the nidus, it typically appears as a hyperemic, distinct "cherry-red" granular tissue bed contrasting sharply with the white sclerotic bone. The nidus is meticulously removed using small curets and sent for pathological examination.
7. Cavity Preparation: The resulting cavity is lightly treated again with the power burr to ensure that the entire nidus and its microscopic margins have been eradicated.
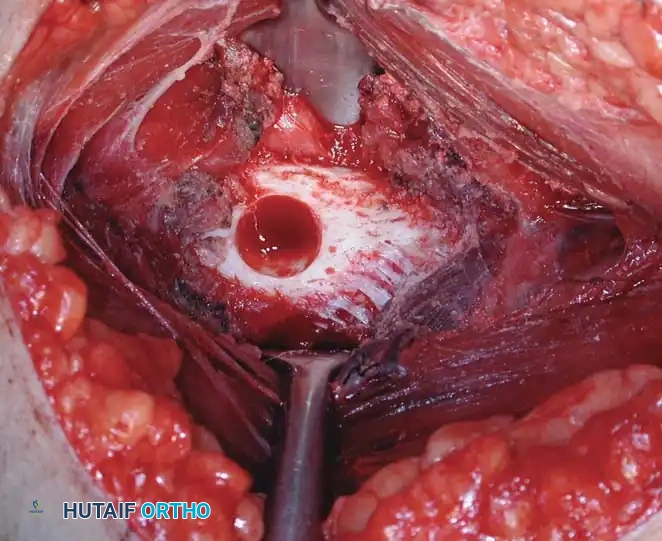
Intraoperative photograph demonstrating the cortical defect following the precise burr-down procedure and complete curettage of the hyperemic nidus.
Postoperative Protocol:
Because the burr-down technique preserves the structural integrity of the surrounding cortical bone, prophylactic internal fixation is rarely indicated. Patients are typically allowed weight-bearing as tolerated, with crutches for comfort. Recurrence rates with this meticulous technique remain less than 10%.
Bone-Forming Tumors: Bone Island (Enostosis)
Bone islands, clinically termed enostoses, are benign, hamartomatous lesions of cancellous bone. They represent a focus of mature, compact cortical bone located entirely within the medullary cavity.
Pathophysiology and Clinical Presentation
Bone islands are almost universally asymptomatic and are discovered incidentally on radiographs obtained for other reasons. They can involve any bone in the skeleton, though they are frequently seen in the pelvis, proximal femur, and ribs.
The exact etiology remains debated; it is unclear whether they represent a developmental abnormality (a failure of osteoclastic resorption during endochondral ossification) or a benign neoplastic process. Regardless of their origin, the vast majority remain completely quiescent throughout the patient's life.
Osteopoikilosis:
Osteopoikilosis ("spotted bone disease") is a rare, benign sclerosing bone dysplasia characterized by the presence of multiple, widespread bone islands throughout the skeleton. It is often inherited in an autosomal dominant pattern, frequently associated with mutations in the LEMD3 gene. When associated with connective tissue nevi (disseminated lenticular dermatofibrosis), it is termed Buschke-Ollendorff syndrome.
Diagnostic Imaging
The diagnosis of a bone island is almost exclusively radiological.
Plain Radiographs:
Bone islands present as small, round, or oval areas of homogeneously increased radiodensity (sclerosis) located within the cancellous bone. The pathognomonic feature is the presence of radiating bony spicules at the periphery of the lesion. These spicules merge seamlessly with the native trabeculae, creating a characteristic "brush-like" or "thorny" border. There is no associated bone destruction, radiolucent nidus, or periosteal reaction.
Advanced Imaging:
* Bone Scintigraphy: Bone islands typically show normal or only mildly increased uptake on technetium bone scans. A markedly "hot" scan should immediately raise clinical suspicion for a more aggressive osteoblastic lesion.
* Computed Tomography (CT): CT scans confirm the presence of thickened, dense trabeculae that merge imperceptibly with the surrounding normal cancellous bone.
* Magnetic Resonance Imaging (MRI): Because they are composed of dense cortical bone lacking marrow elements, bone islands are characteristically well-defined and uniformly dark (low signal intensity) on both T1- and T2-weighted images. Crucially, there is no surrounding bone marrow edema or soft tissue mass.
Histopathology
The microscopic appearance perfectly reflects the imaging characteristics. A bone island consists entirely of mature, lamellar bone with markedly thickened trabeculae. At the periphery, these lesional trabeculae merge directly with the normal host cancellous bone. There is no fibrovascular stroma, no sclerotic rim, and no cellular atypia. Occasionally, minor foci of woven bone may be present, but mature lamellar bone predominates.
Differential Diagnosis and Management
The primary clinical significance of a bone island lies in its differential diagnosis. While an enostosis is benign, it can radiographically mimic highly aggressive osteoblastic lesions. The differential diagnosis includes:
* Osteoblastic metastasis (particularly from prostate or breast carcinoma)
* Sclerosing osteosarcoma
* Sclerotic myeloma
* Osteoma
Management Protocol:
The overwhelming majority of patients with bone islands require only observation. As long as the lesion remains asymptomatic and demonstrates no interval growth on serial plain radiographs, no further intervention is indicated.
SURGICAL WARNING: Biopsy is strictly indicated if a presumed bone island becomes painful, demonstrates rapid enlargement on serial imaging, or exhibits aggressive features on MRI (such as surrounding edema or cortical breakthrough). In such cases, a core needle biopsy is mandatory to rule out blastic metastasis or a primary sclerosing bone sarcoma.
Conclusion
The management of benign bone-forming tumors requires a high degree of clinical acumen. Osteoid osteomas, while benign, cause profound morbidity through prostaglandin-mediated pain and require definitive intervention—most commonly via CT-guided radiofrequency ablation or meticulous open burr-down resection. Conversely, bone islands are benign incidentalomas that demand restraint; the orthopedic surgeon's primary role is to accurately differentiate them from malignant osteoblastic processes, thereby sparing the patient unnecessary surgical intervention. Mastery of the radiographic and clinical nuances of these lesions is essential for the practicing orthopedic surgeon.
📚 Medical References
- osteoid osteoma, Clin Orthop Relat Res 223:237, 1987.
- Perry CR, Evans LG, Fogarty J, et al: A new surgical approach to fractures of the lateral tibial plateau, J Bone Joint Surg 66A:1236, 1984.
- Pogrund H: A practical approach for lateral meniscectomy, J Trauma 16:365, 1976.
- Putti V: Arthroplasty of the knee joint, J Orthop Surg 2:530, 1920.
- Wilson PD: Posterior capsularplasty in certain flexion contractures of the knee, J Bone Joint Surg 11:40, 1929.
- Femur Bosworth DM: Posterior approach to the femur, J Bone Joint Surg 26:687, 1944.
- Checroun AJ, Mekhail AO, Ebraheim NA, et al: Extensile medial approach to the femur, J Orthop Trauma 10:481, 1996.
- Henry AK: Exposure of the humerus and femoral shaft, Br J Surg 12:84, 1924–1925.
- Thompson JE: Anatomical methods of approach in operations on the long bones of the extremities, Ann Surg 68:309, 1918.
- Hip Abbott LC: Surgical approaches to the joints. In Cole WH, ed: Operative technic in specialty surgery, New York, 1949, Appleton-Century-Crofts. Brackett E: A study of the different approaches to the hip joint, with special reference to the operations for curved trochanteric osteotomy and for arthrodesis, Boston Med Surg J 166:235, 1912.
- Ferguson AB Jr: Primary open reduction of congenital dislocation of the hip using a median adductor approach, J Bone Joint Surg 55A:671, 1973.
- Frndak PA, Mallory TH, Lombardi AV: Translateral surgical approach to the hip: the abductor muscle “split,” Clin Orthop Relat Res 295:135, 1993.
- Gibson A: Vitallium cup arthroplasty of the hip joint, J Bone Joint Surg 31A:861, 1949.
- Gibson A: Posterior exposure of the hip joint, J Bone Joint Surg 32B:183, 1950.
- Gibson A: The posterolateral approach to the hip joint, AAOS Instr Course Lect 10:175, 1953.
- Hardinge K: The direct lateral approach to the hip, J Bone Joint Surg 64B:17, 1982.
- Harris WH: A new lateral approach to the hip joint, J Bone Joint Surg 49A:891, 1967.
- Harris WH: Extensive exposure of the hip joint, Clin Orthop Relat Res 91:58, 1973.
- Henry AK: Exposures of long bones and other surgical methods, Bristol, England, 1927, John Wright & Sons. Hoppenfeld S, deBoer P : Surgical exposures in orthopaedics: the anatomic approach, Philadelphia, 2003, Lippincott Williams & Wilkins. Iyer KM: A new posterior approach to the hip joint, Injury 13:76, 1981.
- Kaplan EB: The blood vessels of the gluteal region, Bull Hosp Jt Dis 7:165, 1946.
- Kocher T: Textbook of operative surgery, 3rd ed, London, 1911, Adam & Charles Black (Translated by HJ Stiles, CB Paul). Langenbeck B von: Über die Schussverletzungen des Huftgelenks, Arch Klin Chir 16:263, 1874.
- Ludloff K: Zur blutigen Einrenkung der angeborenen Huftluxation, Z Orthop Chir 22:272, 1908.
- Mallon WJ, Fitch RD: The medial approach to the hip revisited, Orthopedics 16:39, 1993.
- Marcy GH, Fletcher RS: Modification of the posterolateral approach to the hip for insertion of femoral-head prosthesis, J Bone Joint Surg 36A:142, 1954.
- McFarland B, Osborne G: Approach to the hip: a suggested improvement on Kocher’s method, J Bone Joint Surg 36B:364, 1954.
- McLauchlan J: The Stracathro approach to the hip, J Bone Joint Surg 66B:30, 1984.
- Moore AT: The self-locking metal hip prosthesis, J Bone Joint Surg 39A:811, 1957.
- Moore AT: The Moore self-locking Vitallium prosthesis in fresh
