Comprehensive Introduction and Patho-Epidemiology
The elbow is a highly congruent, complex hinge joint that presents unique challenges to the orthopedic surgeon. Its inherent osseous stability, derived from the precise articulation of the ulnohumeral and radiohumeral joints, is augmented by a robust soft tissue envelope comprising strong collateral ligaments and dynamic muscular stabilizers. However, this dense anatomical configuration means that the key neurovascular structures traversing the upper extremity pass intimately close to the joint capsule, both anteriorly and posteriorly. Consequently, mastering surgical approaches to the elbow requires a profound understanding of three-dimensional regional anatomy, internervous planes, and the specific patho-epidemiology of the lesion being addressed. The surgeon must constantly balance the need for maximal articular exposure against the risk of iatrogenic neurovascular injury and postoperative soft tissue morbidity.
Epidemiologically, trauma to the elbow exhibits a bimodal distribution. High-energy mechanisms, such as motor vehicle collisions and falls from height, typically result in complex, intra-articular distal humerus fractures, terrible triad injuries, and severe fracture-dislocations in the younger, active demographic. Conversely, low-energy falls in the osteoporotic elderly population frequently yield comminuted articular fractures that challenge the limits of internal fixation and increasingly necessitate total elbow arthroplasty (TEA). In both cohorts, the choice of surgical approach dictates the quality of the reduction, the security of the fixation, and ultimately, the functional outcome.
Historically, the evolution of elbow approaches has been driven by the pursuit of extensile exposure without compromising the extensor mechanism. While medial and lateral approaches inherently avoid the most critical neurovascular bundles, their utility is largely restricted by the bony architecture of the epicondyles and columns, limiting access to the central articular surface. Anterior and posterior approaches yield vastly superior visualization of the joint surfaces but bring the surgeon into direct conflict with the median, radial, and ulnar nerves, as well as the brachial artery.
Of the myriad surgical corridors described in the literature, the posterior approach—whether utilizing an olecranon osteotomy or a triceps-sparing/reflecting technique—remains the absolute workhorse for complex elbow reconstruction. The posterior approach with an olecranon osteotomy provides unparalleled, 360-degree visualization of the distal humerus articular surface, making it the gold standard for AO Type C distal humerus fractures. However, it mandates the creation of an iatrogenic fracture that carries its own set of complications. Therefore, modern orthopedic practice demands that the surgeon possess a versatile armamentarium of approaches, tailoring the incision and deep dissection precisely to the fracture morphology, the patient's physiologic status, and the planned reconstructive implants.
Detailed Surgical Anatomy and Biomechanics
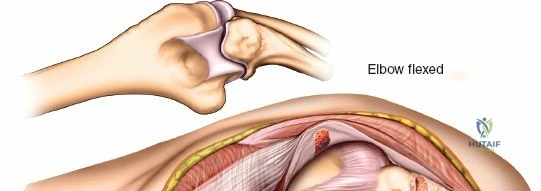
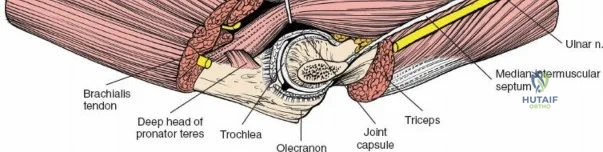
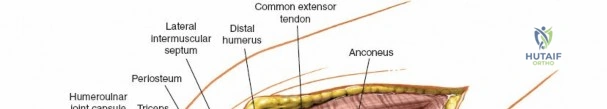
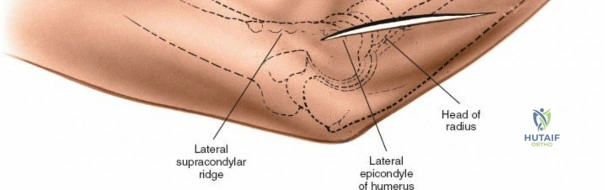
A rigorous command of elbow osteology and soft tissue anatomy is the foundation of safe surgical exposure. The distal humerus flares into the medial and lateral columns, which support the central articular segment composed of the trochlea and the capitellum. The trochlea, a spool-shaped structure, articulates with the greater sigmoid notch of the ulna, providing the primary flexion-extension hinge mechanism. The capitellum, a hemispherical structure located anteriorly and laterally, articulates with the radial head, facilitating forearm pronation and supination while acting as a secondary stabilizer against valgus stress. The structural integrity of this "triangle" (the two columns and the articular block) is paramount; failure to anatomically restore the columns leads to rapid post-traumatic arthrosis and mechanical failure.

The ligamentous anatomy dictates the limits of safe soft-tissue stripping during any approach. The medial collateral ligament (MCL) complex, specifically its anterior bundle, is the primary restraint to valgus instability. It originates from the anteroinferior aspect of the medial epicondyle and inserts onto the sublime tubercle of the proximal ulna. The lateral collateral ligament (LCL) complex, particularly the lateral ulnar collateral ligament (LUCL), is the primary restraint to posterolateral rotatory instability (PLRI). The LUCL originates from the lateral epicondyle and inserts onto the supinator crest of the ulna. Surgical approaches must meticulously preserve these origins and insertions; overzealous subperiosteal stripping, particularly during lateral or medial exposures, can result in catastrophic postoperative instability.
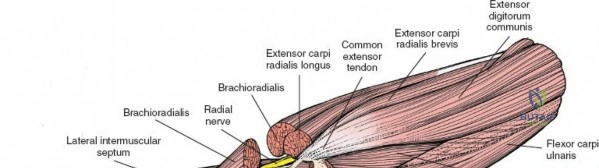
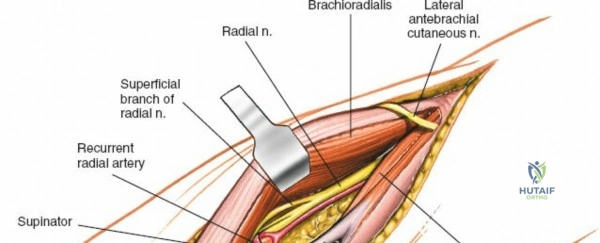
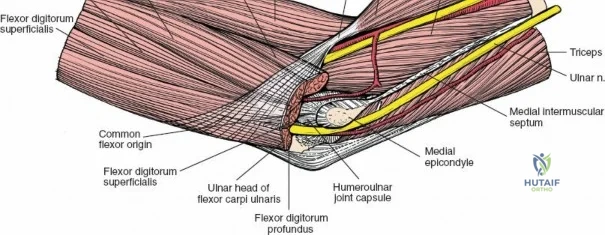
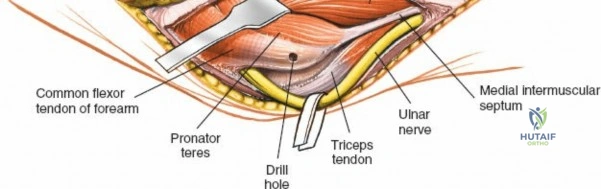
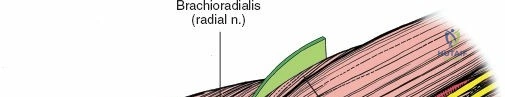
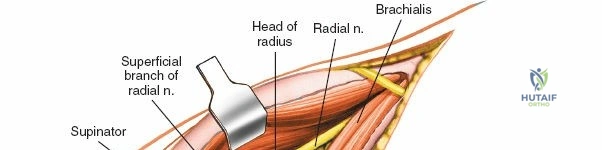
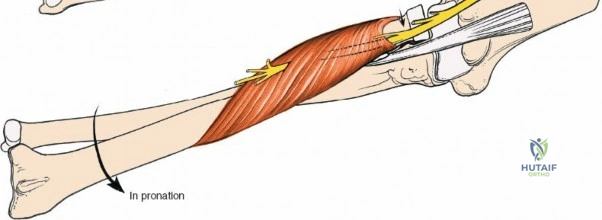
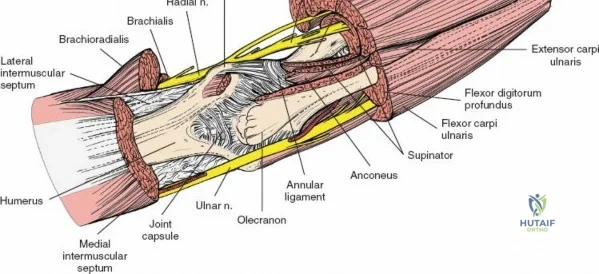
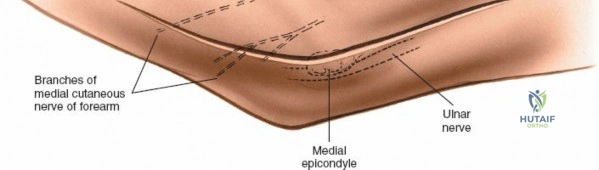
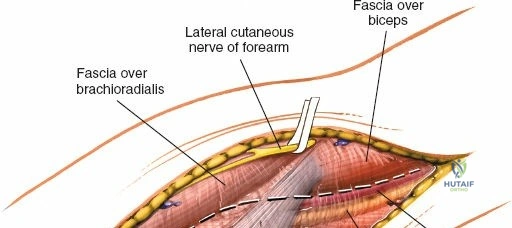
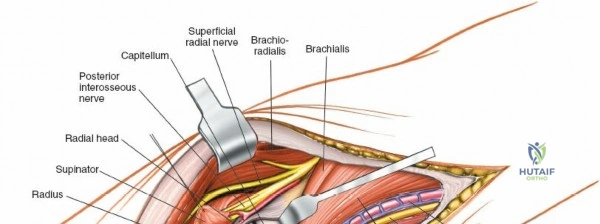
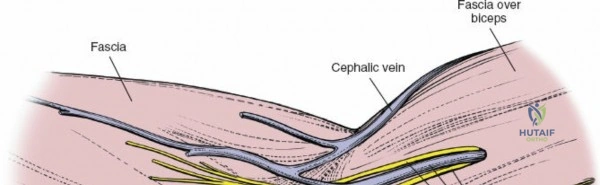
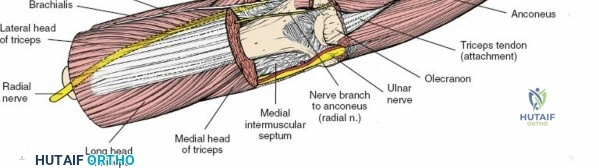
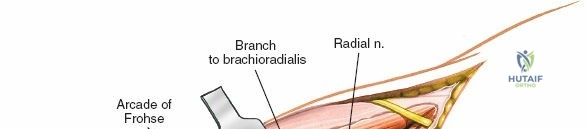
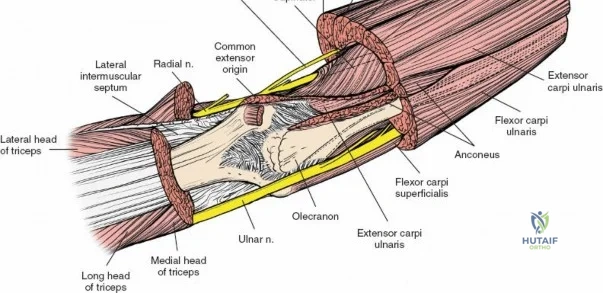
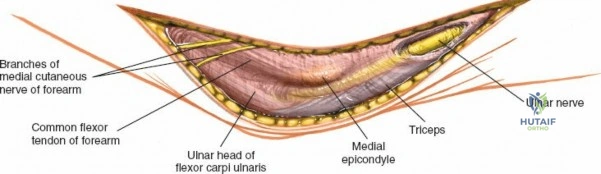
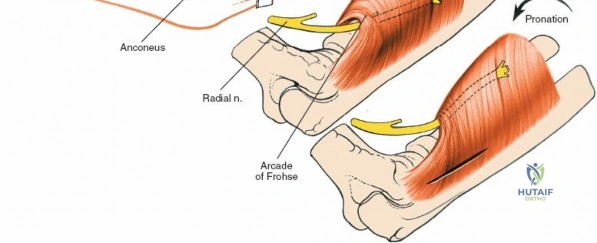
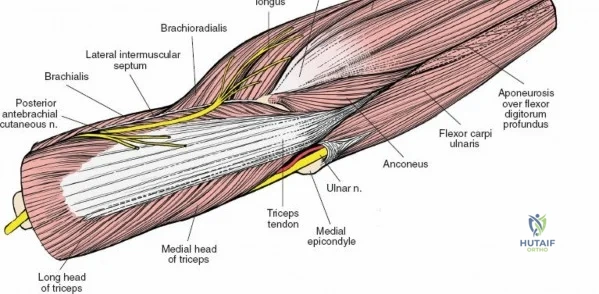
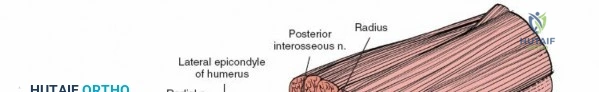
Neurovascular geography represents the greatest hazard during elbow surgery. The ulnar nerve is the most frequently encountered and manipulated structure. It courses through the posterior compartment of the arm, passes behind the medial intermuscular septum, and enters the cubital tunnel posterior to the medial epicondyle. Its intimate relationship with the posterior capsule makes it highly vulnerable during posterior and medial approaches. The radial nerve pierces the lateral intermuscular septum approximately 10 cm proximal to the lateral epicondyle, transitioning from the posterior to the anterior compartment. This anatomical landmark strictly limits the proximal extension of any posterior or lateral approach.
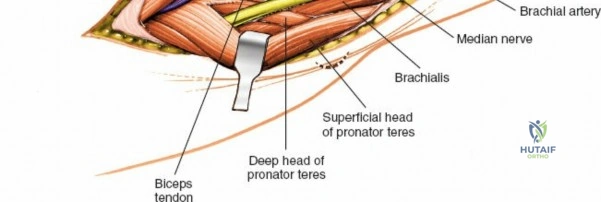
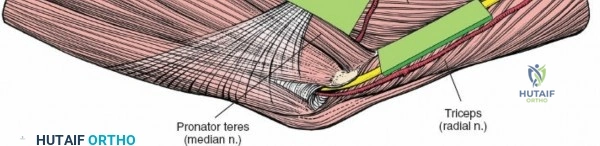
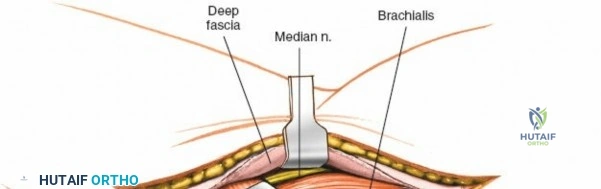
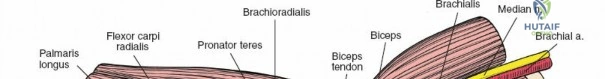
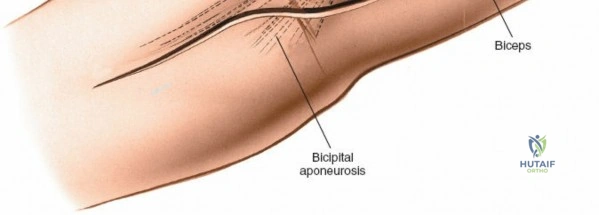
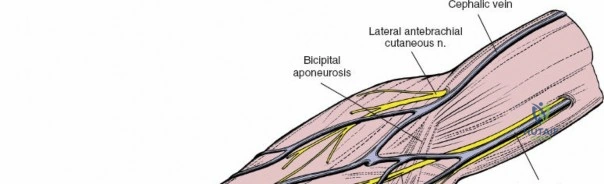
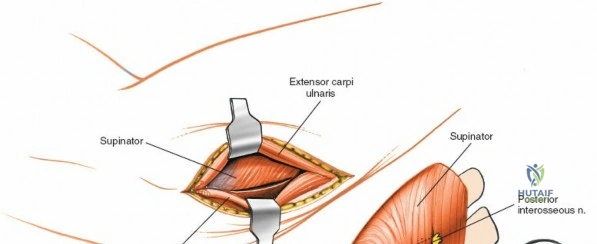
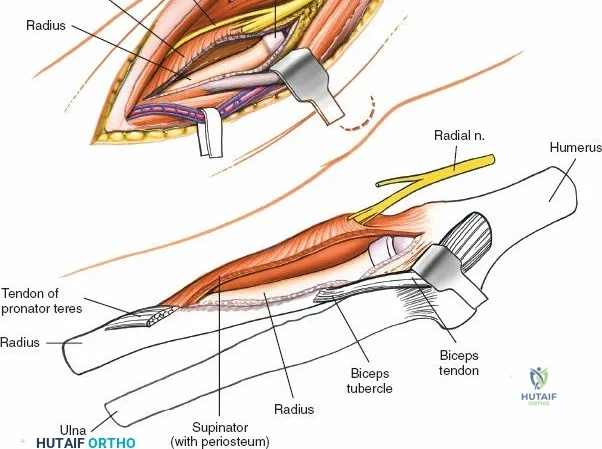
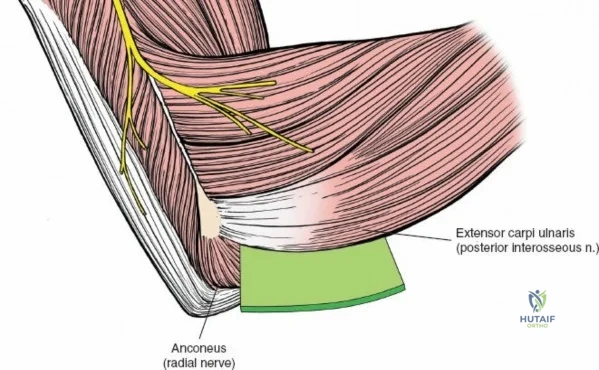
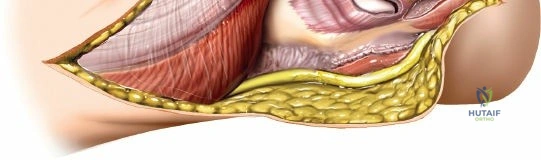
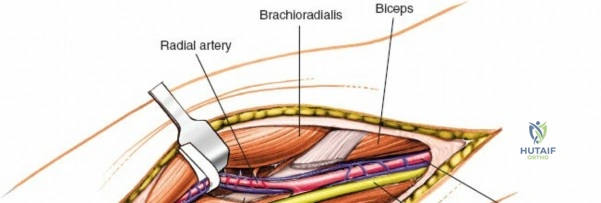
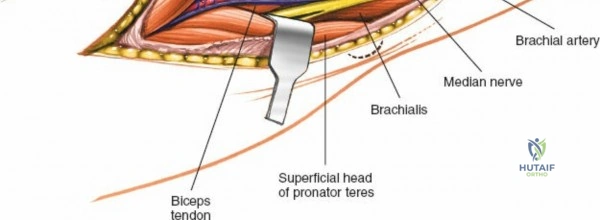
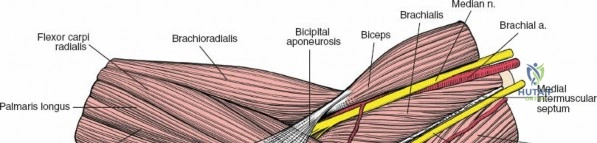
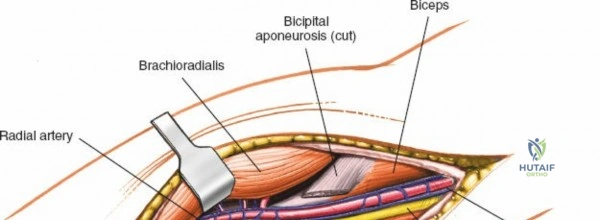
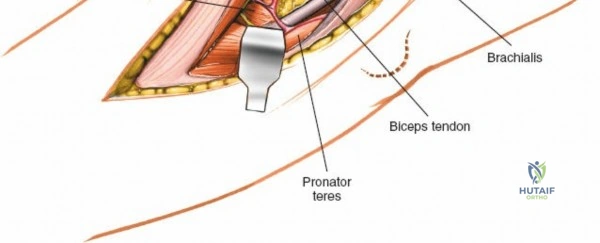
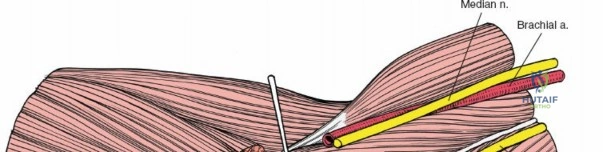
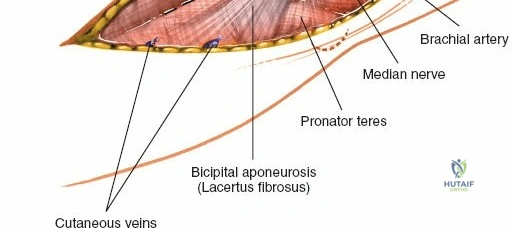
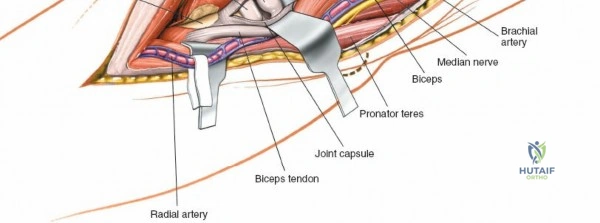
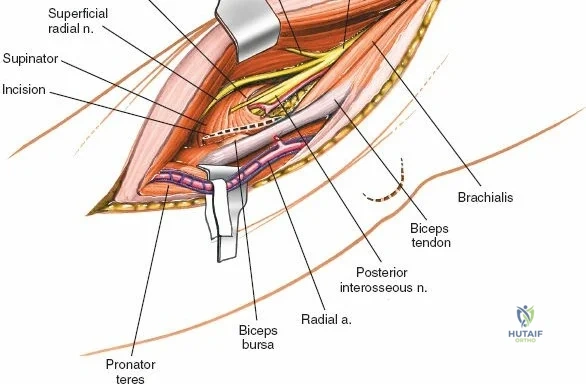
Anteriorly, the cubital fossa houses the median nerve and the brachial artery. These structures lie deep to the bicipital aponeurosis (lacertus fibrosus) and medial to the biceps tendon. The median nerve is at risk during anterior approaches, anterior capsular releases, or if instruments inadvertently plunge through the anterior capsule during a posterior approach. Understanding these spatial relationships allows the surgeon to exploit internervous planes—such as the interval between the anconeus (radial nerve) and the extensor carpi ulnaris (posterior interosseous nerve) in the Kocher approach—to safely navigate to the osseous pathology.
Exhaustive Indications and Contraindications
Selecting the optimal surgical approach is a nuanced decision predicated on fracture morphology, the necessity for articular visualization, and the planned intervention (e.g., internal fixation vs. arthroplasty). The surgeon must synthesize preoperative imaging with a deep understanding of the limitations of each anatomical corridor.
The posterior approach with an olecranon osteotomy remains the definitive choice for complex, intra-articular fractures of the distal humerus (AO/OTA 13-C2 and C3). It is indicated when absolute, anatomic visualization of the trochlear groove and capitellum is required to restore the articular puzzle. It is also utilized for the treatment of distal humerus nonunions and the extraction of elusive intra-articular loose bodies. However, it is relatively contraindicated in pediatric patients with open physes, in cases where a total elbow arthroplasty (TEA) is highly probable (as an intact extensor mechanism is crucial for TEA rehabilitation), and in patients with severe osteopenia where fixing the osteotomy may prove impossible.
The posterior approach without olecranon osteotomy (incorporating triceps-sparing, triceps-reflecting, or TRAP techniques) is the preferred approach for total elbow arthroplasty, extra-articular distal humerus fractures, and some simple intra-articular patterns. It preserves the structural integrity of the olecranon, eliminating the risk of osteotomy nonunion. The primary contraindication is a highly comminuted articular fracture that demands direct, orthogonal visualization of the joint surface, which a triceps-reflecting approach simply cannot provide without excessive and potentially damaging retraction.
| Surgical Approach | Primary Indications | Relative/Absolute Contraindications |
|---|---|---|
| Posterior (Olecranon Osteotomy) | Complex intra-articular distal humerus fractures (AO Type C), articular nonunions, massive loose bodies. | Planned Total Elbow Arthroplasty (TEA), severe osteopenia, open olecranon physis, active local infection. |
| Posterior (Triceps Sparing/Reflecting) | Total Elbow Arthroplasty, extra-articular distal humerus fractures, distal humerus osteotomies. | Highly comminuted intra-articular fractures requiring 360-degree visualization of the trochlea. |
| Anteromedial | Medial epicondyle fractures, coronoid fractures (via medial facet), MCL reconstruction, ulnar nerve transposition. | Pathology extending into the lateral column or capitellum; complex central trochlear fractures. |
| Posterolateral (Kocher) | Radial head/neck fractures, radial head arthroplasty, capitellum fractures, LCL reconstruction. | Pathology extending to the medial column; fractures requiring extensive proximal humeral exposure. |
| Anterior (Cubital Fossa) | Brachial artery repair, median nerve exploration, distal biceps tendon repair, anterior capsulectomy. | Posterior articular pathology; routine fracture fixation of the distal humerus. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the cornerstone of successful elbow surgery. Standard orthogonal radiographs (anteroposterior and lateral) provide a baseline, but fine-cut Computed Tomography (CT) scans with 3D reconstructions are now considered mandatory for all complex intra-articular fractures. CT imaging delineates the exact number of articular fragments, the presence of coronal shear fractures (e.g., capitellum or trochlea), and the degree of metaphyseal comminution. This allows the surgeon to mentally rehearse the reduction sequence and accurately template the required implants, such as anatomically pre-contoured parallel or orthogonal locking plates.
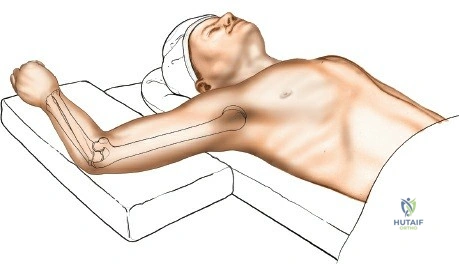
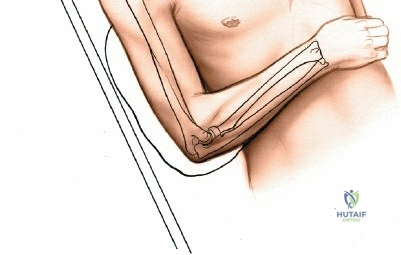
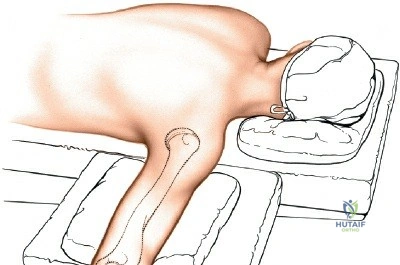
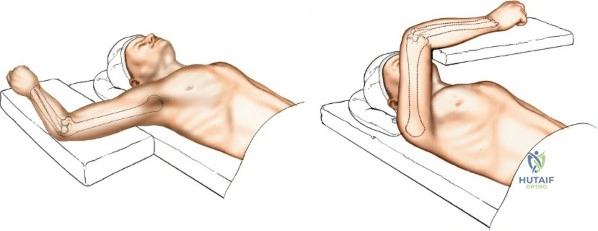
Patient positioning is a critical variable that dictates the ease of the procedure. For posterior approaches, the patient is almost universally placed in the prone position or the lateral decubitus position. In the prone position, the patient must be carefully intubated, and the chest and pelvis padded to allow uninhibited abdominal excursion for ventilation. The operative arm is abducted to 90 degrees, and the upper arm is supported by a padded post or sandbag, allowing the elbow to flex freely to 120 degrees over the side of the table. This position allows gravity to assist in reducing the distal humerus and provides the surgeon with an unobstructed, ergonomic view of the posterior articular surface.
Alternatively, the lateral decubitus position can be utilized, with the operative arm draped over a sterile bar or supported by an arm positioner. This position is highly advantageous for patients with severe polytrauma, cervical spine instability, or significant pulmonary compromise who cannot tolerate prone positioning. It also allows simultaneous access to the anterior and posterior aspects of the joint, which is beneficial for complex fracture-dislocations requiring both anterior and posterior stabilization.
A sterile tourniquet is routinely applied as proximally on the brachium as possible. The limb is exsanguinated by elevation for 3 to 5 minutes prior to tourniquet inflation. Careful draping must allow full mobility of the elbow from full extension to hyperflexion. The surgeon must ensure that intraoperative fluoroscopy (C-arm) can be easily maneuvered into the field to obtain true AP and lateral views without compromising the sterile field or requiring awkward manipulation of the unstable limb.
Step-by-Step Surgical Approach and Fixation Technique
Posterior Approach with Olecranon Osteotomy
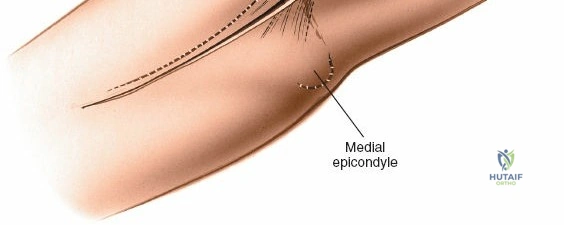
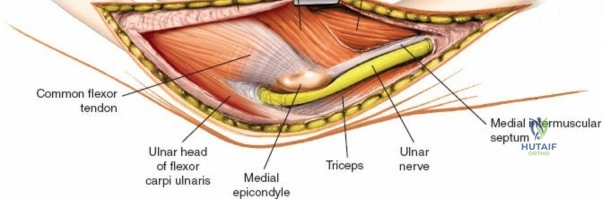
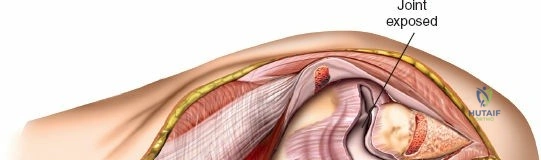
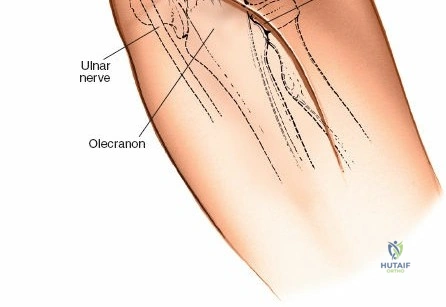
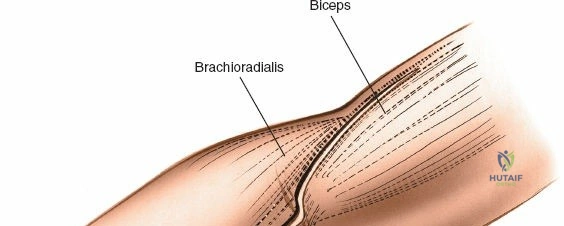
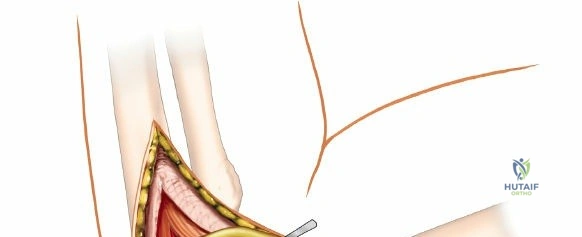
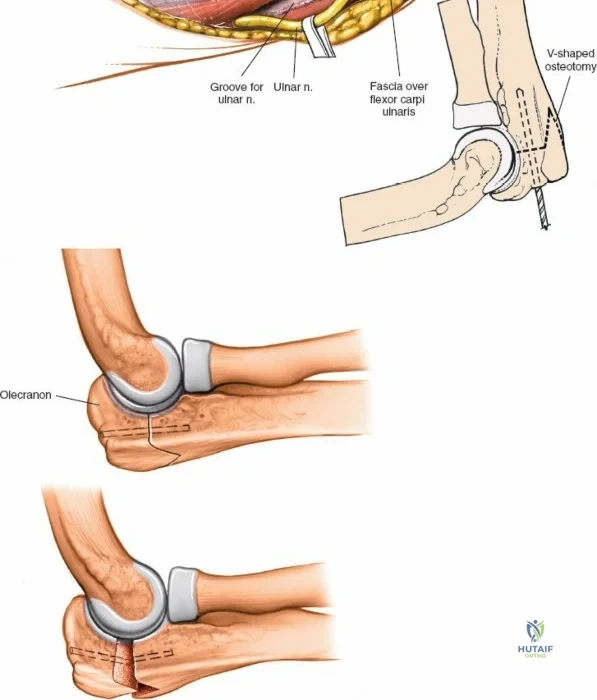
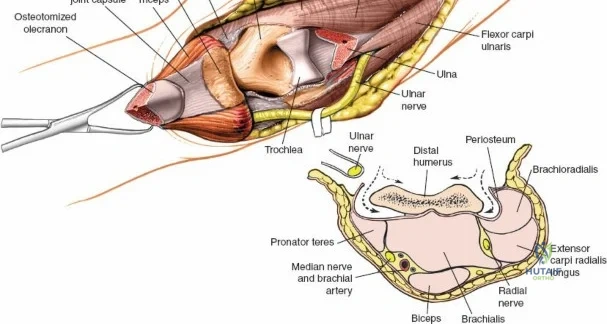
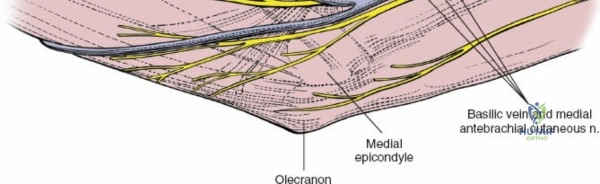
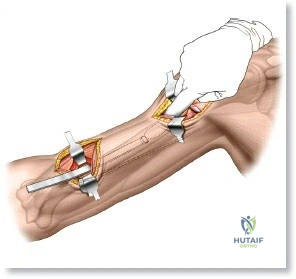
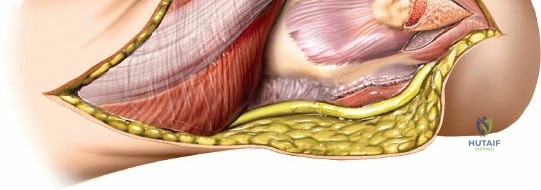
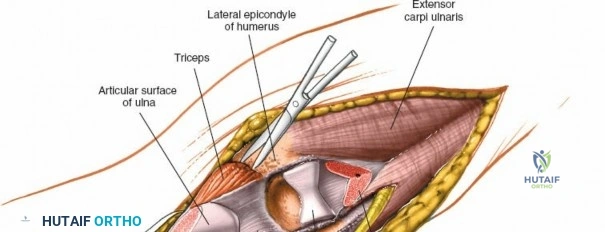
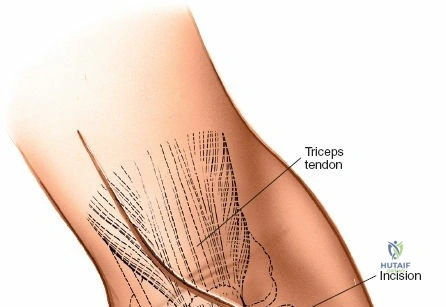
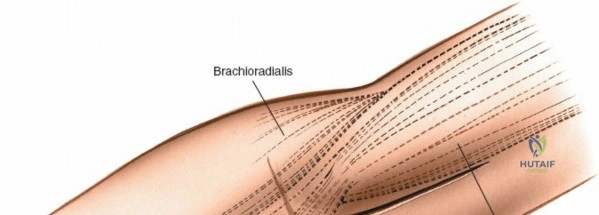
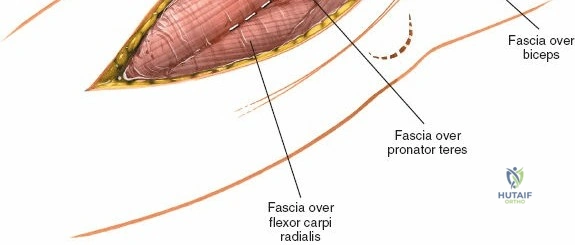
The posterior approach with an olecranon osteotomy provides the most extensile, unhindered view of the distal humerus. The landmarks are the medial and lateral epicondyles and the tip of the olecranon. A longitudinal incision is made beginning approximately 5 cm proximal to the olecranon in the posterior midline. As the incision approaches the olecranon tip, it must be curved laterally to bypass the bony prominence. This lateral curve is critical; it prevents the final surgical scar from resting directly over the weight-bearing tip of the elbow, which can lead to painful postoperative bursitis or wound breakdown. The incision is then curved back medially to run along the subcutaneous border of the ulnar shaft.
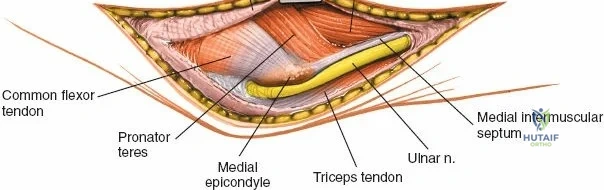
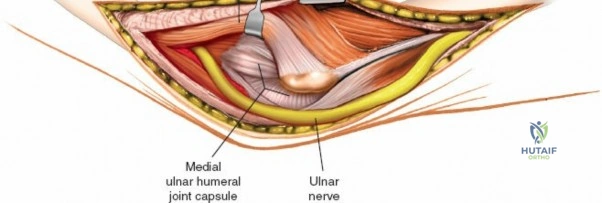
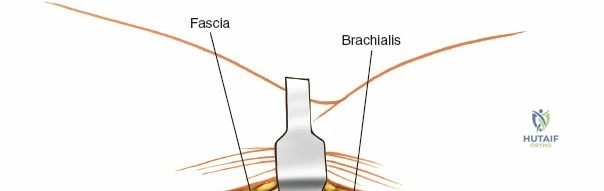
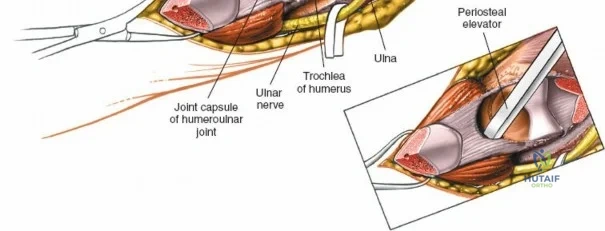
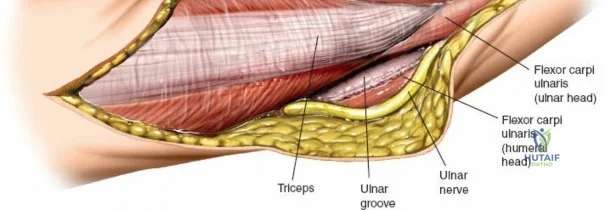
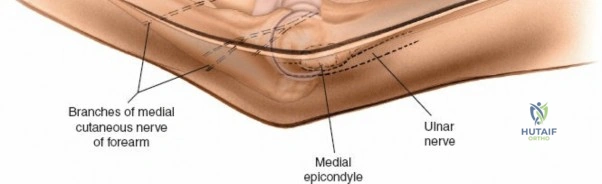
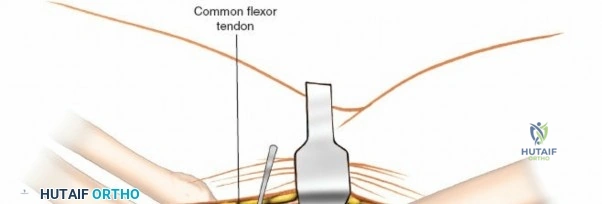
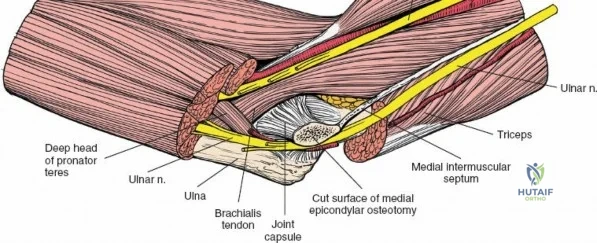
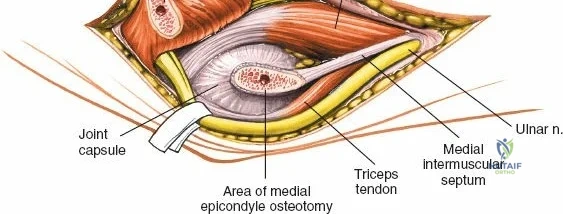
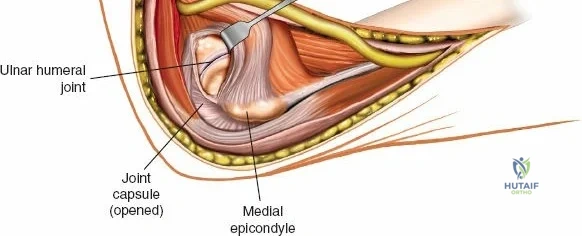
There is no true internervous plane for this approach; it relies on the mechanical detachment of the extensor mechanism. Deep dissection begins with the identification and protection of the ulnar nerve. The deep fascia over the medial epicondyle is incised, and the ulnar nerve is carefully unroofed from the cubital tunnel. The nerve must be mobilized circumferentially and protected with a vessel loop. Crucially, the vessel loop must never be used to apply continuous traction, as the ulnar nerve is exquisitely sensitive to stretch-induced ischemic neuropraxia. The nerve should simply be mobilized enough to allow it to be safely swept out of the operative field.
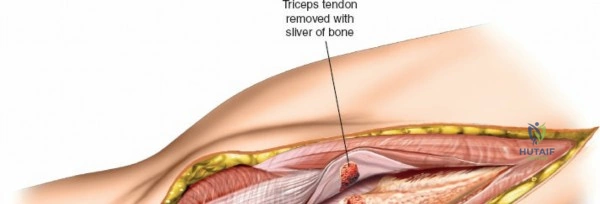
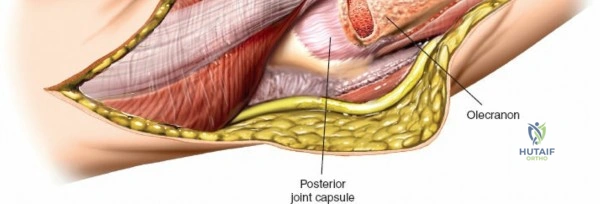
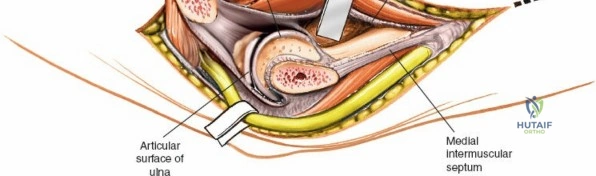
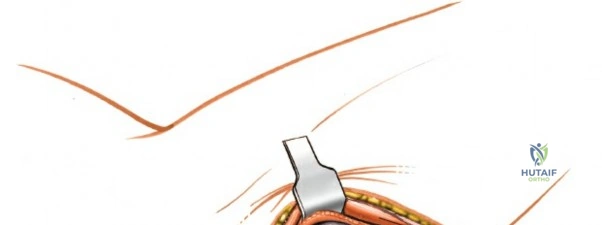
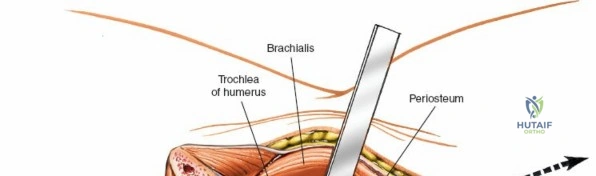
Prior to performing the osteotomy, the olecranon must be pre-drilled and tapped for the planned fixation (typically a 6.5mm or 7.3mm partially threaded cancellous screw, or a pre-contoured olecranon plate). Pre-drilling before the osteotomy ensures perfect anatomic alignment during closure. The articular surface of the olecranon is then exposed. A V-shaped (chevron) osteotomy is marked approximately 2 to 2.5 cm distal to the olecranon tip, with the apex pointing distally. The chevron shape provides inherent rotational stability when reduced. An oscillating saw is used to cut through the dorsal cortex and cancellous bone, stopping just short of the articular cartilage. An osteotome is then inserted into the kerf and twisted to crack the subchondral bone and articular cartilage. This technique creates a rough, interdigitating fracture line that facilitates exact anatomic reduction and enhances primary bone healing.
Once the osteotomy is complete, the proximal olecranon fragment, with the triceps tendon firmly attached, is reflected proximally. The soft tissues are elevated off the medial and lateral columns in a strictly subperiosteal plane to preserve the vascularity of the fracture fragments. The dissection can be extended distally by elevating the flexor carpi ulnaris (FCU) and extensor carpi ulnaris (ECU) off the proximal ulna. Proximal extension is strictly limited by the radial nerve, which crosses the posterior humerus and pierces the lateral intermuscular septum approximately one handbreadth (10 cm) proximal to the lateral epicondyle. Venturing proximal to this safe zone risks catastrophic radial nerve palsy.
Posterior Approach without Olecranon Osteotomy
For scenarios where an osteotomy is contraindicated (e.g., TEA, extra-articular fractures), triceps-sparing or triceps-reflecting techniques are employed. The skin incision and ulnar nerve management remain identical to the osteotomy approach.
In the Bryan-Morrey (triceps-reflecting) approach, the triceps is detached from the olecranon along with the periosteum of the proximal ulna and reflected laterally as a continuous sleeve. The anconeus is elevated from the ulna, and the entire extensor mechanism is peeled off the posterior humerus and olecranon. This provides excellent exposure for arthroplasty but requires meticulous, robust repair of the triceps mechanism through transosseous drill holes during closure to prevent postoperative extensor lag or rupture.
The Triceps-Reflecting Anconeus Pedicle (TRAP) approach involves elevating the anconeus muscle off the proximal ulna while preserving its vascular pedicle (from the recurrent interosseous artery). The triceps is then split in the midline or reflected, allowing access to the joint. This approach attempts to preserve the dynamic stability provided by the anconeus while avoiding an osteotomy.
Medial and Lateral Corridors
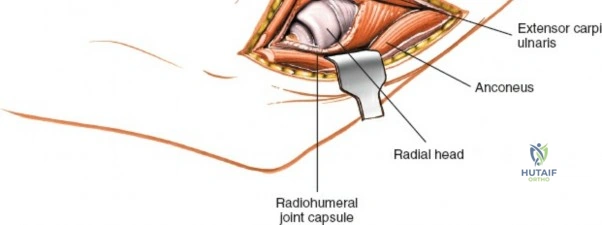
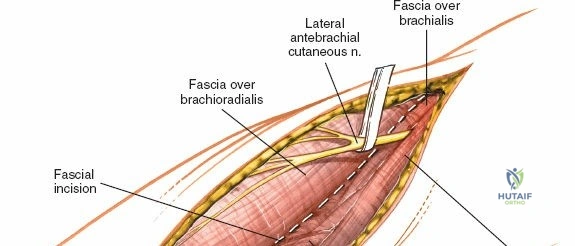
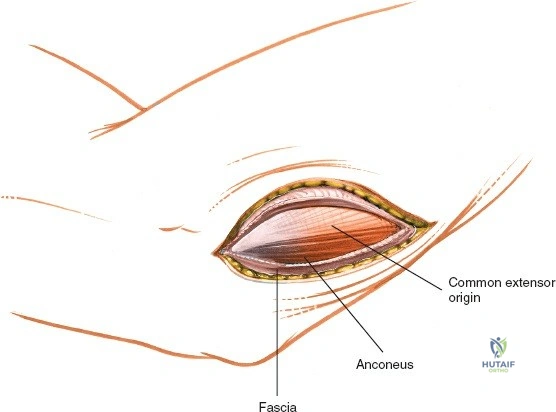
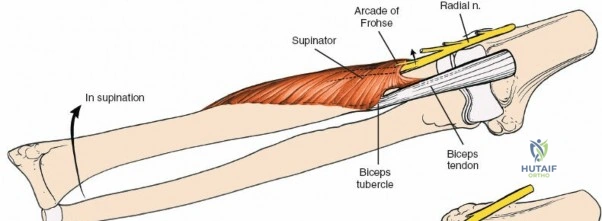
The Posterolateral (Kocher) approach is the gold standard for accessing the radial head and capitellum. The incision runs from the lateral epicondyle distally toward the ulnar styloid. The internervous plane lies between the anconeus (radial nerve) and the extensor carpi ulnaris (posterior interosseous nerve). The LCL complex must be fiercely protected; the arthrotomy is performed anterior to the lateral ulnar collateral ligament (LUCL) to prevent iatrogenic posterolateral rotatory instability. Distal extension is limited by the posterior interosseous nerve (PIN) as it wraps around the radial neck within the supinator muscle.
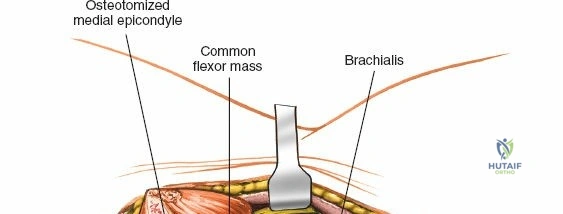
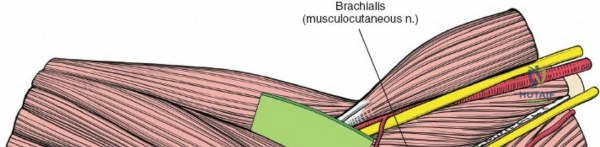
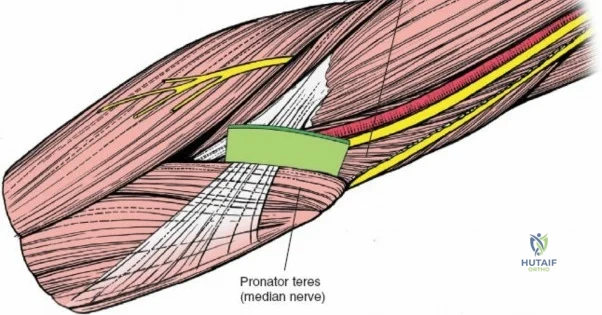
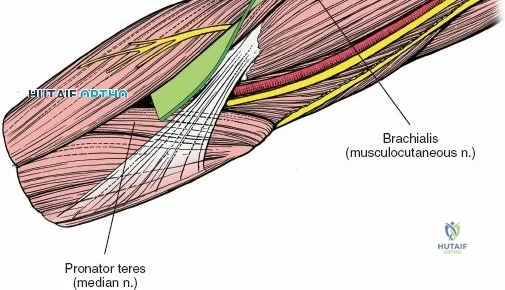
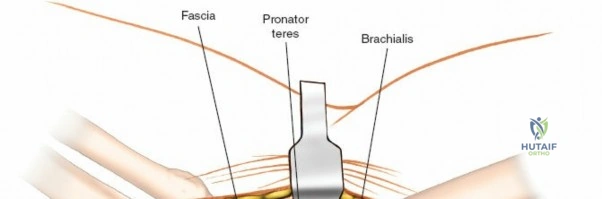
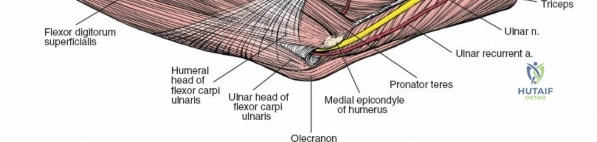
The Anteromedial approach is utilized for coronoid fractures and medial collateral ligament repairs. The incision is centered over the medial epicondyle. The internervous plane is between the pronator teres (median nerve) and the brachialis (musculocutaneous nerve). The ulnar nerve is identified and protected posteriorly. This approach allows direct visualization of the sublime tubercle and the anteromedial facet of the coronoid.
Complications, Incidence Rates, and Salvage Management
Surgical approaches to the elbow are fraught with potential complications, driven by the dense neurovascular anatomy and the joint's propensity for stiffness. The surgeon must be proactive in anticipating these issues and possess the skills for salvage management.
Ulnar neuropathy is the most frequent complication following posterior and medial approaches, with incidence rates reported between 5% and 15% in complex fracture fixations. This can result from direct intraoperative trauma, excessive traction from vessel loops, or postoperative compression from hematoma or heterotopic ossification. If the nerve subluxates over the medial epicondyle during flexion intraoperatively, or if prominent hardware is placed on the medial column, an anterior subcutaneous transposition should be performed. If postoperative neuropathy develops, initial management is conservative (gabapentinoids, bracing), but recalcitrant cases require surgical neurolysis and transposition.
Olecranon osteotomy nonunion or delayed union occurs in up to 5% of cases. Risk factors include poor fixation technique (e.g., failure to achieve compression), severe osteopenia, and early aggressive active extension. Tension band wiring, while classic, has a high rate of symptomatic hardware requiring removal (up to 40%). Modern techniques utilizing pre-contoured locking plates or heavy intramedullary screws have reduced nonunion rates. If a nonunion occurs, salvage requires revision open reduction, bone grafting (autograft or orthobiologics), and rigid plate fixation.
Heterotopic Ossification (HO) is a devastating complication that severely restricts range of motion. It is particularly common following high-energy trauma, delayed surgery, and concomitant traumatic brain injury. Prophylaxis is critical in high-risk patients. A single dose of localized radiation therapy (700 cGy) administered within 48 hours postoperatively, or a 3- to 6-week course of oral Indomethacin, significantly reduces the incidence of clinically relevant HO. If massive HO develops, surgical excision is indicated, but it must be delayed until the bone has matured (typically 6 to 12 months post-injury) and normal trabecular markings are seen on radiographs, to prevent immediate recurrence.
| Complication | Estimated Incidence | Prevention Strategies | Salvage / Management |
|---|---|---|---|
| Ulnar Neuropathy | 5% - 15% | Avoid traction on nerve; routine anterior transposition if hardware is prominent or nerve subluxates. | EMG/NCS evaluation; surgical neurolysis and anterior transposition. |
| Osteotomy Nonunion | 2% - 5% | Chevron-shaped cut; rigid fixation (plate or large screw); preserve soft tissue attachments. | Revision ORIF with autologous bone grafting and rigid locking plate fixation. |
| Heterotopic Ossification | 10% - 20% (High-energy) | Indomethacin (75mg SR daily) or single-dose radiation therapy (700 cGy) within 48 hours. | Delayed surgical excision (after 6-12 months) once HO is radiographically mature. |
| Symptomatic Hardware | 20% - 40% (Tension bands) | Countersink K-wires; use low-profile plates; bury knots deep to fascia. | Hardware removal after radiographic confirmation of solid union (minimum 6 months). |
| Iatrogenic Instability | 1% - 3% | Respect LUCL during lateral approaches; avoid over-resection of radial head. | Ligamentous reconstruction (e.g., palmaris longus autograft for LUCL). |

Phased Post-Operative Rehabilitation Protocols
The elbow is notoriously unforgiving of prolonged immobilization. The joint capsule rapidly thickens and contracts, leading to profound and often permanent stiffness. Therefore, the overarching goal of any surgical approach and subsequent fixation must be to achieve stability that allows for early, safe range of motion. Rehabilitation protocols must be meticulously communicated between the surgeon and the physical therapist, tailored to the specific approach and the security of the internal fixation.
Phase I: Immediate Post-Operative Phase (Weeks 0-2)
The primary goals in this phase are wound healing, edema control, and the prevention of capsular contracture. If rigid fixation of a fracture (or secure repair of an osteotomy/triceps reflection) was achieved, the elbow is typically immobilized in a well-padded posterior splint at 60 to 90 degrees of flexion for only 3 to 7 days to allow the soft tissues to rest. Active-assisted and passive range of motion (ROM) exercises for the shoulder, wrist, and digits are initiated immediately. By day 7, if the wound is stable, the splint is removed, and gravity-assisted active and active-assisted ROM of the elbow is initiated. If a triceps-reflecting approach was used, active extension against gravity is strictly prohibited to protect the repair.
Phase II: Intermediate Phase (Weeks 2-6)
During this phase, the focus shifts to maximizing arc of motion while protecting healing bone and soft tissue. Sutures or staples are removed at 14 days. Patients are transitioned to a hinged elbow brace, which can be locked at night to prevent flexion contractures (a common complication). Active ROM is encouraged, but forceful passive stretching is avoided, as it can induce microtrauma to the capsule, exacerbating inflammation and promoting heterotopic ossification. Isometrics for the biceps and triceps are introduced. For olecranon osteotomies, active triceps extension is permitted, but heavy resistive extension is avoided until early radiographic union is visible.
Phase III: Advanced Strengthening and Return to Function (Weeks 6-12+)
By week 6, clinical and early radiographic union of osteotomies or fractures should be evident. The hinged brace is discontinued. Progressive resistive exercises (PREs) are initiated to rebuild upper extremity strength. Dynamic splinting (e.g., turnbuckle splints) may be utilized if the patient has plateaued in their ROM and significant contractures remain. Full return to heavy manual labor or contact sports is generally delayed until 4 to 6 months postoperatively, contingent upon full osseous consolidation and the restoration of near-normal strength and motion.
Summary of Landmark Literature and Clinical Guidelines
The evolution of elbow approaches is heavily grounded in landmark biomechanical and clinical studies. A thorough understanding of this literature is essential for the academic orthopedic surgeon.
The concept of elbow stability and the critical role of the lateral ulnar collateral ligament (LUCL) was pioneered by O'Driscoll et al. Their work defined posterolateral rotatory instability (PLRI) and established the absolute necessity of preserving or reconstructing the LUCL during lateral approaches (such as the Kocher approach). This shifted the paradigm of lateral elbow surgery, mandating arthrotomies that remain anterior to the LUCL origin.
Regarding distal humerus fractures, the AO Foundation principles, championed by Jupiter and Mehne, established the standard of care for bicolumnar fixation. Their clinical series demonstrated that the posterior approach with an olecranon osteotomy provided the superior visualization required to achieve the rigid, orthogonal or parallel plate fixation necessary for early mobilization. This body of work solidified the olecranon osteotomy as the gold standard for AO Type C fractures.
More recently, the shift toward triceps-sparing approaches has been driven by the work of Bryan and Morrey, and later modifications by O'Driscoll (the TRAP approach). Morrey's extensive research on elbow biomechanics and total elbow arthroplasty highlighted the morbidity associated with extensor mechanism disruption. Clinical guidelines now strongly recommend triceps-sparing or reflecting approaches for TEA and extra-articular fractures to eliminate the risk of osteotomy nonunion and facilitate accelerated extensor rehabilitation.
Finally, the management of the ulnar nerve remains a topic of active debate. Meta-analyses comparing in situ decompression versus routine anterior transposition during distal humerus fixation have shown mixed results. However, current clinical consensus guidelines suggest that while routine transposition is not strictly mandatory, it should be aggressively employed if the nerve demonstrates intraoperative instability, if hardware is placed on the medial epicondyle, or if the patient exhibits preoperative ulnar neuritis.