Introduction & Epidemiology
Pediatric scoliosis encompasses a spectrum of spinal deformities characterized by a three-dimensional deviation of the vertebral column. Clinically, it presents with lateral curvature in the coronal plane, rotation in the axial plane, and alterations in the sagittal plane. Affecting approximately 2-3% of adolescents, the most prevalent form is Adolescent Idiopathic Scoliosis (AIS), typically presenting after 10 years of age. Other etiologies include Early-Onset Scoliosis (EOS) – defined as onset before 10 years of age, congenital scoliosis resulting from vertebral malformations, and neuromuscular scoliosis associated with conditions like cerebral palsy or muscular dystrophy. Syndromic scoliosis, linked to conditions such as Marfan's or neurofibromatosis, represents another distinct category.
The natural history of scoliosis is crucial for management decisions. Progression risk is multifactorial, influenced by curve magnitude at presentation, skeletal maturity (Risser sign, menarche status), and curve pattern. While most curves remain stable or progress minimally, significant progression, particularly in skeletally immature patients, can lead to functional impairment, pain, psychological distress, and, in severe cases, cardiopulmonary compromise. Understanding the specific etiology and its associated prognosis is paramount for guiding intervention, whether conservative or operative. Our focus herein is on the critical assessment and management of curves necessitating surgical intervention, transcending the initial diagnostic 'degrees' to the 'what's next' in definitive care.
Surgical Anatomy & Biomechanics
A comprehensive understanding of spinal surgical anatomy and biomechanics is fundamental to successful scoliosis correction. The vertebral column is a complex osteoligamentous structure designed for stability, mobility, and protection of the neural elements.
- Vertebral Anatomy: Each typical vertebra comprises a body, pedicles, laminae, and posterior elements (spinous and transverse processes, articular facets). In scoliosis, these structures undergo characteristic adaptive changes, including vertebral body wedging, pedicle elongation and thinning on the convex side, and compensatory rotation. The Cobb angle, while a primary measure of coronal deformity, fails to fully capture the 3D nature, which includes rotational deformity and often a flattened thoracic kyphosis or compensatory lumbar lordosis.
- Ligamentous Structures: The anterior and posterior longitudinal ligaments, ligamentum flavum, interspinous, and supraspinous ligaments contribute significantly to spinal stability. During deformity correction, controlled release or distraction of these structures can facilitate correction.
- Musculature: The paraspinal musculature plays a dynamic role. Asymmetry in muscle tension or weakness, as seen in neuromuscular scoliosis, can profoundly influence curve progression and stability. Subperiosteal dissection during surgery necessitates meticulous attention to preserving the muscle envelope where possible to minimize post-operative paraspinal muscle atrophy and improve long-term fusion success.
- Spinal Cord and Nerves: The spinal cord and nerve roots are highly susceptible to injury during deformity correction. The critical blood supply to the spinal cord, particularly the artery of Adamkiewicz, must be identified and protected. Neuromonitoring (SSEP and MEP) is indispensable during these procedures.
- Biomechanics of Deformity: Scoliotic curves involve a complex interplay of compressive and tensile forces. The Hueter-Volkmann principle, describing how compression inhibits and tension stimulates growth, partially explains curve progression in the growing spine. Surgical correction aims to restore spinal balance in the coronal and sagittal planes, detorque the spine, and achieve solid arthrodesis. Instrumentation constructs leverage principles of distraction, compression, translation, and direct vertebral rotation (DVR) to achieve these goals. The goal is not just a straight spine, but a balanced spine that preserves physiological sagittal contours (thoracic kyphosis, lumbar lordosis) and avoids proximal or distal junctional issues.
Indications & Contraindications
The decision for operative intervention in pediatric scoliosis is complex, balancing the risks of surgery against the natural history of the deformity and its potential impact on quality of life, pulmonary function, and pain.
Indications for Operative Intervention
Surgical correction is primarily indicated for progressive curves that are unresponsive to conservative management or curves that have reached a magnitude associated with adverse outcomes. The specific thresholds vary by etiology, skeletal maturity, and curve pattern.
Table: Operative vs. Non-Operative Indications
| Factor | Operative Indications | Non-Operative Indications |
|---|---|---|
| Curve Magnitude | AIS: >45-50° in skeletally immature or progressing curves. EOS: >40-50° with failure of conservative measures (casting/bracing). Neuromuscular: >40-50° with pelvic obliquity or functional impairment. Congenital: Progressive curves of any magnitude threatening neurological function or severe trunk imbalance. | AIS: <40-45° in skeletally immature patients (observational or bracing). <50-55° in skeletally mature patients (observational). EOS: <40° with potential for growth modulation (serial casting, bracing). Neuromuscular: Mild curves without functional deficit. |
| Progression | Documented progression of ≥5-10° despite bracing. | Stable curves without significant progression. |
| Skeletal Maturity | Immature or mature, if curves meet magnitude criteria and cause significant functional issues. | Skeletally immature patients with smaller curves where bracing may alter natural history. |
| Curve Pattern | Severe thoracic curves (thoracic kyphosis > 40°, apex T7-T10) threatening pulmonary function. Significant double major or triple major curves leading to trunk decompensation. | Balanced, non-progressive curves. |
| Patient Symptoms | Intractable back pain unresponsive to conservative measures. Significant cosmetic deformity leading to psychosocial distress. Neurological deficit related to spinal deformity (e.g., tethered cord in congenital scoliosis). | Mild, manageable back pain. Minimal cosmetic concern. |
| Associated Issues | Significant truncal decompensation. Progressive pelvic obliquity (neuromuscular). Balance issues interfering with ambulation. | No associated cardiopulmonary or neurological compromise. |
| Failed Conservative Treatment | Continued curve progression despite appropriate bracing/casting protocols. | Successful arrest of curve progression with bracing/casting. |
Contraindications for Operative Intervention
Absolute contraindications are rare but include:
- Uncontrolled Systemic Infection: Active osteomyelitis, sepsis, or localized soft tissue infection at the operative site.
- Unstable Medical Condition: Severe cardiopulmonary compromise, coagulopathies, or other life-threatening medical comorbidities that cannot be optimized pre-operatively and render the anesthetic or surgical risk prohibitively high.
- Severe Neurological Deficit (Post-Op): For some patients with profound pre-existing neurological deficits where further instrumentation offers no functional benefit and carries significant risk.
- Lack of Informed Consent: Inability to obtain appropriate informed consent from the patient or legal guardian.
Relative contraindications often involve optimizing the patient's condition to reduce surgical risk:
- Poor Nutritional Status: Hypoalbuminemia can impair wound healing and increase infection risk.
- Poor Pulmonary Function: Pre-operative pulmonary optimization, including bronchodilators, chest physiotherapy, and consideration of non-fusion techniques (e.g., growing rods, VEPTR) or staged procedures for very severe cases.
- Anticoagulation: Careful management of antiplatelet or anticoagulant medications pre-operatively.
- Previous Spinal Surgery/Radiation: May complicate exposure and increase risk of complications.
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is crucial for minimizing complications and optimizing outcomes in pediatric scoliosis surgery.
Pre-Operative Evaluation
-
Clinical Assessment:
- Detailed History: Curve progression, pain, neurological symptoms, cardiopulmonary history, previous treatments, and psychosocial impact.
- Physical Examination: Assess trunk balance, shoulder height, rib hump, pelvic obliquity, limb length discrepancy, neurological status (motor, sensory, reflexes, clonus), and flexibility of the curve (side-bending radiographs).
- Skeletal Maturity: Risser sign, Tanner staging, menarcheal status.
-
Radiographic Assessment:
- Standing Posteroanterior (PA) and Lateral Whole Spine Radiographs: Essential for Cobb angle measurement, assessment of sagittal balance (thoracic kyphosis, lumbar lordosis), and identification of transitional vertebrae.
- Side-Bending Radiographs: Evaluate curve flexibility, identify structural vs. compensatory curves, and aid in determining fusion levels.
- Supine Push-Prone or Traction Radiographs: Further assess flexibility and potential for correction.
- CT Scan: Indicated for complex congenital deformities, pseudoarthrosis, or anomalous vertebral anatomy to accurately assess pedicle morphology and canal dimensions, particularly when pedicle screw fixation is planned.
- MRI Scan: Mandatory for patients with atypical curves (e.g., left thoracic curve in AIS), rapid progression, neurological symptoms, pain, congenital deformities, or early-onset scoliosis to rule out intraspinal pathology (e.g., syrinx, tethered cord, tumor, Chiari malformation).
- 3D Reconstruction: Can be invaluable for severe, complex deformities, aiding in pedicle screw trajectory planning.
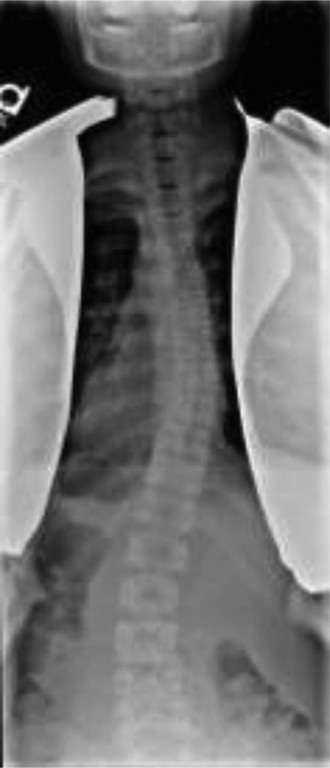
 Illustrative pre-operative radiographic series demonstrating severe pediatric scoliosis.
Illustrative pre-operative radiographic series demonstrating severe pediatric scoliosis.
-
Pulmonary Function Tests (PFTs): Essential for patients with severe thoracic curves (>70-80°), neuromuscular scoliosis, or other respiratory comorbidities. May necessitate pre-operative pulmonary optimization.
- Cardiac Evaluation: Especially for syndromic scoliosis (e.g., Marfan's, Ehlers-Danlos) or significant neuromuscular disease.
- Blood Work: Complete blood count, coagulation profile, blood type and cross-match (for potential transfusions).
- Anesthetic Consultation: For complex cases or patients with significant comorbidities, a dedicated pre-operative anesthesia evaluation is crucial.
Pre-Operative Patient Positioning
Patient positioning significantly impacts surgical exposure, intraoperative blood loss, and neurological safety.
- Jackson Table or Relatable Spinal Frame: This specialized table allows for adequate abdominal decompression, minimizing epidural venous bleeding and improving visibility. It also facilitates chest expansion and maintains physiological sagittal contours.
-
Prone Position:
- Padding: Ensure adequate padding to all pressure points (cheeks, eyes, shoulders, hips, knees, ankles) to prevent nerve compression, skin breakdown, and ophthalmological complications.
- Head Positioning: Neutral head position, protected eyes (e.g., with gel pads or a horseshoe headrest), ensuring no direct pressure on the globes.
- Arms: Gently abducted and flexed to less than 90° at the shoulders and elbows, with palms down, secured to avoid brachial plexus traction injuries.
- Chest and Pelvic Supports: Ensure weight is distributed on the iliac crests and bilateral lateral chest walls, allowing the abdomen to hang freely. This minimizes intra-abdominal pressure, reduces venous congestion in the epidural plexus, and decreases blood loss.
- Lower Extremities: Knees slightly flexed with pillows underneath to prevent hyperextension, and ankles padded.
- Neuromonitoring Setup: Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs) are standard. Electrodes are meticulously placed by the neurophysiologist and baseline readings obtained before incision.
- Surgical Prep and Drape: Wide sterile field from the occiput to the sacrum, extending laterally to include the iliac crests if autograft harvest is anticipated.
Detailed Surgical Approach / Technique
Posterior Spinal Fusion (PSF) with segmental pedicle screw instrumentation is the gold standard for most forms of pediatric scoliosis requiring surgical correction. This section details a general approach; variations exist based on surgeon preference, curve type, and patient factors.
1. Incision and Exposure
- Midline Incision: Typically a straight midline incision directly over the spinous processes, extending from the proximal to distal fusion levels. The length is determined by the number of segments to be instrumented.
- Subperiosteal Dissection: Using electrocautery and Cobb elevators, the paraspinal muscles are meticulously stripped subperiosteally from the spinous processes, laminae, and transverse processes. This dissection proceeds laterally to the tips of the transverse processes, exposing the entire posterior elements.
- Level Identification: Accurate identification of spinal levels is critical. Intraoperative fluoroscopy or radiographs are used, often employing a marker (e.g., an 18-gauge needle) placed at the desired starting or ending vertebra. Counting from S1 or C7 can also be used as a cross-reference.
2. Pedicle Screw Placement
This is the most critical and potentially hazardous step. Pedicle screws offer superior biomechanical stability and correction capabilities compared to hooks or wires, allowing for 3D correction.
-
Entry Point Determination:
- Thoracic Vertebrae: The intersection of the pars interarticularis and the junction of the superior articular facet with the transverse process.
- Lumbar Vertebrae: The intersection of the vertical line through the lateral border of the superior articular facet and the horizontal line bisecting the transverse process.
- Pedicle Preparation: A high-speed burr or an awl is used to create a small cortical opening at the entry point.
- Probe/Sounding: A pedicle probe is carefully advanced through the pedicle into the vertebral body, feeling for resistance. The probe should advance smoothly. If bone is hard, remain cortical. If it pushes through spongy bone, an endosteal trajectory is confirmed. The trajectory is medial and slightly cephalad for thoracic pedicles, and slightly convergent for lumbar pedicles.
- Palpation: After creating the pedicle tract, a ball-tipped pedicle sounder is used to palpate all five walls (medial, lateral, superior, inferior, anterior) of the tract to confirm contained breach and prevent neurovascular injury. A breach warrants repositioning or using an alternative fixation point.
- Tapping and Screw Insertion: The pedicle tract is typically tapped (undersized compared to the screw) to create threads, especially in dense cortical bone. Polyaxial pedicle screws are then inserted, ensuring optimal length and diameter without breaching the anterior cortex or spinal canal.
-
Navigation/Robotics: In complex cases, or for routine enhanced accuracy, image-guided navigation or robotic systems can be employed to assist with precise pedicle screw placement.
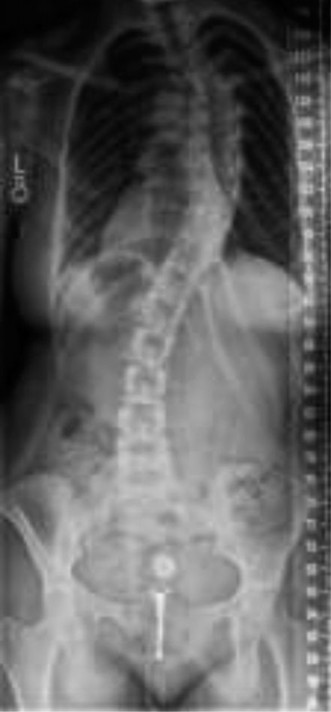 Intraoperative image demonstrating pedicle screw placement, crucial for segmental correction.
Intraoperative image demonstrating pedicle screw placement, crucial for segmental correction.
3. Deformity Correction
Once all screws are placed, two contoured rods (typically cobalt-chrome or titanium alloy) are used to correct the deformity. The sequence of correction varies by surgeon and curve type.
- Rod Introduction: The first rod is often placed on the concave side. It is contoured to the desired sagittal profile (thoracic kyphosis, lumbar lordosis) and then carefully seated into the screw heads.
-
Reduction Maneuvers:
- Direct Vertebral Rotation (DVR): By rotating the rod (or specific vertebral bodies via cantilever or segmental derotation tools), the apical vertebral rotation is corrected. This is a powerful 3D correction technique.
- Translation: Using reduction screws or persuaders, the vertebral bodies are translated towards the pre-contoured rod.
- Compression/Distraction: Compression across the convexity of the curve and distraction across the concavity can help reduce the curve and restore sagittal alignment. This must be performed carefully to avoid neurological injury.
- Cantilever Technique: A contoured rod is inserted into screws at one end of the construct and then sequentially cantilevered into the remaining screws to achieve correction.
- Second Rod Placement: The second rod, usually on the convex side, is then introduced. It often serves to enhance stability, provide additional corrective forces, or lock in the correction achieved with the first rod. Cross-links between the rods increase construct rigidity.
4. Fusion Bed Preparation
- Decortication: After correction, the posterior elements (laminae, facet joints, transverse processes) within the fusion segment are meticulously decorticated using a high-speed burr or osteotome. This creates a bleeding bony surface to promote arthrodesis.
-
Bone Grafting: Autogenous bone graft (local bone from spinous processes, laminae, transverse processes) is typically used. Allograft (demineralized bone matrix, cancellous chips) or synthetic bone graft substitutes can be supplemented, especially in cases where local autograft is insufficient. The graft material is packed thoroughly over the decorticated surfaces.
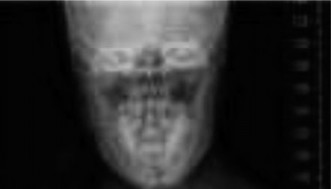 Post-operative AP radiograph illustrating satisfactory correction and instrumentation alignment.
Post-operative AP radiograph illustrating satisfactory correction and instrumentation alignment.
5. Final Steps
- Neuromonitoring Check: Before closure, neuromonitoring signals are re-checked to ensure no changes post-correction.
- Hemostasis: Meticulous hemostasis is achieved with electrocautery, topical hemostatic agents (e.g., fibrin sealant, gelatin sponges), and absorbable packing.
- Drainage: Suction drains (e.g., Jackson-Pratt) are often placed deep to the muscle layer to minimize hematoma formation.
-
Wound Closure: The paraspinal fascia is reapproximated with strong absorbable sutures. Subcutaneous tissue and skin are closed in layers.
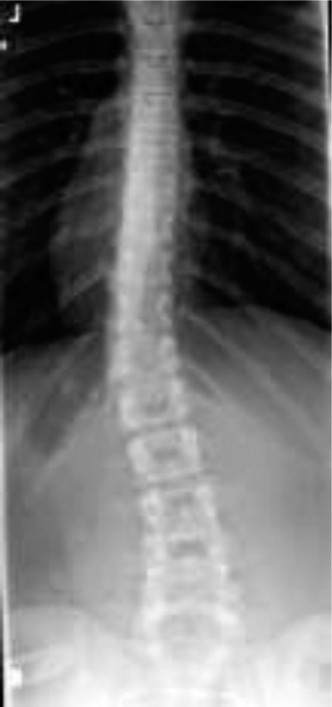 Post-operative lateral radiograph, emphasizing the restored sagittal profile and adequate instrumentation.
Post-operative lateral radiograph, emphasizing the restored sagittal profile and adequate instrumentation.
Complications & Management
Despite significant advances in surgical techniques and perioperative care, complications can occur in pediatric scoliosis surgery. Vigilance, early recognition, and prompt management are crucial.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (Approx.) | Salvage Strategies |
|---|---|---|
| Neurological Injury | 0.5-2% | Intraoperative: Wake-up test, rod removal, reduce distraction/compression, administer steroids, consider hypotension protocol. Postoperative: Immediate MRI, aggressive neurological assessment, re-exploration if compressive lesion identified, neurosurgical consultation. |
| Infection (SSI) | 1-5% | Superficial: Oral/IV antibiotics. Deep: Surgical debridement, washout, IV antibiotics (culture-specific), potentially implant removal if persistent. |
| Excessive Blood Loss | Variable | Pre-operative optimization (anemia correction), antifibrinolytics (TXA), controlled hypotension, cell saver, staged procedures, judicious use of blood products. |
| Implant-Related Complications | 2-10% | Screw Malposition: Revision/repositioning of misplaced screws (intraoperative or delayed). Rod Breakage/Pullout: Revision surgery with removal of failed hardware, potential extension of fusion, additional fixation. |
| Pseudarthrosis / Non-Union | 2-5% (lower with modern techniques) | Revision surgery with debridement of fibrous tissue, aggressive decortication, generous bone grafting (autograft/allograft), consideration of BMP, potentially new instrumentation or construct revision. |
| Proximal Junctional Kyphosis (PJK) / Failure (PJF) | 5-20% | PJK (non-symptomatic): Observation. PJF (symptomatic/progressive): Revision surgery with proximal extension of fusion, often with larger diameter rods and additional fixation points. Prevention: maintain appropriate sagittal balance, avoid over-distraction. |
| Distal Junctional Kyphosis (DJK) / Failure (DJF) | <5% | DJK (non-symptomatic): Observation. DJF (symptomatic/progressive): Revision surgery with distal extension of fusion. Prevention: careful selection of lowest instrumented vertebra (LIV). |
| Crankshaft Phenomenon | <1% (in skeletally immature) | Occurs in growing children after anterior-only fusion or posterior fusion that allows continued anterior growth. Salvage: Posterior instrumentation and fusion. Prevention: Combined anterior/posterior approach or posterior instrumentation to neutral and stable vertebrae. |
| Dural Tear / CSF Leak | 1-3% | Intraoperative repair (sutures, patch, fibrin sealant), lumbar drain for several days, bed rest. |
| Vascular Injury | Rare (<0.1%) | Immediate surgical repair, vascular surgery consultation. |
| Visceral Injury | Rare (<0.1%) | Surgical repair of injured viscus, appropriate specialty consultation. |
Post-Operative Rehabilitation Protocols
Post-operative management in pediatric scoliosis surgery aims to facilitate recovery, prevent complications, and integrate the patient back into normal activities while ensuring a solid fusion.
Immediate Post-Operative Period (Days 0-7)
- Intensive Care Unit (ICU): Most patients, especially after extensive procedures, spend 24-48 hours in the ICU for close monitoring of neurological status, vital signs, pain, and fluid balance.
- Pain Management: Multimodal analgesia is standard, typically involving patient-controlled analgesia (PCA) with opioids initially, transitioning to oral opioids and non-opioid medications (NSAIDs, acetaminophen) as tolerated. Regional blocks (e.g., erector spinae plane block) can be beneficial.
- Neurological Monitoring: Frequent neurological checks (motor and sensory) are paramount. Any new deficit warrants immediate investigation (MRI, neurological consultation).
-
Mobilization:
Early mobilization is encouraged to prevent pulmonary complications and deep vein thrombosis (DVT).
- Day 1: Log-roll to sitting at the edge of the bed, assisted ambulation to a chair.
- Day 2-3: Progressive ambulation in the room and hallway, increasing distance and independence.
- Pulmonary Care: Incentive spirometry, deep breathing exercises, and cough encouragement are vital to prevent atelectasis and pneumonia.
- Wound Care: Daily inspection for signs of infection (erythema, discharge, fever). Dressings are typically changed as needed. Drains are usually removed when output is minimal (<50 ml/24 hours).
- Diet: Clear liquids post-operatively, advancing to a regular diet as bowel function returns and nausea subsides.
Intermediate Post-Operative Period (Weeks 1-6)
- Hospital Discharge: Typically 4-7 days post-surgery, once pain is controlled with oral medication, the patient is ambulating independently, and there are no immediate post-operative complications.
-
Activity Restrictions:
- No bending, lifting (heavy objects), or twisting (BLT) for 3-6 months.
- Avoid strenuous activities, contact sports, and running.
- Limited sitting tolerance initially, encouraged to alternate sitting, standing, and walking.
- Return to school: Often within 2-4 weeks, with accommodations such as lighter backpack, modified seating, and reduced physical education.
- Pain Management: Gradual weaning from opioid medication, reliance on NSAIDs and acetaminophen.
- Physical Therapy: Focus on gentle core strengthening, posture education, and progressive ambulation. Avoid aggressive stretching or spinal manipulation.
- Bracing: Generally not required after PSF with modern segmental instrumentation, as the construct provides sufficient stability. Some surgeons may consider a soft brace for comfort or specific patient scenarios.
Long-Term Rehabilitation (Months 3-12 and beyond)
- Radiographic Follow-up: Regular post-operative radiographs (AP and lateral) at 3, 6, and 12 months to assess fusion status, instrumentation integrity, and maintenance of correction.
-
Activity Progression:
- 3-6 Months: Gradual return to light activities. No contact sports. Swimming and cycling often permitted.
- 6-12 Months: Progressive increase in activity level. Most non-contact sports may be considered after 6 months if fusion is evident.
- 12 Months+: Fusion is typically solid. Most restrictions are lifted. Return to full activity, including contact sports, is generally permitted after 12 months and radiographic confirmation of solid fusion. Individual assessment and discussion with the surgeon are essential.
- Pain Management: Residual pain is managed with NSAIDs or other non-pharmacological methods. Chronic pain should be investigated for pseudarthrosis or other complications.
- Body Mechanics and Posture: Continued emphasis on proper body mechanics and posture to minimize stress on the fused and adjacent segments.
Summary of Key Literature / Guidelines
The management of pediatric scoliosis is guided by extensive research and consensus guidelines from prominent organizations such as the Scoliosis Research Society (SRS), the American Academy of Orthopaedic Surgeons (AAOS), and the Pediatric Orthopaedic Society of North America (POSNA).
-
Scoliosis Research Society (SRS) Guidelines:
The SRS provides comprehensive guidance on the diagnosis, bracing, and surgical management of various forms of scoliosis. Their work emphasizes evidence-based decision-making for indications, surgical techniques, and outcome assessment. Key areas include:
- Adolescent Idiopathic Scoliosis (AIS): The classic Bracing in Adolescent Idiopathic Scoliosis Trial (BRAIST) demonstrated the efficacy of bracing in preventing progression in moderate curves. Surgical indications for AIS generally remain >45-50° in growing patients or curves causing significant progressive deformity or pain in skeletally mature patients.
- Early-Onset Scoliosis (EOS): Management of EOS prioritizes controlling the curve while maximizing spinal and thoracic growth to prevent thoracic insufficiency syndrome. Techniques like serial casting, growing rods (traditional or magnetically controlled), and vertebral body tethering (VBT) are employed, often in a staged approach. The early fusion debate highlights the critical balance between curve control and preserving lung development.
- Neuromuscular Scoliosis: Surgical intervention aims to improve sitting balance, prevent pelvic obliquity, and alleviate pain. Fusion often extends to the pelvis (sacral fixation) to achieve stability in patients with weak trunk musculature.
- Pedicle Screw Instrumentation: Numerous studies have established the superiority of all-pedicle screw constructs over hook-based or hybrid systems for achieving greater correction, improved segmental control, and lower rates of pseudarthrosis in AIS. This has become the standard of care.
- Sagittal Plane Restoration: Modern surgical philosophy emphasizes not only coronal plane correction but also meticulous restoration of physiological sagittal alignment (thoracic kyphosis and lumbar lordosis). Neglecting sagittal balance can lead to proximal/distal junctional problems, pain, and suboptimal functional outcomes. The importance of the C7 plumb line, T1 spinopelvic inclination, and pelvic parameters (pelvic incidence, sacral slope, pelvic tilt) is increasingly recognized in surgical planning.
- Neuromonitoring: The routine use of intraoperative neuromonitoring (SSEPs and MEPs) has significantly reduced the incidence of irreversible neurological deficits during scoliosis surgery. Protocols for managing neuromonitoring changes (e.g., wake-up test, temporary rod removal, blood pressure elevation, steroid administration) are well-established.
- Blood Management: Tranexamic acid (TXA) has become standard for reducing intraoperative blood loss and transfusion requirements in spinal deformity surgery, supported by high-level evidence. Controlled hypotension and cell salvage are also widely utilized strategies.
- Evidence on Long-Term Outcomes: Studies on adult outcomes of surgically treated pediatric scoliosis demonstrate generally good long-term function and patient satisfaction, though a small percentage may experience persistent back pain or require revision surgery. Continued research focuses on minimizing complications, optimizing fusion rates, and improving patient-reported outcomes.
The evolution of pediatric scoliosis management continues, driven by technological advancements, refined surgical techniques, and a deeper understanding of spinal biomechanics and growth. The overarching goal remains to provide durable correction and improve the quality of life for these young patients.
Clinical & Radiographic Imaging