Introduction & Epidemiology
Multiple Hereditary Exostoses (MHE), also known as diaphyseal aclasia or osteochondromatosis, is an autosomal dominant skeletal disorder characterized by the formation of multiple osteochondromas. These benign cartilage-capped bony outgrowths typically arise from the juxta-epiphyseal region of long bones, particularly affecting the metaphyses, and grow away from the adjacent joint. The condition is genetically heterogeneous, primarily linked to mutations in either the EXT1 gene (8q24.1) or EXT2 gene (11p11-p12), which encode glycosyltransferases involved in heparan sulfate proteoglycan synthesis. These proteoglycans are critical for normal chondrocyte differentiation and growth plate function.
The incidence of MHE is estimated to be approximately 1 in 50,000 live births, making it the most common hereditary bone disorder. Penetrance is nearly 100%, though expressivity can vary significantly, even within families. Patients present with a wide spectrum of clinical manifestations, ranging from a few asymptomatic exostoses to widespread involvement leading to severe skeletal deformities, pain, and functional limitations. Exostoses typically develop during childhood and cease to grow with skeletal maturity. However, their presence can disrupt normal endochondral ossification, leading to a constellation of secondary deformities. Lifelong monitoring is crucial for detecting and managing complications, including neurological or vascular impingement, mechanical irritation, growth disturbances, and the most serious risk: malignant transformation into secondary peripheral chondrosarcoma.
Surgical Anatomy & Biomechanics
Exostoses in MHE are true osteocartilaginous lesions, meaning they are composed of a cartilage cap that undergoes endochondral ossification, forming a bony stalk continuous with the underlying parent bone. They originate from ectopic growth plate cartilage cells that herniate from the physis. This pathological growth mechanism explains why they grow away from the joint and why they cease to enlarge at skeletal maturity, as the ectopic physis itself fuses.
The predilection sites for exostoses include:
*
Distal femur and proximal tibia/fibula:
Common for genu valgum, ankle valgus, and leg length discrepancy.
*
Distal radius and ulna:
Leads to characteristic forearm deformities (Madelung-like deformity), ulnar shortening, radial bowing, increased interosseous space, and wrist subluxation.
*
Proximal humerus:
Can restrict shoulder range of motion.
*
Pelvis and scapula:
Often large, sessile lesions that can cause pain or impingement.
*
Ribs, vertebrae, and small bones of the hands and feet:
Less common but can cause specific localized problems.
The biomechanical consequences of exostoses are multifaceted:
1.
Growth Plate Tethering and Deformity:
Exostoses located near growth plates can cause focal physeal damage or tethering, leading to angular deformities and limb length discrepancies.
*
Genu Valgum:
A common finding, resulting from asymmetrical growth inhibition of the distal femoral or proximal tibial physis, often aggravated by exostoses on the medial aspects.
*
Forearm Deformity:
Ulnar growth arrest (due to exostoses from the distal ulna or radial head) is characteristic, leading to relative radial overgrowth, radial bowing, positive ulnar variance, and progressive subluxation of the distal radio-ulnar joint (DRUJ) with pain and restricted forearm rotation.
*
Ankle Valgus:
Exostoses arising from the distal tibia or fibula can cause premature fusion or tethering of the respective physis, leading to ankle valgus due to differential growth.
2.
Mechanical Irritation and Impingement:
Exostoses can impinge on adjacent muscles, tendons, ligaments, or joint capsules, causing pain, bursitis, tendinitis, or restricted joint motion.
3.
Neurovascular Compression:
Exostoses can compress peripheral nerves (e.g., common peroneal nerve at the fibular neck, ulnar nerve at the elbow) or vascular structures (e.g., popliteal artery behind the knee), leading to neuropathy, claudication, or even acute vascular compromise.
4.
Fracture:
While rare, an exostosis stalk can fracture following trauma, causing acute pain and swelling.
5.
Malignant Transformation:
The most significant long-term risk. The cartilage cap of an exostosis can undergo malignant transformation into a secondary peripheral chondrosarcoma. This typically occurs in adulthood, with a reported lifetime risk of 0.5% to 5%. Large, centrally located, or progressively growing exostoses, particularly those with a thick cartilage cap (>2 cm in adults), are suspicious.
Monitoring for these risks involves regular clinical examination, assessment of range of motion, neurovascular status, and standing radiographs to evaluate for angular deformities and limb length discrepancies.
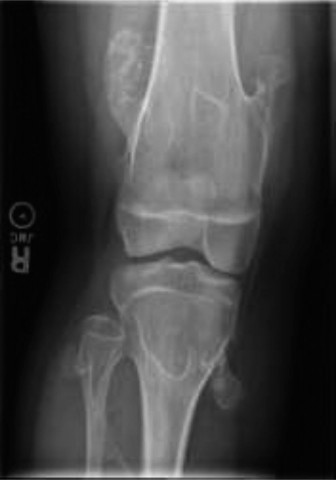
Figure: Representative image of a large exostosis, demonstrating its potential for significant mass effect and distortion of normal skeletal architecture.
Indications & Contraindications
The management of MHE is primarily guided by the presence and severity of symptoms and deformities. A "watch and wait" approach is common for asymptomatic lesions, but close monitoring is essential.
Operative Indications
Surgical intervention is indicated for:
*
Pain:
Persistent pain unresponsive to conservative measures, particularly if related to mechanical irritation, impingement on soft tissues, or bursitis.
*
Neurological Symptoms:
Compression neuropathy (e.g., foot drop due to peroneal nerve compression, ulnar neuropathy) or radiculopathy from spinal involvement.
*
Vascular Compromise:
Arterial or venous compression leading to claudication, reduced pulses, or venous congestion.
*
Malignant Transformation:
Suspicion or confirmed diagnosis of secondary peripheral chondrosarcoma (indicated by rapid growth, new onset of pain in a previously asymptomatic lesion, increased size of cartilage cap on imaging, cortical destruction, or soft tissue mass).
*
Progressive Angular Deformity:
Significant and worsening genu valgum, ankle valgus, or forearm deformities (e.g., Madelung-like deformity) causing functional impairment or cosmetic concern.
*
Limb Length Discrepancy (LLD):
Symptomatic LLD requiring correction.
*
Restricted Joint Motion:
Exostoses causing mechanical block to joint movement.
*
Fracture of the Exostosis Stalk:
Painful and often requires excision.
*
Functional Impairment:
Any exostosis significantly interfering with activities of daily living, sleep, or shoe wear.
Non-Operative Indications
Non-operative management includes:
*
Asymptomatic Exostoses:
No pain, no functional impairment, no neurovascular compromise, no significant deformity.
*
Stable Minor Deformities:
Small angular deformities or mild limb length discrepancies that do not cause functional limitations and are not progressing.
*
Observation for Malignant Transformation:
Routine surveillance for all patients with MHE, especially for lesions that are large, sessile, or in high-risk areas (e.g., pelvis, shoulder girdle, proximal femur). Regular clinical examination and serial radiographs are critical. MRI is indicated for suspicious lesions.
*
Conservative Pain Management:
NSAIDs, physical therapy, activity modification for mild, intermittent pain.
Table: Operative vs. Non-Operative Indications
| Indication Type | Operative | Non-Operative |
|---|---|---|
| Symptoms | Persistent, severe pain; Neurological compression; Vascular compromise | Asymptomatic lesions; Mild, intermittent pain managed conservatively |
| Deformity | Progressive angular deformity (valgus/varus); Significant limb length discrepancy; Joint impingement/blockage | Stable minor deformities not causing functional limitation |
| Pathology | Suspicion/Confirmation of malignant transformation; Fracture of exostosis stalk | Asymptomatic, stable exostoses (requiring surveillance for malignant transformation) |
| Function | Significant functional impairment in ADLs, gait, range of motion | No significant functional impairment |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is paramount in MHE surgery due to the often complex anatomical distortions and the proximity of neurovascular structures.
Imaging
- Plain Radiographs: Standard AP and lateral views of the affected area are essential. Full-length standing radiographs (scanograms or EOS imaging) are critical for evaluating limb alignment (mechanical axis deviation) and limb length discrepancies, particularly in the lower extremities. Hand and wrist radiographs are crucial for forearm deformities.
- Computed Tomography (CT): Recommended for complex deformities, spinal involvement, pelvic or scapular lesions, or when precise 3D anatomy is required for osteotomy planning. CT angiography may be indicated if vascular impingement is suspected.
- Magnetic Resonance Imaging (MRI): Indispensable for evaluating soft tissue involvement, defining the cartilage cap thickness, assessing neurovascular compression, and most importantly, for suspected malignant transformation. A cartilage cap thickness >2 cm in adults is highly suspicious for chondrosarcoma and warrants careful evaluation, potentially with biopsy.
- Ultrasound: Can be used to assess cartilage cap thickness in superficial lesions and to evaluate for vascular flow compromise or nerve compression.
Growth Assessment
In pediatric patients, assessment of remaining growth potential (e.g., using bone age, Green-Anderson charts, or Moseley straight-line graph) is critical, especially when planning growth modulation procedures (hemiepiphysiodesis) or considering the timing of definitive osteotomies.
Surgical Planning
- Excision of Exostoses: Map the exact location relative to neurovascular structures. Plan incisions to allow adequate exposure while minimizing soft tissue damage and cosmetic impact.
-
Deformity Correction:
- Growth Modulation: For angular deformities in growing children with sufficient remaining growth, guided growth techniques (hemiepiphysiodesis using plates or screws) are effective. Plan the precise location for plate placement based on mechanical axis deviation.
- Corrective Osteotomy: For established angular deformities or in skeletally mature patients. Plan the osteotomy level, wedge resection/opening, and fixation method (internal fixation with plates/screws or external fixation). Consider biplanar corrections for complex deformities.
- Limb Lengthening: For significant limb length discrepancy, plan gradual lengthening with external fixators (monorail, circular frames) or internal lengthening nails, considering the rate and total amount of lengthening required.
- Forearm Deformity: Often requires a combination of ulnar lengthening, radial osteotomy, and DRUJ stabilization.
Patient Positioning
- Supine: Most common for lower extremity and anterior approach forearm surgeries. Allows for limb manipulation.
- Prone: For posterior thigh or calf exostoses, or posterior spinal lesions.
- Lateral Decubitus: For hip, pelvis, or shoulder girdle lesions.
- Arm Board/Hand Table: For upper extremity procedures, ensuring proper support and access.
- Tourniquet: Typically used for extremity surgery to provide a bloodless field, aiding in precise dissection and neurovascular protection. Ensure proper padding and inflation pressure.
- Fluoroscopy: Essential for accurate osteotomy cuts, plate/screw placement, and confirming correction.
Detailed Surgical Approach / Technique
Surgical intervention in MHE patients typically involves either direct exostosis excision or osteotomies for deformity correction, or a combination thereof.
1. Exostosis Excision (Exostosectomy)
Principles:
*
Complete Excision:
The goal is complete removal of the cartilage cap and the underlying bony stalk to prevent recurrence. Incomplete removal of the cartilage cap is the primary cause of recurrence.
*
Neurovascular Protection:
Meticulous dissection is paramount, as exostoses often abut or encase vital neurovascular structures. Identification and protection of these structures should be the first priority.
*
Physeal Sparing:
In children, care must be taken to avoid damaging the adjacent physis to prevent iatrogenic growth arrest or deformity.
General Steps:
1.
Incision:
Plan an incision that provides adequate exposure of the exostosis and surrounding structures, ideally longitudinal and in line with skin tension lines, avoiding crossing joints perpendicularly.
2.
Dissection:
* Carefully incise skin and subcutaneous tissue.
* Identify and retract muscle planes. Use internervous planes where possible (e.g., between biceps brachii and brachialis for anterior humerus, between tibialis anterior and gastrocnemius for anterior tibia).
*
Crucial Step:
Systematically identify all adjacent nerves and vessels (e.g., common peroneal nerve at the fibular neck, popliteal vessels behind the knee, ulnar nerve at the elbow, radial artery in the forearm). Trace them proximal and distal to the exostosis to ensure their safety.
* The periosteum overlying the exostosis is incised.
3.
Excision:
* Once the exostosis is adequately exposed and neurovascular structures are protected, use an osteotome or burr to excise the bony stalk at its base, flush with the cortical surface of the parent bone.
* Ensure complete removal of the cartilage cap. The excised specimen should be sent for histological examination to confirm benignity or identify malignant transformation.
* For large defects, bone wax can be applied to control bleeding. Bone grafting is generally not required for simple exostosectomy.
4.
Hemostasis and Closure:
Achieve meticulous hemostasis. Irrigate the wound. Close in layers, ensuring no dead space. Drains may be used for larger excisions.
Specific Considerations:
*
Spinal Exostoses:
Extremely challenging due to proximity to the spinal cord and nerve roots. Often requires neurosurgical co-management. Laminectomy or laminotomy may be necessary.
*
Pelvic/Scapular Exostoses:
Can be very large and deep, requiring extensive muscle dissection and careful attention to major vessels and nerves (e.g., sciatic nerve, femoral vessels, brachial plexus).
*
Forearm:
Exostoses from the distal ulna often tether the physis. Excision here must be precise to avoid damaging the distal ulnar growth plate further.
2. Deformity Correction
a. Growth Modulation (Hemiepiphysiodesis)
- Indication: Growing children with angular deformities (e.g., genu valgum, ankle valgus) and sufficient remaining growth.
- Technique: Percutaneous placement of a tension band plate (e.g., 8-plate) or staples across the physis on the convex side of the deformity. This temporarily tethers growth on one side, allowing the concave side to "catch up" and correct the alignment.
- Removal: Plates are typically removed once correction is achieved or when skeletal maturity is reached.
b. Corrective Osteotomy
- Indication: Established angular deformities in skeletally mature patients or significant deformities in children unsuitable for growth modulation.
- Principles: Precise bone cuts to realign the mechanical axis. Can be opening wedge, closing wedge, or dome osteotomies.
-
General Steps (e.g., Distal Femoral Varus Osteotomy for Genu Valgum):
- Approach: Medial or lateral approach depending on the desired correction and fixation.
- Osteotomy: Using fluoroscopic guidance, make a precise osteotomy cut (e.g., supracondylar distal femur). A Gigli saw or oscillating saw can be used. Ensure neurovascular protection during cuts.
- Correction: Correct the angular deformity, often guided by pre-operative planning and intra-operative navigation or alignment guides. Ensure patellar tracking is optimized.
- Fixation: Secure the osteotomy with internal fixation (e.g., locking plates, intramedullary nails) or external fixation. Strong fixation is crucial for early mobilization.
c. Limb Lengthening
- Indication: Significant limb length discrepancy (>2-3 cm) with functional implications.
- Technique: Osteotomy (cortical or metaphyseal) followed by gradual distraction using an external fixator (e.g., Ilizarov, Taylor Spatial Frame) or an internal lengthening nail.
- Rate: Typically 0.75-1.0 mm/day, adjusted based on bone formation and soft tissue tension.
- Monitoring: Regular radiographs to assess regenerate bone formation and check for complications (e.g., nerve palsy, joint contractures).
d. Forearm Deformity Correction (Madelung-like Deformity)
- Pathology: Often involves ulnar shortening and distal radial bowing with dorsal subluxation of the ulna at the DRUJ.
-
Techniques:
- Distal Ulnar Exostosis Excision: If present and tethering growth.
- Ulnar Lengthening Osteotomy: For significant ulnar shortening, often performed with an external fixator or internal plate with gradual distraction.
- Distal Radial Closing Wedge Osteotomy: To correct radial bowing and redirect the articular surface.
- DRUJ Stabilization: Procedures may be needed for chronic instability or painful impingement.
- Complexity: These corrections are highly complex and require careful pre-operative planning and experienced hand/upper extremity surgeons.
Complications & Management
MHE surgery is associated with a specific set of complications, ranging from general surgical risks to those inherent to the condition and the reconstructive procedures.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence | Management / Salvage Strategy | Salvage Strategy control |
