Operative Repair of the Deep Branch of the Ulnar Nerve
Key Takeaway
The repair of the deep branch of the ulnar nerve demands meticulous microsurgical technique due to its complex motor innervation of the intrinsic hand muscles. Utilizing the modified Boyes technique, surgeons can achieve tension-free neurorrhaphy by rerouting the motor fascicles through the carpal tunnel. This guide details the precise anatomical dissection, fascicular mobilization, and postoperative protocols required to optimize functional recovery of the hand's intrinsic musculature.
INTRODUCTION AND BIOMECHANICAL SIGNIFICANCE
The ulnar nerve is the primary motor nerve of the hand, responsible for the intricate balance and fine motor control required for power grip and precision pinch. Injury to the deep branch of the ulnar nerve is particularly devastating, as it is a purely motor nerve (with the exception of minor articular branches to the carpal joints). Severance of this branch results in an "intrinsic-minus" hand, characterized by clawing of the ring and small fingers, profound weakness in key pinch (Froment’s sign), and loss of finger abduction and adduction (Wartenberg’s sign).
Because the deep branch of the ulnar nerve is deeply situated within the palm, injuries are often the result of penetrating trauma, crush injuries, or iatrogenic damage during deep palmar dissections. The primary goal of surgical intervention is the restoration of motor continuity. Whenever possible, primary end-to-end neurorrhaphy is preferred over nerve grafting, as motor recovery is significantly superior when a single neurorrhaphy site is utilized. To achieve a tension-free repair in the presence of a nerve gap, advanced mobilization techniques—such as the modified Boyes technique—are employed to reroute the nerve and gain critical length.
💡 Clinical Pearl
The deep branch of the ulnar nerve is highly fascicular and purely motor. Misalignment during repair will result in irreversible loss of intrinsic muscle function. Always utilize high-powered loupe magnification for the initial dissection and an operating microscope for the final neurorrhaphy to ensure precise fascicular matching.
SURGICAL ANATOMY
Understanding the complex three-dimensional anatomy of the ulnar nerve at the wrist and palm is paramount for successful repair.
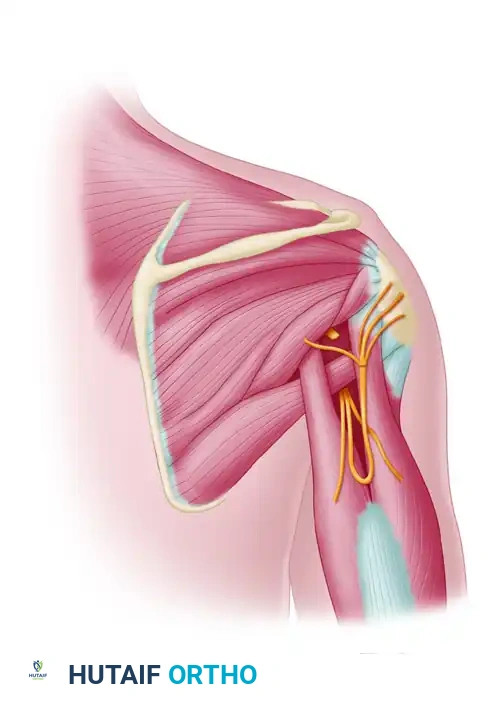
The ulnar nerve enters the hand through Guyon’s canal, bordered medially by the pisiform and laterally by the hook of the hamate. Within or just distal to the canal, the nerve bifurcates into a superficial (predominantly sensory) branch and a deep (purely motor) branch.
* Superficial Branch: Supplies the palmaris brevis muscle and provides sensation to the volar aspect of the small finger and the ulnar half of the ring finger.
* Deep Branch: Dives dorsally and radially, passing between the origins of the abductor digiti quinti (ADQ) and flexor digiti quinti (FDQ). It courses around the hook of the hamate, piercing the opponens digiti quinti (ODQ), and travels across the deep palmar arch. It innervates the hypothenar muscles, all dorsal and volar interossei, the third and fourth lumbricals, the adductor pollicis, and the deep head of the flexor pollicis brevis.
PREOPERATIVE PLANNING AND INDICATIONS
Surgical repair is indicated for any acute transection of the deep branch of the ulnar nerve. Delayed repairs (within 3 to 6 months) are also indicated, though the prognosis for motor recovery declines as motor endplates undergo irreversible degeneration.
In cases of severe crush injuries, gunshot wounds, or delayed presentations where a significant nerve gap exists, the surgeon must be prepared to perform either a nerve graft or a nerve rerouting procedure. The modified Boyes technique is the gold standard for overcoming moderate gaps (up to 2-3 cm) without the need for interpositional grafting.
SURGICAL TECHNIQUE: REPAIR OF THE DEEP BRANCH (MODIFIED BOYES TECHNIQUE)
The modified Boyes technique is a masterful approach designed to gain length by intraneurally dissecting the motor fascicles from the main ulnar nerve trunk in the distal forearm and rerouting them through the carpal tunnel.
1. Positioning and Setup
- The patient is positioned supine with the arm extended on a radiolucent hand table.
- A well-padded pneumatic tourniquet is applied to the proximal arm.
- The procedure requires standard hand instrumentation, a microsurgical tray, and an operating microscope.
2. Incision and Exposure
- Begin the incision in the midpalm, curving it distal and parallel to the thenar crease.
- Extend the incision ulnarward over the hook of the hamate to the flexion crease of the wrist.
- Proceed proximally and medially, crossing the wrist crease obliquely, and extend the incision along the ulnar aspect of the distal forearm.
- Reflect the skin flaps carefully. Identify the palmaris brevis muscle at its insertion. Divide the palmaris brevis and reflect it ulnarward.
- Surgical Warning: Extreme care must be taken during the reflection of the palmaris brevis to avoid disturbing its nerve supply, which arises from the superficial branch of the ulnar nerve.
3. Deep Dissection and Muscle Release
- Identify and retract the ulnar artery and veins toward the thumb to protect them during the deep dissection.
- To expose the deep branch as it dives into the hypothenar eminence, divide the origins of the abductor digiti quinti, flexor digiti quinti, and opponens digiti quinti muscles.
- Retract the tendons of the flexor digitorum superficialis and profundus radially.
- The course of the deep branch of the ulnar nerve is now fully exposed from the pisiform to the midpalm.

FIGURE 68-12A: Main trunk and deep branch of the ulnar nerve have been exposed, and the volar carpal ligament has been divided.
4. Distal Exposure (If Required)
- If the injury is located further distally in the palm, extend the incision toward the index metacarpal.
- Retract the flexor tendons along with the lumbrical muscles ulnarward. This maneuver allows the nerve to be identified and followed as it passes through the transverse fibers of the adductor pollicis.
5. Nerve Preparation and Freshening
- Once the zone of injury is identified, gently free the nerve proximally and distally to the point of damage. In clean, sharp lacerations, this local mobilization usually provides enough length for a tension-free suture.
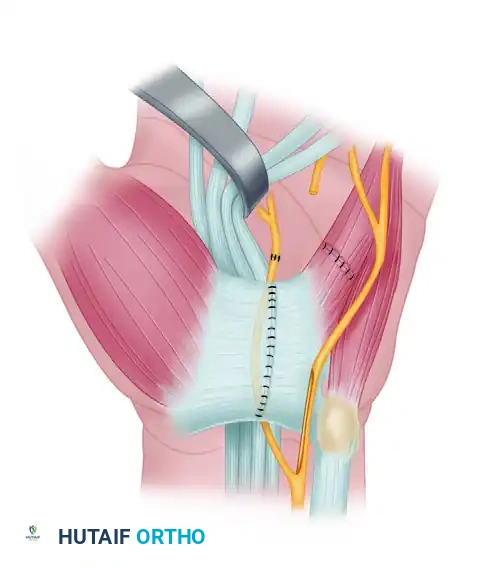
- Under the operating microscope, use microsurgical scissors or a diamond knife to freshen the ends of the nerve until healthy, pouting fascicles are visualized.
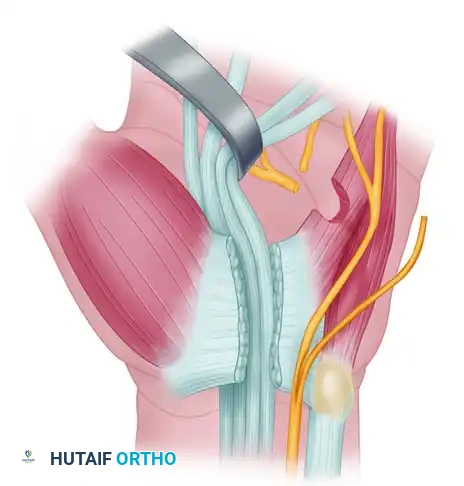
FIGURE 68-12B: Ends of the deep branch have been freshened using microsurgical instruments.
6. Management of Nerve Gaps: The Rerouting Maneuver
If a gap exists due to a gunshot wound, severe crush injury, or delayed presentation with neuroma resection, a primary repair may be under excessive tension. To avoid a nerve graft, the motor component can be rerouted.
- Intraneural Dissection: Under high magnification, carefully dissect the motor fascicular group from the main ulnar nerve trunk well into the distal forearm. This requires meticulous division of the epineurium and separation of the motor and sensory bundles.
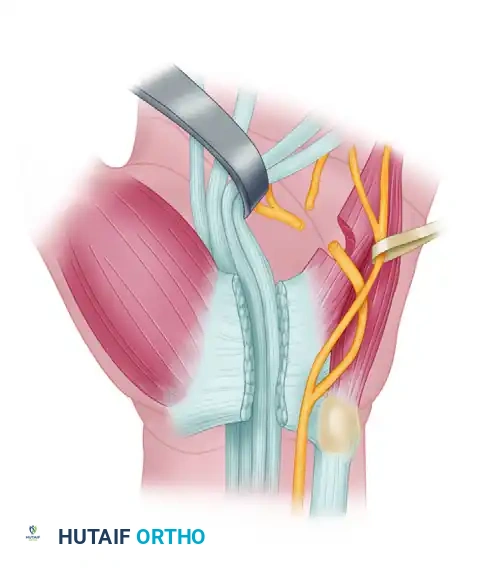
FIGURE 68-12C: The deep branch has been split intraneurally into the distal forearm to allow for independent mobilization.
- Carpal Tunnel Preparation: Divide the volar carpal ligament (transverse carpal ligament) completely. Free the ulnar bursa that lines the carpal tunnel from the ulnar side of the carpus.
- Transposition: Displace the mobilized proximal end of the deep motor branch into the carpal tunnel.
- By flexing the wrist, the proximal end can now be brought directly into the midpalm, bridging the gap.
- Note: In some instances, branches to the hypothenar muscles may still be intact. Gentle dissection and mobilization of these specific bundles allow the branches to be preserved while still permitting the main trunk of the deep branch to be rerouted.
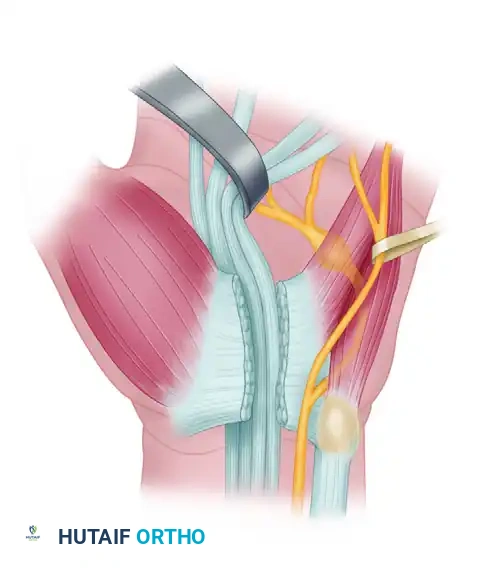
FIGURE 68-12D: The deep branch has been rerouted through the carpal tunnel, and its ends have been sutured without tension.
7. Microsurgical Neurorrhaphy
- With the nerve ends apposed without tension, perform the repair using an epiperineurial or a combination of epiperineurial and perineurial techniques.
- Utilize 8-0 or 9-0 non-absorbable monofilament suture (e.g., nylon).
- Ensure perfect rotational alignment by matching the fascicular topography and longitudinal epineurial vessels.
🚨 Surgical Pitfall
Attempting to close a nerve gap under tension will inevitably lead to ischemia at the repair site, excessive scar formation, and failure of axonal regeneration. If rerouting and wrist flexion do not provide a completely tension-free repair, you must abandon primary neurorrhaphy and proceed with an interpositional nerve graft (e.g., medial antebrachial cutaneous nerve).
SURGICAL TECHNIQUE: DORSAL BRANCH OF THE ULNAR NERVE
The dorsal branch of the ulnar nerve (dorsal ulnar cutaneous nerve) provides critical sensation to the dorsoulnar aspect of the hand and the dorsal surfaces of the small and ulnar half of the ring fingers. It typically branches from the main ulnar nerve trunk approximately 5 cm proximal to the ulnar styloid and courses dorsally, crossing the ulnar styloid superficially.
Repair Principles
- The dorsal branch is large enough at the wrist, and just distal to it, to be repaired using techniques similar to those used for a digital nerve.
- Exposure: Identify the nerve through a longitudinal or lazy-S incision over the dorsoulnar aspect of the distal forearm and wrist.
- Gaining Length: If extra length is needed to appose the severed ends (a common scenario due to the nerve's superficial and vulnerable location), the nerve can be made to branch from the main trunk more proximally.
- Perform an intraneural dissection of the dorsal branch away from the main ulnar nerve trunk in the distal forearm. Once freed proximally, the nerve can be routed more directly to the dorsum of the hand, effectively bypassing its normal anatomical curve and gaining valuable millimeters for a tension-free repair.
- Perform an epineurial repair using 8-0 or 9-0 microsuture.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a peripheral nerve repair is heavily dependent on strict adherence to postoperative immobilization and subsequent structured rehabilitation.
Phase 1: Immobilization (Weeks 0-4)
- Deep Branch Repair: If the wrist was flexed to achieve a tension-free repair (Boyes technique), the hand and wrist must be immobilized in a dorsal blocking splint. The wrist is typically held in 20 to 30 degrees of flexion, with the metacarpophalangeal (MCP) joints flexed to 70 degrees to prevent intrinsic contractures.
- Dorsal Branch Repair: The wrist should be held in extension for 3 to 4 weeks after surgery to remove tension from the dorsal ulnar cutaneous nerve.
Phase 2: Protected Mobilization (Weeks 4-8)
- The splint is gradually adjusted to bring the wrist to a neutral position over a period of 2 to 3 weeks.
- Active and active-assisted range of motion (ROM) exercises for the digits are initiated to prevent tendon adhesions, particularly if the flexor tendons were retracted or manipulated during the deep palmar dissection.
Phase 3: Strengthening and Re-education (Weeks 8+)
- Once the wrist reaches neutral and the repair site is deemed stable, progressive strengthening begins.
- Patients with deep branch injuries will require extensive motor re-education. Electrical stimulation may be utilized to maintain muscle bulk in the intrinsic muscles while awaiting reinnervation.
OUTCOMES AND PROGNOSIS
The rate of recovery following the repair of the deep branch of the ulnar nerve is dictated by the distance from the repair site to the motor endplates. Axonal regeneration occurs at a rate of approximately 1 mm per day (or 1 inch per month) after an initial latency period of 3 to 4 weeks.
Because the deep branch innervates muscles sequentially from proximal to distal (hypothenar muscles → ulnar lumbricals → interossei → adductor pollicis → first dorsal interosseous), clinical recovery will follow this anatomical progression. The first dorsal interosseous (FDI) is the most distal muscle innervated by the ulnar nerve and is therefore the last to recover.

FIGURE 68-13: Rate of recovery of voluntary function of the first dorsal interosseous muscle after repair of the deep branch of the ulnar nerve. The chart demonstrates the time interval (in months) from operation to the recovery of the first interosseous muscle across various clefts and hypothenar muscles.
Patients must be counseled that full recovery of the first dorsal interosseous and adductor pollicis may take 12 to 18 months, and in some cases, complete normal strength may never be fully restored. However, achieving protective sensation (in the case of the dorsal branch) and functional grip strength (in the case of the deep branch) is highly achievable with meticulous microsurgical technique and dedicated postoperative therapy. Regular clinical examinations monitoring the advancing Tinel's sign and serial electromyography (EMG) at 3-month intervals are recommended to track reinnervation progress.
📚 Medical References
- ulnar nerve repair: a cadaver study, J Hand Surg (Am) 23:244, 1998.
- Berger A, Millesi H: Nerve grafting, Clin Orthop Relat Res 133:49, 1978.
- Boswick JA Jr, Schneewind J, Stromberg W Jr: Evaluation of peripheral nerve repairs below elbow, Arch Surg 90:50, 1965.
- Braun RM: Epineurial nerve suture, Clin Orthop Relat Res 163:50, 1982.
- Brooks DM: Tendon transplantation in the forearm and arthrodesis of the wrist, Proc R Soc Med 42:838, 1949.
- Cabaud HE, Rodkey WG, McCarroll HR Jr, et al: Epineurial and perineurial fascicular nerve repairs: a critical comparison, J Hand Surg 1:131, 1976.
- Cabaud HE, Rodkey WG, Nemeth TJ: Progressive ultra-structural changes after peripheral nerve transection and repair, J Hand Surg 7A:353, 1982.
- Doolabh VB, Mackinnon SE: FK 506 accelerates functional recovery following nerve grafting in a rat model, Plast Reconstr Surg 103:1928, 1999.
- Ducker TB, Hayes GJ: A comparative study of the technique of nerve repair, Surg Forum 18:443, 1967.
- Edshage S: Peripheral nerve suture: a technique for improved intraneural topography: evaluation of some suture materials, Acta Chir Scand Suppl 331:1, 1964.
- Finsterbush A, Porat S, Rousso M, et al: Prevention of peripheral nerve entrapment following extensive soft tissue injury, using silicone cuffi ng: an experimental study, Clin Orthop Relat Res 162:276, 1982.
- Fu SY, Gordon T: Contributing factors to poor functional recovery after delayed nerve repair: prolonged axotomy, J Neurosci 15:3876, 1995.
- Fu SY, Gordon T: Contributing factors to poor functional recovery after delayed nerve repair: prolonged denervation, J Neurosci 15:3885, 1995.
- Ganel A, Farine I, Aharonson Z, et al: Intraoperative nerve fascicle identifi cation using choline acetyltransferase: a preliminary report, Clin Orthop Relat Res 165:228, 1982.
- Giddins GEB, Wade PJF, Amis AA: Primary nerve repair: strength of repair with different gauges of nylon suture material, J Hand Surg 14B:301, 1989.
- Grabb WC, Bement SL, Koepke GH, et al: Comparison methods of
You Might Also Like