Upper Extremity Compartment Syndrome and Volkmann Ischemic Contracture: A Master Surgical Guide
Key Takeaway
Upper extremity compartment syndrome is a surgical emergency necessitating immediate fasciotomy to prevent irreversible muscle necrosis and subsequent Volkmann's ischemic contracture. This comprehensive guide details the pathophysiology, diagnostic criteria, and step-by-step surgical techniques for forearm and hand fasciotomies. Furthermore, it outlines the reconstructive management of established Volkmann's contracture and adducted thumb deformities, providing evidence-based protocols for orthopedic residents and practicing consultants.
Introduction to Upper Extremity Compartment Syndrome
Acute compartment syndrome (ACS) of the upper extremity represents a catastrophic orthopedic emergency. It is defined by an elevation of interstitial pressure within a closed osteofascial compartment that compromises microvascular perfusion, leading to tissue ischemia, muscle necrosis, and nerve damage. If left untreated or inadequately decompressed, ACS progresses to the devastating sequela known as Volkmann’s ischemic contracture—a condition characterized by irreversible fibrosis, muscle contracture, and profound functional impairment of the limb.
While fractures of the distal radius and forearm (both-bone fractures) are the most common etiologies, ACS can also arise from crush injuries, vascular trauma, prolonged tourniquet times, intravenous regional anesthesia complications, and even low-velocity gunshot wounds. Recognizing the insidious onset of this condition and executing a flawless, anatomically sound fasciotomy is a fundamental requirement for every practicing orthopedic surgeon.
Clinical Pearl: The deep volar compartment of the forearm, housing the flexor digitorum profundus (FDP) and flexor pollicis longus (FPL), is the most frequently and severely affected compartment in upper extremity ACS. Ischemia here dictates the classic flexed posture seen in established Volkmann’s contracture.
Pathophysiology and Biomechanics
The pathophysiology of compartment syndrome is governed by the arteriovenous gradient theory. As intracompartmental pressure rises—whether from decreased compartment volume (e.g., tight casts, tight fascial closures) or increased compartment content (e.g., hemorrhage, edema, reperfusion injury)—venous outflow is obstructed. This venous congestion further increases capillary hydrostatic pressure, leading to a vicious cycle of fluid transudation, worsening tissue edema, and eventual collapse of the arteriolar microcirculation.
Skeletal muscle can tolerate ischemia for approximately 4 hours before irreversible damage begins. By 8 hours, irreversible necrosis is virtually guaranteed. Peripheral nerves exhibit neuropraxia within 30 minutes of ischemia and sustain irreversible axonal damage after 12 to 24 hours.
Diagnostic Principles
The diagnosis of ACS remains primarily clinical, supplemented by intracompartmental pressure (ICP) monitoring in equivocal cases or obtunded patients.
Clinical Evaluation
The classic "5 Ps" (Pain, Pallor, Pulselessness, Paresthesia, Paralysis) are historically taught but are dangerously late indicators.
* Pain out of proportion to the primary injury is the earliest and most reliable symptom.
* Pain with passive stretch of the muscles within the involved compartment (e.g., passive extension of the fingers stretching the volar flexors) is the most sensitive clinical sign.
* Palpable tenseness or a "wood-like" feeling of the compartment is highly suggestive but subjective.
Intracompartmental Pressure (ICP) Monitoring
When clinical signs are ambiguous, particularly in polytrauma, pediatric, or comatose patients, ICP measurement is mandatory. Techniques include the use of a Wick catheter, slit catheter, or a solid-state transducer (e.g., Stryker needle).
Surgical Warning: Absolute pressure readings are less reliable than the differential pressure ($\Delta$P). A $\Delta$P (Diastolic Blood Pressure minus Intracompartmental Pressure) of less than 30 mm Hg is an absolute indication for emergent fasciotomy.
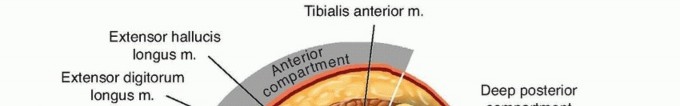
Surgical Anatomy of the Forearm Compartments
Successful decompression requires an intimate understanding of forearm fascial anatomy. The forearm is divided into three primary compartments:
1. Volar Compartment: Subdivided into superficial and deep spaces. The deep space contains the FDP, FPL, and pronator quadratus.
2. Dorsal Compartment: Contains the extensor muscles.
3. Mobile Wad Compartment: Contains the brachioradialis, extensor carpi radialis longus (ECRL), and extensor carpi radialis brevis (ECRB).
Operative Technique: Forearm and Hand Fasciotomy
The goal of surgery is the complete, unhindered release of all fascial envelopes. Prophylactic or limited incisions have no role in the management of ACS.
Patient Positioning and Preparation
- Position: Supine with the affected arm extended on a radiolucent hand table.
- Tourniquet: A pneumatic tourniquet may be applied but should not be inflated unless catastrophic hemorrhage is encountered. Inflating the tourniquet exacerbates ischemia.
- Anesthesia: General anesthesia is preferred. Regional blocks are generally avoided preoperatively as they mask the clinical symptoms of compartment syndrome, complicating postoperative evaluation.
The Volar Approach (Extended Henry)
The volar incision must decompress the superficial volar, deep volar, and mobile wad compartments, while also releasing the carpal tunnel.
- Incision Design: Begin just proximal to the medial epicondyle. Extend the incision distally and obliquely across the antecubital fossa to the mobile wad. Continue distally along the volar forearm, curving ulnarly at the wrist to avoid crossing the flexion crease at a right angle, and extend into the palm along the thenar crease.
- Superficial Dissection: Incise the skin and subcutaneous tissue. Identify and protect the medial antebrachial cutaneous nerve and the palmar cutaneous branch of the median nerve.
- Fascial Release: Incise the superficial fascia over the flexor carpi ulnaris (FCU) and flexor carpi radialis (FCR).
- Deep Compartment Release: Retract the superficial flexors (FDS) to expose the deep compartment. The fascia over the FDP and FPL must be meticulously incised.
- Epimysiotomy: If the muscle bellies remain tight and pale after fasciotomy, perform an epimysiotomy (incising the epimysium directly over the muscle belly) to allow further expansion.
- Carpal Tunnel Release: Divide the transverse carpal ligament completely to decompress the median nerve, as forearm swelling invariably increases carpal tunnel pressure.
The Dorsal Approach
While the volar approach decompresses the majority of the forearm, a separate dorsal incision is often required to release the dorsal compartment.
- Incision Design: Make a straight longitudinal incision from the lateral epicondyle to the midline of the distal radioulnar joint.
- Fascial Release: Incise the dorsal fascia longitudinally. Identify the interval between the extensor digitorum communis (EDC) and the extensor carpi radialis brevis (ECRB) to ensure both the mobile wad and dorsal compartments are fully decompressed.
Hand Fasciotomy
Compartment syndrome of the hand involves the interosseous, thenar, and hypothenar compartments.
- Dorsal Interosseous Release: Make two longitudinal dorsal incisions over the second and fourth metacarpals. Retract the extensor tendons to access and incise the fascia of the dorsal and volar interossei.
- Thenar and Hypothenar Release: Make a longitudinal incision along the radial border of the first metacarpal for the thenar compartment, and along the ulnar border of the fifth metacarpal for the hypothenar compartment.
Postoperative Management and Wound Closure
Fasciotomy wounds must never be closed primarily.
- Initial Dressing: Apply sterile, non-adherent dressings. Negative pressure wound therapy (NPWT) is highly recommended at 50 to 75 mm Hg continuous pressure to manage exudate and reduce edema.
- The Vessel Loop Shoelace Technique: To prevent excessive skin retraction, vessel loops can be interlaced through skin staples placed along the wound edges. This provides gentle, continuous traction, facilitating delayed primary closure.
- Definitive Closure: Return to the operating room at 48 to 72 hours for a second look, debridement of any frankly necrotic muscle, and attempted closure. If delayed primary closure is impossible by day 5 to 7, split-thickness skin grafting is mandatory.
Pitfall: Aggressive attempts to pull fascial edges together during delayed closure will recreate the compartment syndrome. If the wound does not close without tension, use a skin graft.
Volkmann’s Ischemic Contracture
When compartment syndrome is missed or inadequately treated, the necrotic muscle is gradually replaced by dense, inelastic scar tissue. This results in Volkmann’s ischemic contracture, classically presenting with elbow flexion, forearm pronation, wrist flexion, metacarpophalangeal (MCP) joint extension, and interphalangeal (IP) joint flexion.
Tsuge Classification and Management Strategy
The severity of Volkmann’s contracture dictates the reconstructive approach, as classified by Tsuge:
1. Mild Type
- Pathology: Localized ischemia, primarily affecting the FDP (usually the middle and ring fingers).
- Presentation: Flexion contracture of two or three fingers. Sensation is typically intact.
- Surgical Treatment: Fractional lengthening of the affected flexor tendons at the musculotendinous junction.
2. Moderate Type
- Pathology: Involvement of the FDP, FPL, and FDS. The median and ulnar nerves often exhibit compression neuropathy due to surrounding fibrosis.
- Presentation: Classic claw hand deformity with wrist flexion. Sensory deficits are common.
- Surgical Treatment:
- Muscle Slide Operation (Page-Scaglietti Technique): This involves the proximal release of the flexor-pronator origin from the medial epicondyle. The entire muscle mass is advanced distally by 2 to 3 centimeters.
- Neurolysis: Meticulous neurolysis of the median and ulnar nerves is performed simultaneously to restore sensation and intrinsic function.
3. Severe Type
- Pathology: Extensive necrosis of both flexor and extensor compartments. Profound nerve ischemia.
- Presentation: A severely contracted, non-functional, insensate limb.
- Surgical Treatment:
- Radical Debridement: Excision of all fibrotic, non-contractile muscle.
- Free Functional Muscle Transfer (FFMT): The gold standard for severe cases. The gracilis muscle is harvested with its neurovascular pedicle. It is transferred to the forearm, with the obturator nerve anastomosed to the anterior interosseous nerve (AIN) or a fascicle of the median nerve, and the vascular pedicle anastomosed to the radial or ulnar artery.
- Tendon Transfer: If local expendable muscles (e.g., brachioradialis, ECRL) survived, they may be transferred to the FDP to restore finger flexion.
Management of the Adducted Thumb Contracture
A frequent concomitant deformity in severe Volkmann’s contracture, or following direct crush injuries to the hand, is the adducted thumb. Fibrosis of the adductor pollicis and the first dorsal interosseous muscle obliterates the first web space, destroying the hand's ability to grasp or pinch.
Pathoanatomy
The contracture involves not only the muscle bellies but also the overlying fascia, the skin of the first web space, and occasionally the carpometacarpal (CMC) joint capsule.
Surgical Reconstruction Techniques
Restoring the first web space requires a stepwise approach, addressing skin, fascia, muscle, and joint capsule.
1. Skin and Web Space Deepening
Simple Z-plasties are rarely sufficient for severe contractures.
* Four-Flap Z-plasty or Jumping Man Flap: Provides superior deepening and widening of the web space.
* Sliding Flap (Brand and Milford Technique): A dorsal rotational flap is mobilized to resurface the deepened web space, shifting the tension away from the critical apex of the web.
* Skin Grafting: If local flaps are inadequate, a full-thickness skin graft (FTSG) harvested from the groin or lower abdomen is utilized to cover the defect after release.
2. Musculofascial Release
- Adductor Pollicis Release: The origin of the adductor pollicis is released from the third metacarpal shaft. In severe cases, the transverse head may need to be completely excised.
- First Dorsal Interosseous Release: The fascial envelope of the first dorsal interosseous is released dorsally.
3. Capsulotomy
If the thumb remains adducted after soft tissue and muscle release, a dorsal capsulotomy of the trapeziometacarpal (CMC) joint is performed.
Surgical Warning: Following release, the thumb must be pinned in wide abduction and opposition using a smooth Kirschner wire (K-wire) driven across the first and second metacarpals. This maintains the web space during the critical 4-to-6-week healing phase.
Conclusion
The management of upper extremity compartment syndrome demands high clinical vigilance, rapid surgical intervention, and an uncompromising approach to fascial decompression. The transition from an acute compartment syndrome to a Volkmann’s ischemic contracture represents a failure of early recognition, transforming a reversible ischemic insult into a lifelong reconstructive challenge.
For the orthopedic surgeon, mastery of the volar and dorsal forearm approaches, coupled with a deep understanding of reconstructive salvage procedures—ranging from muscle slides to free functional muscle transfers—is essential. Strict adherence to these evidence-based principles ensures the preservation of limb viability and the restoration of complex hand mechanics.
You Might Also Like