Patient Presentation & History
A 3-year-old male presented to our pediatric orthopedic clinic with a chief complaint of progressive right lower extremity bowing and a noticeable limp. The deformity had been insidious in onset, first observed by his parents around 18 months of age, and had gradually worsened. He ambulated independently but with a noticeable Trendelenburg gait and occasional tripping. There was no history of trauma, specific injury, or acute pain episodes. The parents also reported the presence of multiple "birthmarks" on his torso and extremities since infancy.
His birth history was uncomplicated, full-term, via spontaneous vaginal delivery. Developmental milestones were met within normal limits. Past medical history was significant for a previously diagnosed mild scoliosis, currently managed with observation. There was no known family history of similar orthopedic conditions or genetic syndromes. On further questioning, the parents noted that the patient had undergone an ophthalmology evaluation at 2 years of age due to suspicious lesions on his iris, which were identified as Lisch nodules. No specific genetic testing had been performed prior to this presentation.
Clinical Examination
General Assessment
The patient was an alert, cooperative 3-year-old male. His vital signs were stable, and he appeared well-nourished. Height and weight were at the 50th percentile for age.
Musculoskeletal Inspection
- Lower Extremities: A significant anterolateral bowing deformity of the right tibia was evident, particularly in the distal and middle thirds. The apex of the deformity was prominent, with overlying skin appearing thin and taut. A subtle leg length discrepancy (LLD) was noted, with the right lower extremity appearing approximately 1-1.5 cm shorter.
- Skin: Multiple café-au-lait macules were observed over the trunk, bilateral upper extremities, and left lower extremity. Six macules greater than 0.5 cm in their greatest dimension were clearly identified, fulfilling a diagnostic criterion for Neurofibromatosis Type 1 (NF1). There was no axillary or inguinal freckling (Crowe's sign) appreciated at this time. No cutaneous or plexiform neurofibromas were palpated or visible.
- Gait: The patient ambulated with a right-sided limp, consistent with a Trendelenburg pattern and a subtle circumduction of the right lower limb, likely compensating for the LLD and angular deformity.
Palpation
- Deformity Site: Palpation over the anterolateral bowing of the right tibia revealed a firm, non-tender bony prominence. There was no gross instability or crepitus indicative of an acute fracture or frank pseudarthrosis, but the bone felt distinctly irregular and thickened at the apex of the curve.
- Neurovascular: Distal pulses (dorsalis pedis and posterior tibial) were symmetrical and strong bilaterally. Capillary refill was brisk in all digits. Sensation to light touch was intact throughout the right foot and ankle. Motor strength was graded 5/5 in all major muscle groups of the right lower extremity, with no evidence of foot drop.
Range of Motion (ROM)
- Hip and Knee: Full, pain-free range of motion was demonstrated bilaterally.
- Ankle: Dorsiflexion and plantarflexion were full and symmetrical. Subtle stiffness in the subtalar joint was noted on the right, but global ankle ROM was preserved.
Systemic Examination
- Neurological: Gross neurological examination was unremarkable.
- Ophthalmological: The historical finding of Lisch nodules was consistent with an NF1 diagnosis. No proptosis or signs of optic pathway glioma were observed on gross inspection.
- Cardiac: Regular rate and rhythm, no murmurs.
- Abdominal: Soft, non-tender, no organomegaly.
Imaging & Diagnostics
Radiographs
Initial anteroposterior (AP) and lateral radiographs of the right tibia and fibula were obtained. These revealed:
* Significant anterolateral bowing of the distal third of the right tibia.
* A classic "hourglass" deformity with marked sclerosis and narrowing of the medullary canal at the apex of the bowing.
* Evidence of cortical thickening and obliteration of the medullary canal at the site of maximal angulation, suggesting a dysplastic segment rather than a frank pseudarthrosis at this stage.
* No overt fracture line was present.
* The fibula was intact but appeared thin and bowed, conforming to the tibial deformity.
* An estimated 1.5 cm leg length discrepancy was noted on a scanogram of both lower extremities.
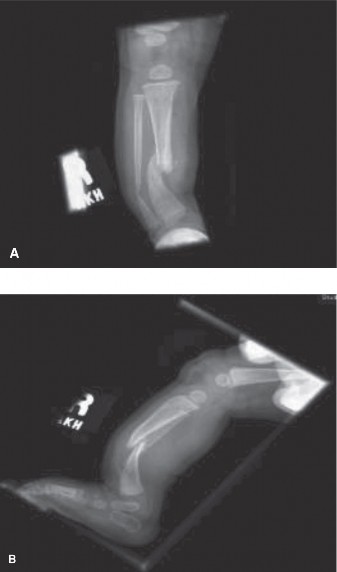
Figure 1: Pre-operative anteroposterior radiograph of the right lower extremity demonstrating severe anterolateral bowing of the distal tibia with sclerotic narrowing of the medullary canal and an "hourglass" deformity characteristic of congenital pseudarthrosis of the tibia (CPT) Type II/III.
Further imaging was performed to complete the diagnostic workup for potential NF1-related skeletal manifestations:
*
Full-spine AP/Lateral radiographs:
Mild thoracolumbar scoliosis, consistent with his previous diagnosis, without significant vertebral dysplasia.
*
Skull radiographs:
No evidence of calvarial dysplasia or sphenoid wing dysplasia.
Figure 2: Pre-operative lateral radiograph of the right lower extremity, further illustrating the anterolateral tibial bowing and the severe dysplastic changes at the apex of the deformity.
Further Diagnostics
- Genetic Consultation: Confirmed clinical diagnosis of Neurofibromatosis Type 1 based on fulfillment of diagnostic criteria (6+ café-au-lait macules >0.5cm, Lisch nodules, characteristic skeletal lesion). Genetic testing for NF1 gene mutation was discussed with the family for prognostic purposes and to confirm the diagnosis on a molecular level.
- Ophthalmology Review: Repeat ophthalmological examination confirmed Lisch nodules and excluded optic pathway gliomas.
- Neurology Consultation: Baseline MRI of the brain and spine was recommended to screen for neurofibromas, optic pathway gliomas, or other central nervous system manifestations.
Differential Diagnosis
The presentation of progressive anterolateral tibial bowing in a young child, coupled with characteristic skin stigmata and Lisch nodules, is highly suggestive of Congenital Pseudarthrosis of the Tibia (CPT) associated with Neurofibromatosis Type 1 (NF1). However, other conditions can present with lower extremity bowing, requiring careful differentiation.
| Feature | Congenital Pseudarthrosis of the Tibia (CPT) (associated with NF1) | Hypophosphatemic Rickets (X-linked dominant) | Infantile Blount's Disease (Tibia Vara) |
|---|---|---|---|
| Etiology | Dysplastic bone, often neurofibromatous tissue infiltration. Strong association with NF1 (50-80%). | Genetic defect (PHEX gene mutation) leading to renal phosphate wasting and impaired Vitamin D metabolism. | Growth disturbance of the medial proximal tibial physis. Multifactorial, associated with obesity, early walking. |
| Presentation Age | Infancy to early childhood (before 5-7 years). May present as bowing, fracture, or non-union. | Typically presents with bowing upon weight-bearing (1-3 years), short stature, muscle weakness. | Infancy (1-3 years) for infantile type. Progressive genu varum. |
| Bowing Character | Anterolateral bowing, typically unilateral. High risk of fracture and non-union. | Bilateral genu varum (bowed legs), symmetrical. | Unilateral (50-80%) or bilateral genu varum. |
| Associated Features | Café-au-lait macules, Lisch nodules, axillary freckling, neurofibromas (NF1 stigmata). | Short stature, bone pain, muscle weakness, dental abscesses. No skin lesions. | Obesity, early ambulation. No systemic findings. |
| Radiographic Findings | Anterolateral tibial bowing, sclerotic segment, "hourglass" deformity, medullary canal obliteration, impending or frank pseudarthrosis. | Widened, irregular, frayed growth plates (cupping, splaying) at metaphyses; diffuse osteopenia. | Medial metaphyseal beak, depression of medial tibial plateau, irregular physis. Increased metaphyseal-diaphyseal angle. |
| Laboratory Findings | Normal calcium, phosphate, alkaline phosphatase. Genetic testing for NF1. | Low serum phosphate, normal/low serum calcium, elevated alkaline phosphatase, elevated FGF23. | Normal calcium, phosphate, alkaline phosphatase. |
| Treatment | Complex surgical reconstruction (excision, grafting, stable fixation). High re-fracture/non-union rate. Long-term bracing. | Oral phosphate and active vitamin D (calcitriol) supplementation. Osteotomies for severe deformity. | Bracing for mild cases, guided growth, proximal tibial osteotomy for moderate to severe cases. |
Surgical Decision Making & Classification
The patient's presentation, characterized by progressive anterolateral tibial bowing, radiographic evidence of dysplastic changes with severe medullary canal narrowing, and the unequivocal presence of Neurofibromatosis Type 1, led to a diagnosis of Congenital Pseudarthrosis of the Tibia (CPT), specifically a pre-fracture (dysplastic) lesion.
Rationale for Operative Management
Despite the absence of a frank fracture, the progressive nature of the bowing, the characteristic radiographic findings of a sclerotic, hourglass deformity, and the strong association with NF1 indicate a high propensity for pathological fracture and subsequent non-union. Non-operative management with bracing alone has consistently failed to prevent fracture or achieve union in true CPT. Therefore, operative intervention was deemed necessary to:
1. Correct the angular deformity.
2. Induce union of the dysplastic segment.
3. Prevent impending fracture and subsequent difficult-to-treat pseudarthrosis.
4. Address the emerging leg length discrepancy.
Classification
The patient's condition best fits the Boyd Classification of Congenital Pseudarthrosis of the Tibia, Type III . This classification describes a lesion with an hourglass constriction of the tibia, usually associated with NF1, indicating an area of profound dysplasia that is highly prone to fracture and subsequent pseudarthrosis. While the patient had not yet sustained a complete fracture, the advanced dysplastic changes clearly placed him in this high-risk category, warranting early prophylactic surgical intervention.
Surgical Technique / Intervention
The primary goal of surgical intervention for CPT is to achieve bony union across the dysplastic segment, correct the deformity, restore alignment, and maintain limb length while minimizing the risk of refracture. Given the high re-fracture rate and the challenges associated with CPT in NF1 patients, a robust and long-lasting construct is essential.
Pre-operative Planning
Detailed templating using the radiographs was performed to determine the extent of the dysplastic segment, the required length of resection, and the appropriate size and length of intramedullary fixation. The goal was to resect the entire dysplastic segment and achieve fixation extending well into healthy bone proximally and distally, ideally crossing the ankle joint for enhanced stability in young children.
Surgical Procedure
- Patient Positioning: The patient was positioned supine on a radiolucent table. A high thigh tourniquet was applied to the right leg. The entire limb was prepped and draped freely to allow for full manipulation and intraoperative fluoroscopy.
- Surgical Approach: A longitudinal anteromedial incision was made over the apex of the bowing, extending proximally and distally to encompass the entire dysplastic segment.
- Soft Tissue Dissection: The subcutaneous tissues and periosteum were carefully incised. Care was taken to identify and protect the anterior neurovascular structures, which can be intimately involved with neurofibromatous tissue in NF1 cases. Any surrounding dysplastic or neurofibromatous soft tissue was meticulously excised.
- Excision of Dysplastic Segment: The dysplastic and sclerotic bone segment at the apex of the bowing was sharply excised using an osteotome and burr. This involved resecting approximately 2.5 cm of tibial shaft to ensure removal of all unhealthy bone until healthy, bleeding cortical bone with an open medullary canal was identified proximally and distally. The corresponding fibula, which was also bowed and often dysplastic, was not directly addressed in this primary procedure but its role in recurrence was noted.
- Preparation for Grafting: The medullary canals of the proximal and distal tibial segments were reamed to prepare for intramedullary nail insertion and to encourage osteoinductive potential. Extensive decortication of the cortical surfaces adjacent to the osteotomy site was performed to maximize the surface area for graft incorporation and promote healing. This involved creating small cortical windows and roughening the bone with a burr.
- Bone Grafting: A significant volume of autologous cancellous bone graft was harvested from the ipsilateral posterior iliac crest. This graft material was packed circumferentially around the osteotomy site and within the reamed medullary canals to augment union. In addition, a demineralized bone matrix (DBM) putty was mixed with the autograft to enhance osteoinductivity.
-
Internal Fixation:
- Intramedullary Nailing: A small diameter (2.5 mm) flexible titanium intramedullary nail (e.g., Ender nail or Fassier-Duval system if available for this age group) was retrograde inserted through a distal medial malleolar portal, traversing the ankle joint and crossing the pseudarthrosis site, extending well into the proximal tibia. This provided axial stability and alignment. A second, parallel flexible nail was attempted but not feasible due to the small bone diameter. Instead, a single, slightly larger (3.0 mm) titanium elastic nail was selected for better rotational control.
- External Fixation: Given the high risk of non-union and refracture in CPT, a monolateral external fixator (e.g., Orthofix, or a mini-Ilizarov frame) was applied. Two proximal pins were placed into the healthy proximal tibia, and two distal pins were placed into the healthy distal tibia, incorporating the calcaneus for robust stability across the ankle joint, which was necessary to protect the relatively weak distal segment from rotational and bending forces. This provided compression across the osteotomy site and rigid multi-planar stability. The external fixator was chosen over plate fixation due to the poor bone quality, high mechanical demands, and the ability to dynamically adjust compression and address potential leg length discrepancies during healing.
- Wound Closure: The wound was copiously irrigated. The deep fascia was closed over drains, followed by subcutaneous tissue and skin closure in layers.
Final Construct
The final construct involved a rigid intramedullary nail extending from the distal tibial metaphysis (crossing the ankle) into the proximal tibial metaphysis, augmented by an external fixator providing stable compression across the osteotomy and protecting the entire tibial segment. This "hybrid" approach is often preferred for CPT due to its superior stability and biologic environment compared to internal fixation alone.
Post-Operative Protocol & Rehabilitation
Post-operative management for CPT is as critical as the surgical intervention itself, focusing on promoting union, maintaining alignment, and preventing complications.
Immediate Post-Operative Period (Day 0-7)
- Pain Management: Multimodal analgesia including regional blocks, NSAIDs, and opioid analgesics as needed.
- Wound Care: Daily pin site care with chlorhexidine solution to prevent infection. Regular inspection of the surgical incision.
- Weight-Bearing: Strictly non-weight-bearing on the affected limb. The external fixator provides protection but direct weight-bearing is deferred until radiographic signs of early union are evident.
- Elevation: Limb elevated to reduce swelling.
- Physiotherapy: Gentle, passive range of motion exercises for the knee and ipsilateral hip. Active ankle dorsiflexion and plantarflexion, within the limits of the frame, to prevent stiffness and deep vein thrombosis.
Intermediate Post-Operative Period (Weeks 1-12)
- Radiographic Monitoring: Serial AP and lateral radiographs of the tibia and fibula at 4-6 week intervals to assess for early signs of union, maintenance of alignment, and pin integrity.
- External Fixator Adjustments: If a dynamic external fixator (e.g., Ilizarov) was used, gradual compression across the osteotomy site might be initiated, followed by a neutral or slight distraction phase if necessary for limb lengthening, as per surgeon's discretion.
- Weight-Bearing Progression: Once early callus formation and stability are observed on radiographs (typically 8-12 weeks), protected weight-bearing with crutches or a walker may be gradually introduced, increasing as union progresses.
- Rehabilitation: Continued physical therapy focused on quadriceps strengthening, hip abductor strength, and progressive gait training with assistance. Active ankle ROM exercises, emphasizing eversion and inversion.
Long-Term Management (Months 3-18+)
- Union Assessment: Radiographic union can be protracted in CPT, often taking 6-12 months or more. Clinical assessment for tenderness and stability at the fracture site is correlated with radiographic findings.
- External Fixator Removal: Once solid radiographic and clinical union is achieved, the external fixator is removed in the clinic or operating room, depending on patient age and complexity.
- Intramedullary Nail Removal: The intramedullary nail is typically left in situ until skeletal maturity or until growth plate concerns mandate removal. It provides crucial long-term protection against refracture.
- Orthotic Management: Following fixator removal, a custom-molded ankle-foot orthosis (AFO) or knee-ankle-foot orthosis (KAFO) is prescribed for long-term protection of the tibia, often for several years or until skeletal maturity. This is critical to prevent refracture, which remains a significant risk.
- Follow-up: Regular clinical and radiographic follow-up every 3-6 months for several years to monitor for refracture, progressive deformity, leg length discrepancy, and growth plate issues.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Early Diagnosis: Recognize the association between anterolateral tibial bowing and Neurofibromatosis Type 1. The presence of café-au-lait macules in a child with tibial bowing should immediately raise suspicion for CPT, even in the absence of a frank fracture. Early intervention on a dysplastic segment (Boyd Type II or III) often yields better results than treating established pseudarthrosis.
- Aggressive Debridement: Complete excision of all dysplastic bone and surrounding fibrous/neurofibromatous tissue is paramount. Leaving dysplastic tissue is a leading cause of non-union or refracture. Expose healthy, bleeding bone at the ends.
- Extensive Decortication: Maximize the surface area for bone graft incorporation by decorticating the periosteal surface of the healthy bone segments adjacent to the osteotomy. This promotes a robust biologic response.
- Robust Bone Grafting: Utilize ample autologous cancellous bone graft, ideally from the iliac crest. Supplementation with allograft, demineralized bone matrix (DBM), or even vascularized fibular graft (for recalcitrant cases) should be considered.
- Stable Fixation – The Hybrid Approach: The combination of an intramedullary nail and an external fixator (e.g., Ilizarov or monolateral frame) offers superior stability compared to either method alone. The IMN provides axial and rotational control along the length of the tibia, while the external fixator allows for compression across the osteotomy and additional multi-planar stability. The nail should ideally span the entire tibia, often crossing the ankle joint in young children to prevent distal fracture.
- Long-Term Protection: After union is achieved and external fixation removed, long-term protective bracing (AFO/KAFO) is essential for several years or until skeletal maturity to shield the 'neo-tibia' from high-stress activities and reduce refracture rates.
- Multidisciplinary Approach: Management of NF1-associated CPT requires a team approach, including pediatric orthopedics, genetics, neurology, ophthalmology, and physical therapy.
- Managed Expectations: Families must be counselled regarding the high refracture rates (up to 50% or more even with optimal treatment), the potential for multiple surgeries, and the likelihood of residual leg length discrepancy and angular deformities.
Pitfalls
- Inadequate Excision/Debridement: Failure to remove all dysplastic and non-osteogenic tissue is a primary cause of non-union or early refracture.
- Insufficient Fixation: An unstable construct that fails to provide rigid compression and multi-planar stability will lead to early failure, loss of reduction, or refracture. Plate fixation alone is generally contraindicated for primary CPT due to high failure rates.
- Premature Fixation Removal: Removing the external fixator or intramedullary nail too early, before solid radiographic union is unequivocally demonstrated, significantly increases the risk of refracture.
- Ignoring the Fibula: While the tibia is the primary focus, the fibula is often involved in the dysplastic process. Aggressive shortening of the tibia without considering fibular stability can lead to proximal migration of the fibula and distal ankle valgus or malunion.
- Growth Plate Injury: Care must be taken during pin insertion for external fixation and intramedullary nail placement to avoid damage to open physes, especially at the distal tibia and proximal fibula.
- Pin Tract Infection: A common complication of external fixation, requiring meticulous pin site care and prompt antibiotic treatment if infection arises. Deep infection can compromise bone healing.
- Leg Length Discrepancy (LLD): While initial correction is achieved, CPT limbs often have impaired growth potential, leading to progressive LLD over time, which may require subsequent lengthening procedures.
- Ankle Deformity: Residual ankle valgus or varus is common due to altered loading mechanics and residual dysplasia. This may necessitate future osteotomies.
- Recurrence: Despite aggressive management, CPT has a high rate of recurrence, emphasizing the chronic nature of the disease and the need for vigilant, long-term follow-up.
