PHYSEAL-SPARING RECONSTRUCTION OF THE ANTERIOR CRUCIATE LIGAMENT
The management of anterior cruciate ligament (ACL) ruptures in skeletally immature patients presents a unique biomechanical and biological challenge. Traditional transphyseal reconstruction techniques carry a well-documented risk of iatrogenic physeal injury, potentially leading to premature growth arrest, angular deformities, or limb-length discrepancies. To mitigate these catastrophic complications, physeal-sparing techniques have been developed for prepubescent children with significant remaining growth potential.
The procedure popularized by Kocher, Garg, and Micheli is an arthroscopically assisted, physeal-sparing, combined intraarticular and extraarticular reconstruction of the anterior cruciate ligament utilizing an autogenous iliotibial band (ITB) graft. This technique is a modern, pediatric-specific modification of the classic combined intraarticular and extraarticular reconstruction originally described by MacIntosh and Darby.
Key modifications in the Kocher-Micheli technique include its specific application in skeletally immature patients, the integration of arthroscopic assistance to minimize arthrotomy morbidity, optimized graft fixation methods, and a tailored, accelerated rehabilitation protocol. Because the patient cohort is young, rehabilitation must be meticulously geared toward the psychological and physiological maturity of the child.
🔪 Surgical Technique: The Kocher, Garg, and Micheli Procedure
Clinical Pearl: Preoperative radiographic evaluation of bone age (e.g., using the Greulich and Pyle atlas) is mandatory to confirm skeletal immaturity and justify a strictly physeal-sparing approach.
Anesthesia and Positioning
* The procedure is performed under general anesthesia and is typically scheduled as an overnight observation procedure to monitor for acute postoperative pain and neurovascular status.
* Position the child supine on the operating table.
* Apply a well-padded pneumatic tourniquet around the proximal aspect of the operative thigh.
* Perform a thorough examination under anesthesia (EUA) to definitively confirm ACL insufficiency (positive Lachman, pivot-shift, and anterior drawer tests) and assess for concurrent collateral ligament or meniscal pathology.
Graft Harvest and Preparation
* Make an oblique incision of approximately 6 cm extending from the lateral joint line to the superior border of the iliotibial band.
* Carefully separate the iliotibial band proximally from the overlying subcutaneous tissue using a periosteal elevator, creating a working space under the skin of the lateral thigh.
* Incise the anterior and posterior borders of the iliotibial band. Carry these parallel incisions proximally under the skin using a curved meniscotome or specialized tendon stripper to harvest a robust strip of tissue.
* Detach the iliotibial band proximally under the skin, ensuring adequate length for both intraarticular passage and extraarticular tenodesis.
* Crucial Step: Leave the iliotibial band attached distally at its anatomic insertion on Gerdy’s tubercle. This preserves the distal vascular supply and provides a native, immensely strong distal anchor.
* Dissect distally to carefully separate the harvested portion of the iliotibial band from the underlying joint capsule and the lateral patellar retinaculum.
* Tubularize the free proximal end of the iliotibial band using a running whipstitch with a heavy, nonabsorbable suture (e.g., No. 5 Ethibond; Ethicon, Johnson & Johnson, Somerville, NJ). This tubularization facilitates smooth passage through the joint.
Arthroscopic Preparation and Femoral Routing
* Establish standard anterolateral and anteromedial arthroscopic portals.
* Perform a comprehensive diagnostic arthroscopy. Treat any concomitant meniscal tears (repair is strongly preferred in pediatric patients) or chondral injuries.
* Carefully excise the ruptured remnants of the anterior cruciate ligament to prevent postoperative cyclops lesions and impingement.
* Identify the "over-the-top" position on the posterior aspect of the lateral femoral condyle, as well as the "over-the-front" position under the anterior intermeniscal ligament.
Surgical Warning: Perform only a minimal notchplasty. Aggressive bone resection in the intercondylar notch risks iatrogenic injury to the perichondrial ring of the distal femoral physis, which lies in dangerously close proximity to the over-the-top position.
- Pass the free, tubularized end of the iliotibial band graft through the over-the-top position. This can be achieved using a full-length curved clamp or a two-incision, rear-entry guide, eventually retrieving the graft out through the anteromedial portal.
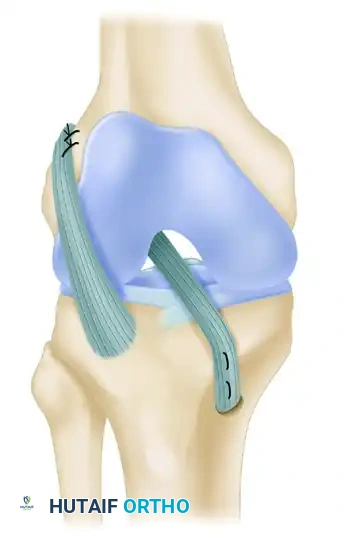
Tibial Routing and Graft Fixation
* Make a second incision of approximately 4.5 cm over the proximal medial aspect of the tibia, centered over the region of the pes anserinus. Carry the dissection sharply through the subcutaneous tissue down to the periosteum.
* Introduce a curved clamp from this medial tibial incision into the joint space, passing under the intermeniscal ligament.
* Using a curved rat-tail rasp, create a small, shallow groove in the anteromedial aspect of the proximal tibial epiphysis (under the intermeniscal ligament). This groove allows the tibial graft placement to sit more posteriorly, improving the biomechanical vector of the reconstructed ligament.
* Pull the free end of the graft through the joint, routing it under the intermeniscal ligament, seating it within the newly created anteromedial epiphyseal groove, and extracting it out through the medial tibial incision.
* Femoral Fixation (Extraarticular): Place the knee in 90 degrees of flexion and 15 degrees of external rotation. Fix the graft on the femoral side through the lateral incision. Use heavy mattress sutures to secure the graft to the lateral femoral condyle at the insertion of the lateral intermuscular septum.
* Tibial Fixation: Transition the knee to 20 degrees of flexion. Apply firm, physiological tension to the graft.
* Confirm the location of the proximal tibial physis using intraoperative fluoroscopy. Make a periosteal incision strictly distal to the physis.
* Create a bony trough in the proximal medial tibial metaphyseal cortex. Suture the graft securely to the robust periosteum at the roughened margins of the trough using multiple mattress sutures.
Postoperative Care and Rehabilitation Protocol
Rehabilitation in the pediatric population requires a delicate balance between protecting the healing graft and preventing arthrofibrosis, all while managing the inherent non-compliance risks of young children.
- Weight Bearing: Touch-down weight bearing (TDWB) is permitted for the first 6 weeks postoperatively.
- Early Motion: Immediate mobilization from 0 to 90 degrees is allowed for the first 2 weeks. Continuous passive motion (CPM) machines set from 0 to 90 degrees are highly recommended during this initial 2-week phase. CPM not only initiates motion but is highly effective at overcoming the fear and anxiety associated with postoperative movement in young children.
- Bracing: A protective hinged knee brace is mandated for 6 weeks. For the first 2 weeks, the brace hinges are locked to limit motion from 0 to 90 degrees.
- Progressive Physical Therapy: Following the initial 2 weeks, therapy progresses to achieve full range of motion. The first 3 months include patellar mobilization, electrical stimulation, pool therapy (hydrotherapy), proprioception drills, and closed-kinetic-chain strengthening exercises.
- Advanced Rehabilitation: After 3 months, the patient may progress to straight-line jogging, plyometrics, sport-cord resistance training, and sport-specific agility drills.
- Return to Play: Return to full, unrestricted activity—including cutting and pivoting sports—is generally permitted at 6 months postoperatively, provided strength and functional testing criteria are met. A custom-made functional derotation knee brace is routinely prescribed for use during cutting and pivoting activities for the first 2 years following the return to sports.
COMPLICATIONS OF ANTERIOR CRUCIATE LIGAMENT RECONSTRUCTION
Despite advancements in surgical technique and graft fixation, ACL reconstruction is not without complications. Understanding the etiology of failure is paramount for the practicing orthopedic surgeon.
Graft Selection and Long-Term Outcomes
Five-year follow-up studies comparing autograft bone-patellar tendon-bone (BPTB) and hamstring autografts demonstrate comparable results regarding overall stability and clinical failure rates. While BPTB grafts exhibit slightly superior initial stiffness and ultimate tensile strength, the functional outcomes remain statistically similar.
Allograft utilization has also evolved. Modern studies at 5- and 7-year follow-ups show allograft outcomes approaching those of autografts. This improvement is largely attributed to refined graft procurement and low-dose irradiation sterilization techniques, which have drastically reduced the incidence of postoperative effusions, bacterial contamination, and apparent immunological graft rejection.
When graft failure is strictly defined as structural rupture, failure rates have stabilized at approximately 7% to 8% at 5-year follow-up. However, if failure is defined by broader clinical parameters—such as KT-1000 arthrometer side-to-side differences >3mm, subjective "giving way" episodes, or failure to return to pre-injury sporting levels—the reported failure rates expand dramatically, ranging from 5% to 52%.
The Burden of Revision Surgery
The incidence of revision ACL surgery continues to rise. This trend is multifactorial, driven by more rigorous follow-up protocols, higher patient expectations, increased athletic demands, and the younger age at which primary reconstructions are now performed.
The socioeconomic and psychological costs of ACL failure are profound. Patients face additional surgical morbidity, prolonged rehabilitation, loss of employment income, and, for student-athletes, the potential loss of collegiate scholarships. Physiologically, chronic instability resulting from a failed ACL reconstruction leads to devastating secondary joint damage. Meniscal tears occur in approximately 40% of patients at 1 year, 60% at 5 years, and 80% at 10 years post-failure. This directly correlates with the incidence of irreversible degenerative joint disease (osteoarthritis) observed at the 10-year mark.
Technical Errors and Prevention
Most surgical failures are preventable through meticulous preoperative planning, strict adherence to anatomic principles, and precise surgical execution.
Tunnel Malposition and Impingement
Surgical failure is most frequently caused by nonphysiometric (non-anatomic) tunnel placement, leading to graft impingement, excessive tension, and ultimate mechanical failure.
* Prevention: Careful identification of native anatomical landmarks is essential. It is highly recommended to initially ream the tunnels with a reamer 2 mm smaller than the definitive graft diameter. This allows for minor directional adjustments using a rasp or eccentric reaming before the final tunnel is committed.
* Notch Impingement: An adequate internal notchplasty must be performed. Intraoperatively, the guidewire must be visualized through a full range of motion to ensure it does not impinge on the roof (Blumensaat's line) or the lateral wall of the intercondylar notch during full extension. Furthermore, the femoral tunnel must not be placed too far posteriorly, which risks impingement against the posterior cruciate ligament (PCL).
Graft Harvest Complications
Stress fractures of the patella can be mitigated by careful BPTB harvest techniques. Bone cuts must be made straight and strictly in line with the tendon fibers. The depth of the patellar bone block should be carefully controlled, and the length should ideally be 20 to 23 mm. The patellar defect should be bone-grafted using reamings from the tunnel preparation to minimize the risk of late postoperative patellar fracture.
Postoperative Complications
Arthrofibrosis and Motion Loss
Arthrofibrosis is a debilitating complication characterized by a restricted range of motion, particularly a loss of terminal extension.
* Management: Initial treatment consists of aggressive, supervised physical therapy (three times weekly), home exercises (three times daily), and nonsteroidal anti-inflammatory drugs (NSAIDs). Prone hangs are specifically emphasized to regain terminal extension.
* If the patient fails to progress over 4 to 6 weeks, or has less than 90 degrees of flexion at 6 weeks postoperatively, gentle manipulation under anesthesia (MUA) and arthroscopic lysis of adhesions should be strongly considered.
* Always review postoperative radiographs to rule out mechanical blocks. A lateral radiograph in full extension is critical to ensure the tibial tunnel is positioned posterior to the slope of the intercondylar roof and that femoral fixation hardware is appropriately seated in the posterior aspect of the notch.
Postoperative Infection
While uncommon in arthroscopic ACL reconstruction (incidence <1%), postoperative septic arthritis is a surgical emergency.
* Diagnosis: Suspect infection if a patient presents 5 to 6 days postoperatively with recurrent fever, escalating disproportionate pain, sudden loss of knee motion, and localized erythema/calor. Joint aspiration is mandatory; a synovial fluid white blood cell (WBC) count ≥ 20,000/µL with a high polymorphonuclear cell percentage is highly suggestive of infection.
* Treatment: Emergent arthroscopic irrigation and extensive débridement are required. Crucially, if the graft remains intact and structurally sound, it should be retained. The joint must be thoroughly lavaged. If symptoms do not drastically improve, repeat irrigation and débridement should be performed at 48 to 72 hours.
* Intravenous organism-specific antibiotics are administered for 2 to 3 weeks, followed by oral antibiotics to complete a comprehensive 6-week course.
POSTERIOR CRUCIATE LIGAMENT (PCL) RECONSTRUCTION
Anatomy and Biomechanics
The posterior cruciate ligament is the primary restraint to posterior tibial translation. It is a robust structure, possessing 1.5 to 2 times the ultimate tensile strength of the anterior cruciate ligament. The broad femoral footprint of the PCL spans approximately 3 cm², making true physiometric (anatomic) reconstruction technically demanding.
The PCL consists of two primary functional bundles and the variable meniscofemoral ligaments (ligaments of Humphrey and Wrisberg):
1. Anteromedial (AM) Band: The larger component, which becomes maximally taut in knee flexion.
2. Posterolateral (PL) Band: While classical teaching notes the PL bundle is tight in extension, specific biomechanical evaluations note its remarkable strength and stiffness, contributing significantly to rotatory stability.
Mechanism of Injury and Clinical Evaluation
The most common mechanism for PCL injury in athletes is a direct blow to the anterior tibia with the knee flexed (e.g., a dashboard injury or falling onto a flexed knee), resulting in forced hyperflexion and posterior tibial translation.
Importantly, isolated PCL injuries are relatively rare; they are frequently associated with concurrent ligamentous damage, most notably to the posterolateral corner (PLC).
Clinical Examination:
* Posterior Drawer Test: The hallmark of PCL insufficiency. A posterior step-off or a posterior drawer of 1 cm (Grade III) indicates a complete rupture.
* Dial Test: If the posterior drawer does not improve with internal rotation, or if there is a 10-degree increase in external rotation at both 30 and 90 degrees of knee flexion compared to the contralateral side, an associated posterolateral corner (PLC) injury must be highly suspected.
Management Strategies
The treatment of PCL injuries remains a topic of ongoing orthopedic debate, heavily influenced by the chronicity of the injury and the presence of concomitant ligamentous damage.
Conservative Management
For isolated, partial PCL injuries (Grade I or II, presenting with 1+ to 2+ posterior laxity), conservative management is often successful. Protocols championed by Shelbourne and Muthukaruppan emphasize initial knee extension bracing and a protective rehabilitation program that strictly avoids active hamstring strengthening (which exacerbates posterior tibial translation). Long-term outcome studies indicate that subjective functional scores do not significantly deteriorate over time in isolated injuries, and clinical outcomes do not strictly correlate with the initial degree of measured laxity.
Operative Reconstruction
For high-demand, active, younger individuals presenting with significant Grade III posterior laxity, or those with combined multi-ligamentous injuries (especially PCL + PLC), surgical reconstruction has become the gold standard of care. The goal of surgery is to restore native joint kinematics, prevent chronic posterior subluxation, and mitigate the early onset of medial compartment and patellofemoral osteoarthritis. The choice of surgical technique—ranging from single-bundle to double-bundle reconstructions, and transtibial versus tibial inlay approaches—must be tailored to the specific anatomical demands of the patient.
